Organometallics
Article
1
129.6, 129.5, 129.3 (d, J = 6.9 Hz), 128.9 (d, J = 4.3 Hz), 123.9, 122.9,
59.5, 50.1, 36.2. 31P NMR (162.0 MHz, DMSO-d6) δ −14.7, −144.2
(m, J = 712.8 Hz). HRMS (ESI-TOF) m/z: [M − PF6]+ Calcd for
C25H25N3P 398.1786. Found 398.1780.
2-(m-Tolyl)benzoxazole (6b). H NMR (CDCl3, 400.1 MHz) δ
8.10 (s, 1H), 8.05 (d, 1H, J = 7.6 Hz), 7.7−7.8 (m, 1H), 7.58 (m, 1H),
7.40 (t, 1H, J = 7.6 Hz), 7.3−7.4 (m, 3H), 2.46 (s, 3H). 13C NMR
(CDCl3, 100.6 MHz) δ 163.3, 150.7, 142.1, 138.8, 132.4, 128.9, 128.2,
127.0, 125.0, 124.8, 124.6, 120.0, 110.6, 21.4.
Synthesis of ({3-{2-{[2-(Diphenylphosphino-κP)-
benzylidene]amino}ethyl}-1-methylimidazole-2-yl)(bischloro)
Palladium(II) (4). Compound 2 (2 mmol, 867 mg), silver oxide (1
mmol, 232 mg), and dichloromethane (20 mL) were added into a 2-
necked flask successively in glovebox. After the mixture was stirred
under darkness for 2 h, it was filtered with Celite. Furthermore,
PdCl2(CNMe)2 (2 mmol, 518 mg) was added into the filtrate and
stirred for another 3 h at ambient temperature. Then, the reaction
mixture was filtered on Celite, and ether (30 mL) was added to form
pale yellow solid. The solid was filtered, washed with ether (5 mL ×
2), and dried under vacuum to give compound 4 (550 mg, 48% yield).
Single crystals suitable for X-ray diffraction measurement were
obtained by diffusing ether into DMF solution of complex 4. 31P
2-(o-Tolyl)benzoxazole (6c). 1H NMR (CDCl3, 400.1 MHz) δ 8.17
(d, 1H, J = 7.2 Hz), 7.80 (m, 1H), 7.60 (m, 1H), 7.3−7.5 (m, 5H),
2.82 (s, 3H). 13C NMR (CDCl3, 100.6 MHz) δ 163.4, 150.3, 142.1,
138.9, 131.8, 130.9, 130.0, 126.3, 126.1, 125.0, 124.4, 120.2, 110.5,
22.2.
1
2-(4-Isopropylphenyl)benzoxazole (6d). H NMR (CDCl3, 400.1
MHz) δ 8.18 (d, 2H, J = 8.4 Hz), 7.77 (m, 1H), 7.58 (m, 1H), 7.3−7.4
(m, 4H), 2.99 (m, 1H), 1.30 (d, 6H, J = 6.8 Hz). 13C NMR (CDCl3,
100.6 MHz) δ 163.3, 152.9, 150.7, 142.2, 127.8, 127.1, 124.9, 124.7,
124.5, 119.9, 110.5, 34.3, 23.8. HRMS (ESI-TOF) m/z: [M + H]+
Calcd for C16H16NO 238.1232. Found 238.1215.
2-(3,5-Dimethylphenyl)benzoxazole (6e). 1H NMR (CDCl3, 400.1
MHz) δ 7.89 (s, 2H), 7.77 (m, 1H), 7.57 (m, 1H), 7.3−7.4 (m, 2H),
7.17 (s, 1H), 2.42 (s, 6H). 13C NMR (CDCl3, 100.6 MHz) δ 163.5,
150.7, 142.1, 138.6, 133.4, 126.9, 125.4, 125.0, 124.5, 119.9, 110.5,
21.3.
1
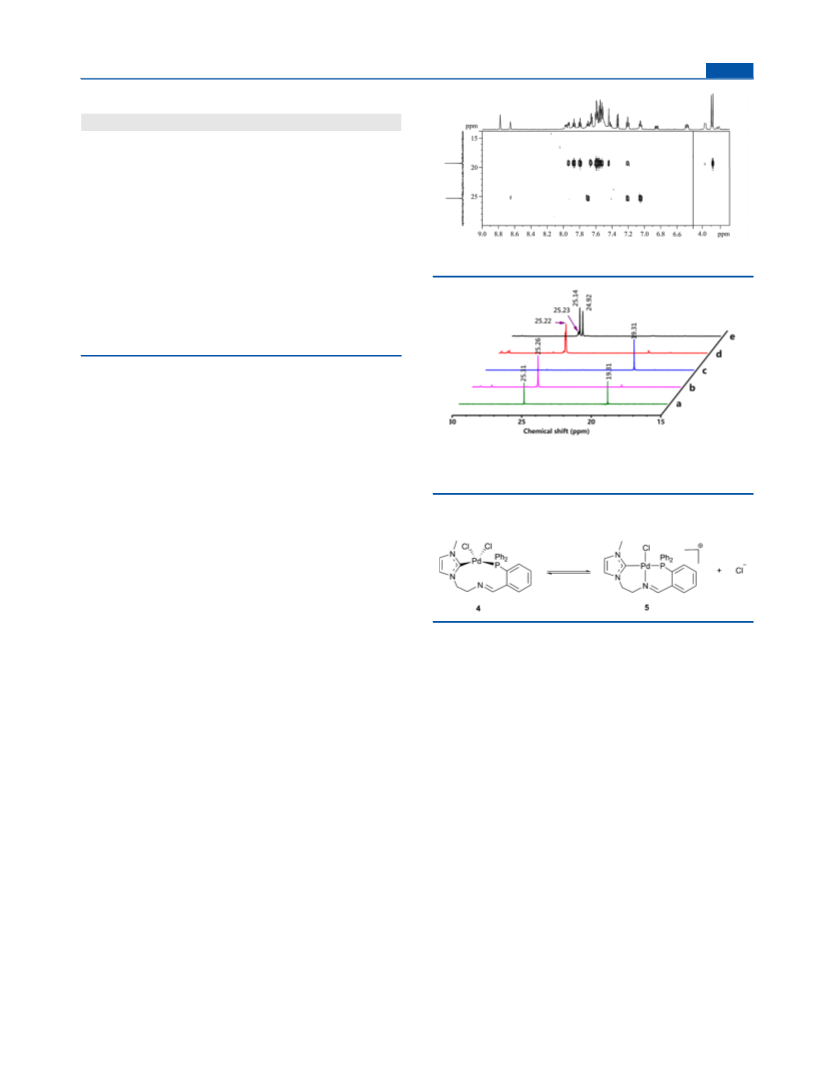
NMR (162.0 MHz, DMSO-d6) δ 25.3, 19.3. H NMR was mixed
spectra. HRMS (ESI-TOF) m/z: [M
−
Cl]+ Calcd for
C25H24ClN3PPd 538.0431. Found 538.0421; [M − 2Cl]2+ Calcd for
C25H24N3PPd 251.5372. Found 251.5362. NMR data for complex 4 +
1
2-(4-Fluorophenyl)benzoxazole (6f). 1H NMR (CDCl3, 400.1
MHz) δ 8.2−8.3 (m, 2H), 7.76 (m, 1H), 7.57 (m, 1H), 7.3−7.4 (m,
2H), 7.21 (t, 2H, J = 8.4 Hz). 13C NMR (CDCl3, 100.6 MHz) δ 164.8
(d, J = 251.0 Hz), 162.2, 150.8, 142.0, 129.9 (d, J = 8.9 Hz), 125.2,
124.7, 123.5 (d, J = 3.1 Hz), 120.0, 116.2 (d, J = 22.2 Hz), 110.6.
10 equiv of LiCl: H NMR (400.1 MHz, DMSO-d6) δ 8.68 (s, 1H),
7.97 (m, 1H), 7.67 (m, 1H), 7.4−7.6 (m, 8H), 7.17 (m, 1H), 7.03 (m,
2H), 6.79 (m, 1H), 6.42 (m, 2H), 4.43 (m, 1H), 4.26 (m, 1H), 3.87 (s,
3H), 3.80 (m, 1H), 3.25 (m, 1H). 13C NMR (100.6 MHz, DMSO-d6)
δ 160.9 (d, J = 9.5 Hz), 159.6, 137.3 (d, J = 5.0 Hz), 135.3 (d, J = 3.4
Hz), 134.7 (d, J = 61.3 Hz), 134.2 (d, J = 25.2 Hz), 133.5 (d, J = 7.3
Hz), 131.4, 131.1, 130.5 (d, J = 9.9 Hz), 129.9 (d, J = 9.9 Hz), 129.8
(d, J = 53.2 Hz), 129.4, 128.7 (d, J = 9.6 Hz), 128.2 (d, J = 11.2 Hz),
124.6, 123.3, 59.7, 51.4, 37.9. 31P NMR (162.0 MHz, DMSO-d6) δ
25.1.
1
2-(4-Methoxyphenyl)benzoxazole (6g). H NMR (CDCl3, 400.1
MHz) δ 8.20 (d, 2H, J = 7.2 Hz), 7.73 (m, 1H), 7.55 (m, 1H), 7.3−7.4
(m, 2H), 7.02 (d, 2H, J = 8.4 Hz), 3.88 (d, 3H, J = 1.2 Hz). 13C NMR
(CDCl3, 100.6 MHz) δ 163.2, 162.3, 150.7, 142.3, 129.4, 124.6, 124.4,
119.7, 119.6, 114.4, 110.4, 55.5.
1
Synthesis of ({3-{2-{[2-(Diphenylphosphino-κP)-
benzylidene]amino-κN}ethyl}-1-methylimidazole-2-yl)(chloro)
Palladium(II) Hexafluorophosphate (5·PF6). Compound 3 (1
mmol, 543 mg), silver oxide (0.5 mmol, 116 mg), and dichloro-
methane (20 mL) were added into a 2-necked flask successively in
glovebox. The mixture was stirred under darkness for 2 h, and then
was filtered with Celite. PdCl2(CNMe)2 (1 mmol, 259 mg) was added
into the filtrate, and the mixture was stirred at ambient temperature for
3 h. At the end of the reaction, the mixture was filtered with Celite
again, and the addition of 20 mL of ether into the filtrate generated
pale yellow solid. The solid substance was filtered, washed with ether
(5 mL × 2), and dried under vacuum to give compound 5·PF6 (200
2-(3-Methoxyphenyl)benzoxazole (6h). H NMR (CDCl3, 400.1
MHz) δ 7.85 (dd, 1H, J = 7.6 Hz, J = 0.8 Hz), 7.7−7.8 (m, 2H), 7.59
(m, 1H), 7.43 (t, 1H, J = 7.6 Hz), 7.3−7.4 (m, 2H), 7.0−7.1 (m, 1H),
3.92 (s, 3H). 13C NMR (CDCl3, 100.6 MHz) δ 163.0, 160.0, 150.8,
142.0, 130.0, 128.3, 125.2, 124.6, 120.1, 120.0, 118.4, 111.9, 110.6,
55.5.
2-(2-Methoxyphenyl)benzoxazole (6i). 1H NMR (CDCl3, 400.1
MHz) δ 8.14 (dd, 1H, J = 7.6 Hz, J = 1.6 Hz), 7.82 (m, 1H), 7.59 (m,
1H), 7.51 (m, 1H), 7.3−7.4 (m, 2H), 7.11 (t, 2H, J = 8 Hz), 4.03 (s,
3H). 13C NMR (CDCl3, 100.6 MHz) δ 161.6, 158.5, 150.3, 142.1,
132.8, 131.3, 125.0, 124.3, 120.7, 120.2, 116.1, 112.1, 110.5, 56.2.
2-(Naphthalen-1-yl)benzoxazole (6j). 1H NMR (CDCl3, 400.1
MHz) δ 9.46 (d, 1H, J = 8.8 Hz), 8.43 (dd, 1H, J = 7.2 Hz, J = 1.2 Hz),
8.02 (d, 1H, J = 8.4 Hz), 7.93 (d, 1H, J = 8.0 Hz), 7.88 (m, 1H), 7.5−
7.8 (m, 4H), 7.39 (m, 2H). 13C NMR (CDCl3, 100.6 MHz) δ 162.8,
150.2, 142.3, 134.0, 132.3, 130.7, 129.3, 128.7, 128.0, 126.5, 126.3,
125.3, 125.0, 124.5, 123.7, 120.3, 110.5.
1
mg, 27% yield). H NMR (400.1 MHz, DMSO-d6) δ 8.77 (s, 1H),
7.93 (m, 1H), 7.87 (m, 1H), 7.79 (m, 1H), 7.5−7.7 (m, 11H), 7.42 (t,
1H, J = 1.6 Hz), 7.21 (t, 1H, J = 8.4 Hz), 4.57 (m, 2H), 3.98 (m, 2H),
3.89 (s, 3H). 13C NMR (100.6 MHz, DMSO-d6) δ 170.1 (d, J = 10.0
Hz), 162.7 (d, J = 186.4 Hz), 137.5 (d, J = 8.5 Hz), 135.9 (d, J = 17.8
Hz), 134.6 (d, J = 6.1 Hz), 134.0 (d, J = 11.6 Hz), 133.7, 132.8, 131.9,
129.1 (d, J = 11.0 Hz), 125.7 (d, J = 51.2 Hz), 123.4 (d, J = 5.1 Hz),
122.0 (d, J = 39.4 Hz), 121.3 (d, J = 4.8 Hz), 59.5, 48.9, 37.3. 31P
NMR (162.0 MHz, DMSO-d6) δ 19.3, −144.2 (m, J = 712.8 Hz).
HRMS (ESI-TOF) m/z: [M − PF6]+ Calcd for C25H24ClN3PPd
538.0431. Found 538.0430. [M − Cl − PF6]2+ Calcd for C25H24N3PPd
251.5372. Found 251.5368.
1
2-(3-Fluoro-4-methylphenyl)benzoxazole (6k). H NMR (CDCl3,
400.1 MHz) δ 7.8−8.0 (m, 2H), 7.77 (m, 1H), 7.57 (m, 1H), 7.3−7.4
(m, 3H), 2.36 (d, 3H, J = 2.0 Hz). 13C NMR (CDCl3, 100.6 MHz) δ
162.1 (d, J = 3.0 Hz), 161.3 (d, J = 244.0 Hz), 150.7, 142.0, 132.1 (d, J
= 5.3 Hz), 129.0 (d, J = 17.4 Hz), 126.6 (d, J = 8.6 Hz), 125.3, 124.7,
123.1 (d, J = 3.5 Hz), 120.1, 114.1 (d, J = 24.9 Hz), 110.6, 14.8 (d, J =
3.5 Hz). HRMS (ESI-TOF) m/z: [M + H]+ Calcd for C14H11FNO
228.0825. Found 228.0816.
Typical Catalytic Procedure of Arylation of Benzoxazole. In
glovebox, benzoxazole (1.5 mmol, 179 mg), complex 4 (0.005 mmol,
2.9 mg) and LiOtBu (1 mmol, 80 mg) were added into a Schlenk tube
successively. Then, PhBr (1 mmol, 93 mg) and DME (4 mL) was
injected into the reaction tube at atmosphere. The tube was heated to
90 °C and stirred for 24 h. After the reaction completed, the mixture
was cooled to room temperature and diluted with EtOAc of 5 mL.
Some insoluble solid was filtered off with silica pad, and the filtrate was
evaporated to dryness under vacuum and purified by column
chromatography to give white solid 6a (186 mg, 95% yield).
1
2-(4-Fluoro-3-methylphenyl)benzoxazole (6l). H NMR (CDCl3,
400.1 MHz) δ 8.12 (dd, 1H, J = 7.2 Hz, J = 1.6 Hz), 8.05 (m, 1H),
7.76 (m, 1H), 7.57 (m, 1H), 7.3−7.4 (m, 2H), 7.14 (t, 1H, J = 8.8
Hz), 2.37 (d, 3H, J = 2 Hz). 13C NMR (CDCl3, 100.6 MHz) δ 163.6
(d, J = 227.5 Hz), 162.2, 150.8, 142.1, 131.1 (d, J = 5.9 Hz), 127.1 (d, J
= 8.8 Hz), 125.9 (d, J = 18.1 Hz), 125.0, 124.6, 123.1 (d, J = 3.4 Hz),
119.9, 115.8 (d, J = 23.1 Hz), 110.5, 14.5 (d, J = 3.5 Hz). HRMS (ESI-
TOF) m/z: [M + H]+ Calcd for C14H11FNO 228.0825. Found
228.0819.
1
2-(3,5-Bis(trifluoromethyl)phenyl)benzoxazole (6m). 1H NMR
(CDCl3, 400.1 MHz) δ 8.72 (s, 2H), 8.03 (s, 1H), 7.83 (m, 1H),
7.65 (m, 1H), 7.4−7.5 (m, 2H). 13C NMR (CDCl3, 100.6 MHz) δ
160.0, 150.9, 141.7, 132.7 (q, J = 33.8 Hz), 129.3, 127.5 (broad),
2-Phenylbenzoxazole (6a). H NMR (CDCl3, 400.1 MHz) δ 8.28
(m, 2H), 7.79 (m, 1H), 7.60 (m, 1H), 7.5−7.6 (m, 3H), 7.3−7.4 (m,
2H). 13C NMR (CDCl3, 100.6 MHz) δ 163.0, 150.7, 142.0, 131.6,
129.0, 127.7, 127.1, 125.2, 124.6, 120.0, 110.6.
H
Organometallics XXXX, XXX, XXX−XXX

 Li, Yaqiu
Li, Yaqiu
 Yu, Xiaojun
Yu, Xiaojun
 Wang, Yangdiandian
Wang, Yangdiandian
 Fu, Haiyan
Fu, Haiyan
 Zheng, Xueli
Zheng, Xueli
 Chen, Hua
Chen, Hua
 Li, Ruixiang
Li, Ruixiang