ORGANIC
LETTERS
2
001
Vol. 3, No. 1
-8
An Azophenol-Based Chromogenic
Anion Sensor
5
Dong Hoon Lee, Kwan Hee Lee, and Jong-In Hong*
School of Chemistry and Molecular Engineering, Seoul National UniVersity,
Seoul 151-747, South Korea
Received October 4, 2000
ABSTRACT
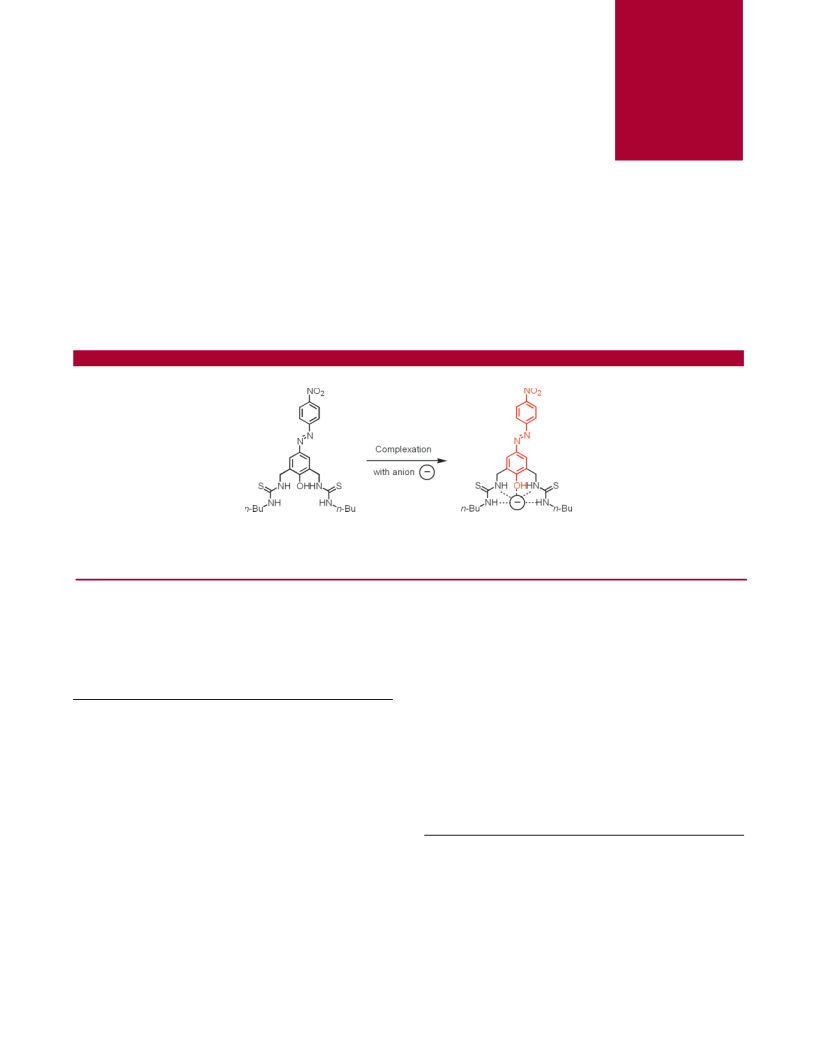
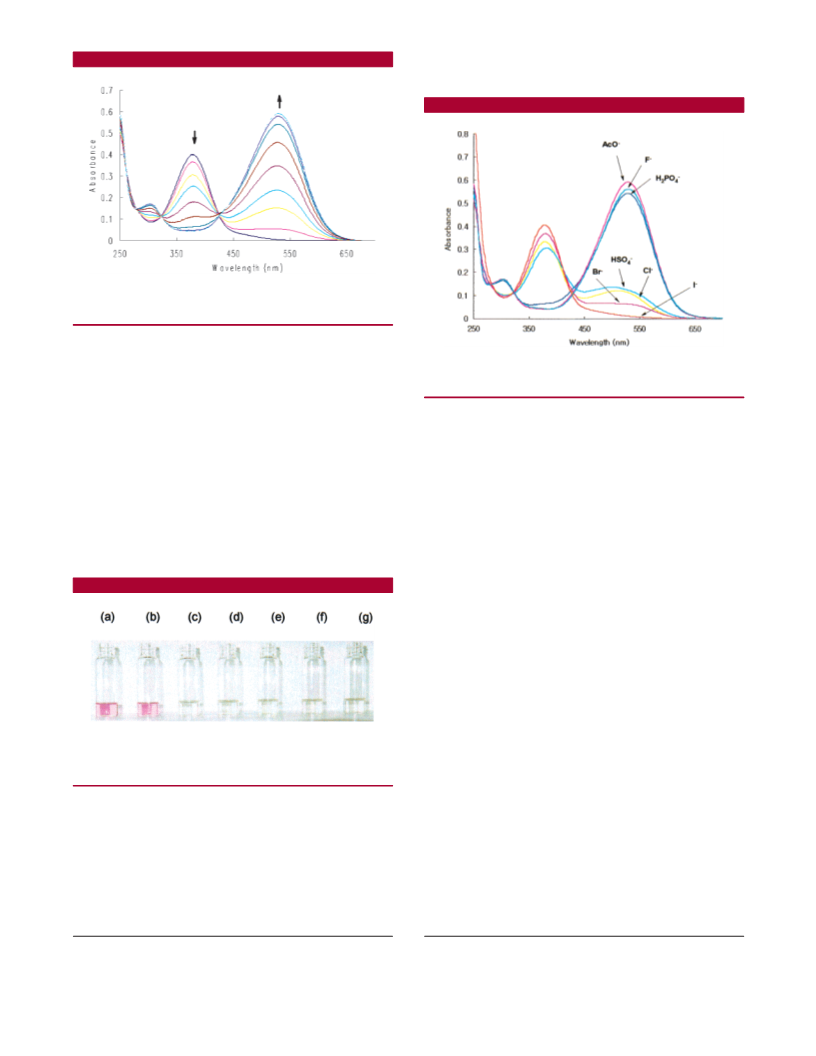
A new chromogenic azophenol−thiourea based anion sensor, 2, has been developed. This system allows for the selective colorimetric detection
-
-
-
of F , H
2 4
PO , and AcO . Selectivity trends turned out to be dependent upon guest basicity and conformational complementarity between 2
and the guest.
Compared to the relatively well-developed cation chemo-
sensors, development of anion binding sensors is only
generally consists of two parts. One is the anion-binding part
employing various combinations of pyrroles, guanidiniums,
1
3
-5
recently emerging as a research area of significant impor-
Lewis acids, amides, and urea/thioureas. The other is the
chromophore which makes binding-induced changes into
2
tance. One of the more attractive approaches in this field
3,4
1,2
involves the construction of optical sensors. This system
optical signals. These two parts are either covalently
4
2c,d,3
attached or intermolecularly associated.
However, because of the near-absence of chromophores
(
1) (a) Takagi, M. In Cation Binding by Macrocycles; Inoue, Y., Gokel,
W. G., Ed.; Marcel Dekker: New York, 1990; Chapter 11,pp 465-496.
b) Topics in Current Chemistry (Supramolecular Chemistry I); Weber, E.,
which are responsive to anions, the number of optical sensors
(
5c,6
available for anionic species is very low.
Only a few
Ed.; Springer: Berlin, 1993; Vol. 165. (c) L o¨ hr, H.-G.; V o¨ gtle, F. Acc.
Chem. Res. 1985, 18, 65-72. (d) Hayashita, T.; Takagi, M. In Compre-
hensiVe Supramolecular Chemistry; Atwood, J. L., Davies, J. E. D.,
MacNicol, D. D., V o¨ gtle, F., Suslick, K. S., Eds.; Pergamon: Oxford, 1996;
pp 635-669.
systems reported by several groups are known.2 In this
-4
Letter we present the synthesis and binding properties of an
2
a,7
azophenol-thiourea system. We show that compound 2
acts as a colorimetric sensor for selected anions by means
of hydrogen-bonding interactions. Intermolecular interactions
(2) (a) Brz o´ zka, Z. In ComprehensiVe Supramolecular Chemistry;
Atwood, J. L., Davies, J. E. D., MacNicol, D. D., V o¨ gtle, F., Suslick, K.
S., Eds.; Pergamon: Oxford, 1996; pp 187-212. (b) Chemosensors of Ion
and Molecular recognition; Desvergne, J.-P., Czarnik, A. W., Eds.;
Kluwer: Dordrecht, 1997; Vol. 492. (c) Niikura, K.; Metzger, A.; Anslyn,
E. V. J. Am. Chem. Soc. 1998, 120, 8533-8534. (d) Metzger, A.; Anslyn,
E. V. Angew. Chem., Int. Ed. 1998, 37, 649-652. (e) Kubo, Y.; Maeda,
S.; Tokita, S.; Kubo, M. Nature. 1996, 382, 522-523.
(5) For reviews, see: (a) Supramolecular Chemistry of anions; Bianchi,
A., Bowman-James, K., Garcia-Espana, E., Eds.; Wilely-VCH: New York,
1997. (b) Lehn, J.-M. Supramolecular Chemistry, Concepts and Perspec-
tiVes; VCH: Weinheim, 1995. (c) Schmidtchen, F. P.; Berger, M. Chem.
ReV. 1997, 97, 1609-1646.
(6) (a) Xie, H.; Yi, S.; Wu, S. Chem. Commun. 1999, 2751-2754. (b)
Nishizawa, S.; Kaneda, H.; Uchida, T.; Teramae, N. J. Chem. Soc., Perkin
Trans. 2 1998, 2325. (c) Kubo, Y.; Tsukahara, M.; Ishihara, S.; Tokita, S.
Chem. Commun. 2000, 653-654. (d) Hayashita, T.; Onodera, T.; Kato, R.;
Nishizawa, S.; Teramae, N. Chem. Commun. 2000, 755-756.
(7) Tsuge A.; Moriguchi, T.; Mataka, S.; Tachiro. M. J. Chem. Soc.,
Perkin Trans. 1993, 2211.
(
3) (a) Lavigene, J. J.; Anslyn, E. V. Angew. Chem., Int. Ed. 1999, 38,
666-3669. (b) Gale, P. A.; Twyman, L. J.; Handlin, C. I.; Sessler, J. L.
Chem. Commun. 1999, 1851-1852.
4) (a) Black, C. B.; Andrioletti, B.; Try, A. C.; Ruiperez, C.; Sessler, J.
3
(
L. J. Am. Chem. Soc. 1999, 121, 10438-10439. (b) Miyaji, H.; Sato, W;
Sessler, J. L. Angew. Chem. Int. Ed. 2000, 39, 1777-1780. (c) Anzenbacher,
P., Jr.; Jurs ´ı kov a´ , K.; Sessler, J. L. J. Am. Chem. Soc. 2000, 122, 9350-
9
351.
1
0.1021/ol006690t CCC: $20.00 © 2001 American Chemical Society
Published on Web 12/16/2000

 Lee, Dong Hoon
Lee, Dong Hoon
 Lee, Kwan Hee
Lee, Kwan Hee
 Hong, Jong-In
Hong, Jong-In