Angewandte
Communications
Chemie
selectivity after isomerization at 608C or above for multiple
Et O), with each plot exhibiting exponential decay of
2
[
19–22]
+
hours.
The activity of catalyst 2 is attributed to the
allylbenzene (Figure 1A). A plot of kobs vs. [Na ] is linear
À4
À1
hemilability of the ether ligand cis to the hydride ligand. The
high selectivity is attributed to the mild reaction conditions,
with the E isomer being thermodynamically favored at lower
(Figure S13) with a non-zero intercept (3.06 ꢀ 10 min )
close to the experimentally observed value for cation-free
isomerization under comparable conditions (Figure S3). The
[
23]
+
temperatures.
presence of an additional Na -dependent term in the rate law
We next sought to tune the activity of 2 using cation–
crown interactions to adjust substrate-binding tendencies
[Eq. (S1)] is consistent with a cation–crown binding equilib-
rium. NMR spectroscopy confirms that 2 is the resting state in
each case.
(
Scheme 1). Carrying out allylbenzene isomerization in the
F
presence of 1 mol% KBAr 4 and 350 mm Et O did not
2
F
provide any rate enhancement. Including 2.2 mol% NaBAr 4
and 78 mm Et O on the other hand, led to a 3-fold rate
2
À1
enhancement (TOF = 5.4 h ) relative to salt-free conditions,
reaching completion after about 100 h. Lithium salts foster
remarkable increases in catalytic activity: Upon injection of
[
24]
allylbenzene into an NMR tube containing 1 mol% 2 and
F
0
.8 mol% LiBAr 4 in CD Cl , complete isomerization oc-
2
2
curred within 10 min. The selective response of catalyst 2 to
+
+
+
Li is striking, given that Na and Li have similar binding
[
25]
affinity with 12-crown-4.
The dramatic increase in activity caused by lithium salts
enabled a reduction in the required amount of precious metal
catalyst. At 0.1 mol% (0.5 mm) 2 in the presence of 10 equiv
F
4
of LiBAr ·3Et O, full conversion of allylbenzene to b-
2
methylstyrene (> 99% trans) was observed in 24 min
À1
(
11.6 min half-life, TOF = 2750 h ). Isomerization by 2
alone, even at 5 mol% loading, proceeded with a 530 min
half-life. The cation-tuning approach thus enabled a 50-fold
reduction in Ir loading while still producing trans-b-methyl-
styrene 46 times faster than 5 mol% 2!
Rapid olefin isomerization is not confined to allybenzene.
The phenylpropenoid 4-methoxyallylbenzene is isomerized
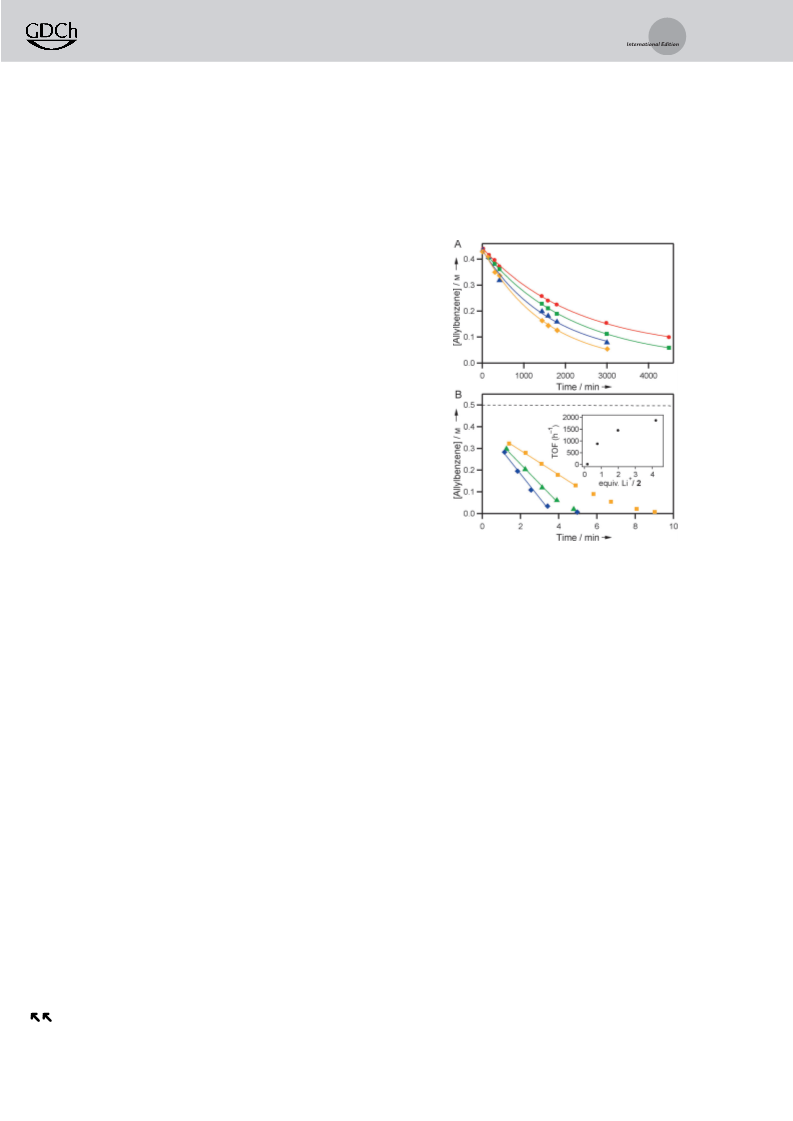
Figure 1. Allylbenzene isomerization by 5 mm 2 (A) with 1.1 (red
+
circles), 2.5 (green squares), 5.0 (blue triangles), and 7.4 (yellow
by the 2/Li system in less than 10 min, forming the fragrance
F
[26]
diamonds) equiv of NaBAr
(
4
, and (B) with 0.8 (orange squares), 2.0
additive anethole in 98% yield and with 97.3% selectivity
F
green triangles), 4.2 (blue diamonds) equiv of LiBAr ·3Et O, and
4 2
+
without salt (dashed line). Inset: Li influence on TOF.
for the trans isomer. Isomerization of 0.5m 1-hexene by
1
4
mol% 2 (without added salt) reached 97% conversion after
0 h (t1/2 = 487 min) with a distribution of isomers favoring
+
trans-2-hexene (81% yield). In the presence of 1.3 mol%
LiBAr ·3Et O, it took only 2 min to reach a similar distri-
bution ( t1 = 0.59 min), indicating an 825-fold rate enhance-
ment. Allowing the Li -containing mixture to react further
led to about 17% yield of 3-hexenes after 190 min, compared
to roughly 3% 3-hexenes in the absence of Li after 40 h,
suggesting that cations can also influence regioselectivity
patterns. The 2/Li system is among the fastest isomerization
catalysts at room temperature.
Detailed kinetic studies were carried out to provide
insight into the mechanism of this cation-tuned catalysis.
Allylbenzene isomerization was monitored by H NMR
spectroscopy. With catalyst 2 (without alkali metal salts),
The dramatic Li -promoted rate enhancement is accom-
F
panied by a striking change in the kinetic profile (Figure 1B).
In the presence of Li , the concentration of allylbenzene
decreases linearly over time, indicating a shift to a regime that
is zero-order in olefin. As the Li /2 ratio increases, the rate of
isomerization increases dramatically before eventually level-
ing off (Figure 1B, inset). The fastest Li -accelerated trial
(TOF = 1870 h and zero-order half-life, t1 = 1.6 min) fea-
tures a 1100-fold enhancement compared to the standard
4
2
0
+
/2
+
+
+
+
+
À1
0
/2
[
4,5,19,26,27]
À1
conditions (TOF = 1.8 h and t1/2 = 1810 min).
A mechanism consistent with the data is shown in
Scheme 2. In the absence of cations, the reaction is first-
order in catalyst and allylbenzene, consistent with a pre-
equilibrium displacement of crown ether by allylbenzene
(Keq,1) influencing the observed rate. The rate accelerates as
the alkali metal salt concentration increases, indicating
a parallel catalytic cycle in which cation–crown interactions
stabilize the proposed olefin adduct A (Keq,2). Li salts shift
Keq,2 further towards A, reaching a regime that is zero-order
in allylbenzene and eventually zero-order in Li , with
turnover-limiting insertion or elimination at Ir. Accordingly,
1
the concentration of allylbenzene decreased exponentially
(
Figure S2 in the Supporting Information), indicating that the
À4
À1
reaction is first-order in olefin (kobs = 3.83 ꢀ 10 min , t
=
1/2
1
810 min). The reaction is also first-order in 2, on the basis of
kobs increasing linearly with increasing catalyst concentration
Figure S6).
The broad mechanistic features did not change in the
+
(
+
+
presence of Na . The rate of isomerization increased linearly
as the Na /2 ratio increased from 1.1 to 7.5 (constant 0.5m
+
the hydride resonance of 2 diminishes (Figure S16) and other
2
ꢀ 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2017, 56, 1 – 6
These are not the final page numbers!

 Kita, Matthew R.
Kita, Matthew R.
 Miller, Alexander J. M.
Miller, Alexander J. M.