NMR of base-modified pyrimidine nucleosides
1
3
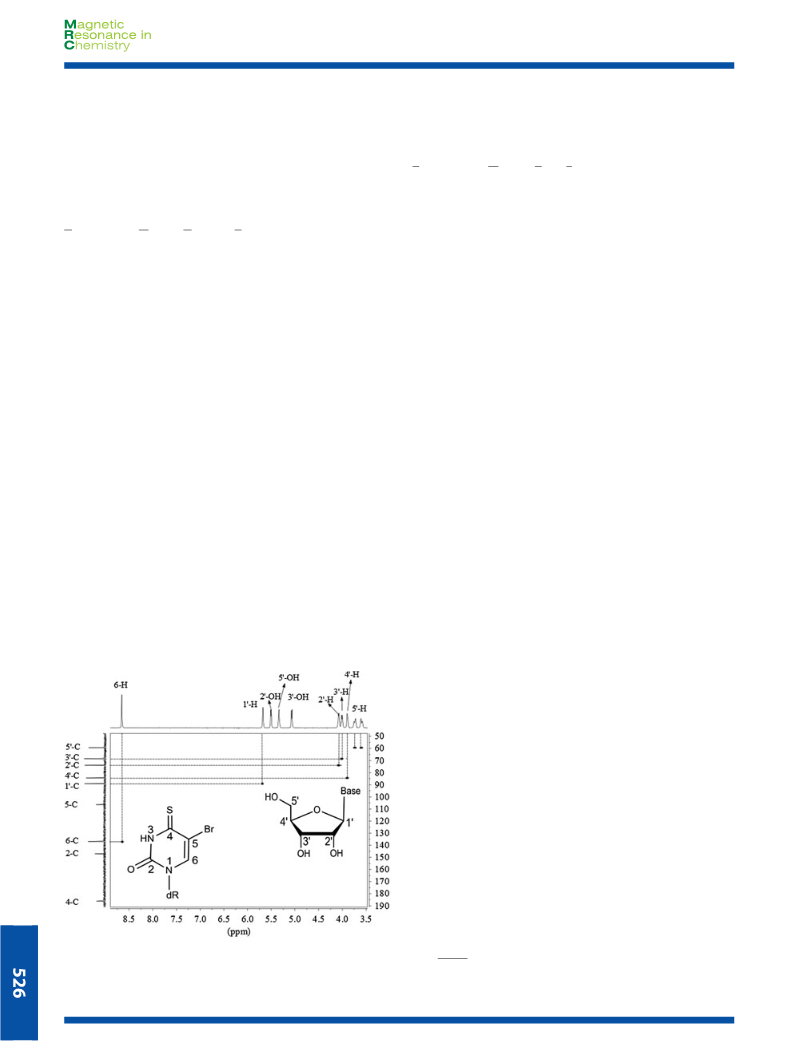
for F and Cl but not for Br and I. The C resonances of 5-C in
-bromonucleosides (1e, 2e, 3e and 4e) and 5-iodo-nucleo-
2', 3', 5'-tri-O-acetyl-5-bromo-4-thiouridine
', 3', 5'-tri-O-acetyl-5-bromouridine (0.90 g, 2.00 mmol) was
5
2
sides (1f, 2f, 3f and 4f) in fact have lower δ values relative
to that of the un-modified uracil nucleosides. Clearly, the elec-
tron-withdrawing effect alone is not enough to explain these
observations. This unusual effect could be explained by the
dissolved in 1, 4-dioxane (30 ml), and P S (0.85 g, 3.84 mmol) was
2
5
added. The mixture was refluxed at 106 °C for 1.5h (monitored by
TLC). The solvent was removed under reduced pressure, and the
residue was treated several times with CH Cl . The residue was
2 2
purified on a silica gel column and eluted with petroleum: ethyl
[
22]
‘
heavy atom effect’
that is when a carbon atom is attached
to a heavy halogen atom (such as Br or I), the diamagnetic in-
teractions arising from the numerous electrons of bromo or
iodine atom increase the shielding effect of the substituted
carbon atom so that the NMR resonances shift upfield.
acetate (3:2, v/v) to give the title compound (0.51 g, 55.6%).
5-bromo-4-thiouridine (4e)
2
', 3', 5'-tri-O-acetyl-5-bromo-4-thiouridine (0.63 g, 1.95 mmol)
Conclusions
was suspended in absolute MeOH (120 ml) and was saturated
with dry ammonia gas by stirring at room temperature for 4.5 h.
The resulting solution was evaporated under reduced pressure
at 35 °C, and the residue was purified on a silica gel column
A series of 5-substituted-4-thio-uridines and their 2'-deoxy ana-
logues have been successfully prepared from their respective
parent nucleosides. Their H and C NMR have been systemically
investigated. Practical methods are established to unambigu-
ously assign all pyrimidine carbons and to distinguish between
1
13
packed in CH
2 2 2 2
Cl : MeOH = 9:1. Column was eluted with CH Cl :
MeOH = 9.5:0.5; the solvent was then removed in vacuo to pro-
vide a solid residue. The residue was recrystallized from H
give the title compound (0.33 g, 71%).
2
O to
1
'-C and 4'-C signals in both types of nucleosides. These assign-
ments can provide useful references for 4-thiopyrimidine nucleo-
sides and other base-modified nucleosides. The finding that the
1
H chemical shifts of the imino proton in these 4-thionucleosides
Acknowledgements
are unusually high would offer an excellent marker in NMR stud-
ies of nucleic acids containing these thio-bases.
The authors are most grateful to Mr Yong-Ting Xu at Liaoning
Normal University, China, for his excellent technical assistance with
NMR. Dr Xu is most grateful to Dr Elaine Moore for critical reading of
the draft. The work is financially supported by the Scientific
Research Foundation for the Returned Overseas Chinese Scholars,
State Education Ministry, China.
Experimental
NMR instruments
5
00 MHz from Bruker (AV-500, FT NMR) and 400 MHz from Bruker
AV-400, FT NMR). The COSY spectra (DMSO-d ) were obtained in
the magnitude mode with 1024 points in the F2 dimension and
56 increments in the F1 dimension. Each increment FID was
obtained with 12 scans with a relaxation delay of 2 s. HMQC spec-
tra (DMSO-d ) were obtained in the magnitude mode with 1024
(
6
References
[
[
[
1] T. Lindahl. Nature 1993, 362, 709–715.
2] E. Ichikawa, K. Kato. J. Synth. Org. Chem. Jpn. 2001, 59, 331–345.
3] S. Porcher, S. Pitsch. Helv. Chim. Acta 2005, 88, 2683–2704.
2
6
[4] M. K. Lakshman. Curr. Org. Synth. 2005, 2, 83–112.
points in the F2 dimension and 256 increments in the F1 dimen-
sion. Each increment FID was obtained with 16 scans with a relax-
ation delay of 2 s.
[5] P. A. Limbach, P. F. Crain, J. A. McCloskey. Nucleic Acids Res. 1994, 22,
2183–2196.
[6] M. N. Prichard, E. R. Kern. Viruses-Basel 2010, 2, 1968–1983.
[7] L. C. J. Gillet, O. D. Scharer. Chem. Rev. 2006, 106, 253–276.
[8] A. Massey, Y. Z. Xu, P. Karran. Curr. Biol. 2001, 11, 1142–1146.
[9] Y. Z. Xu, X. H. Zhang, H. C. Wu, A. Massey, P. Karran. Bioorg. Med.
Chem. Lett. 2004, 14, 995–997.
Materials and methods
All chemicals and solvents, unless stated otherwise, were from
either Aldrich or Sigma. All chemicals and solvents were used
directly without further purification. Nucleosides on TLC were
[10] R. Marek, V. Sklenar, in Annual Reports on NMR Spectroscopy,
vol. 54, 2005, pp. 201–242.
[
[
11] P. F. Agris, S. C. Brown. Methods Enzymol. 1995, 261, 270–299.
12] B. Ancian, in Annual Reports on Nmr Spectroscopy, vol. 69 (Ed: G. A.
Webb), Elsevier Academic Press Inc., San Diego, 2010, pp. 39–143.
identified using p-anisaldehyde/ethanol/H SO (5:90:5) or EtOAc:
2
4
Petro Ether (8: 2) solution that converted the nucleosides
into black spots on heating.
Preparation of 5-substituted- 4-thiopyrimidine nucleosides:
Chemical synthesis of 5-bromo-4-thiouridine is described below as a
typical example.
[13] R. Narukulla, D. E. G. Shuker, V. Ramesh, Y. Z. Xu. Magn. Reson. Chem.
2
008, 46, 1–8.
[14] X. H. Zhang, Y. Z. Xu. Molecules 2011, 16, 5655–5664.
[15] J. Asakura, M. J. Robins. J. Org. Chem. 1990, 55, 4928–4933.
[16] H. Schott, D. Goltz, T. C. Schott, C. Jauch, R. A. Schwendener. Bioorg.
Med. Chem. 2011, 19, 3520–3526.
[
[
17] V. Kumar, S. V. Malhotra. Bioorg. Med. Chem. Lett. 2008, 18, 5640–5642.
18] J. J. Fox, D. Vanpraag, I. Wempen, I. L. Doerr, L. Cheong, J. E. Knoll, M. L.
Eidinoff, A. Bendich, G. B. Brown. J. Am. Chem. Soc. 1959, 81, 178–187.
19] C. B. Reese. Tetrahedron 1978, 34, 3143–3179.
2
', 3', 5'-tri-O-acetyl-5-bromouridine
[
[
To a solution of 5-bromouridine (1e) (0.87 g, 2.70 mmol) in dry
20] V. Kumar, J. Yap, A. Muroyama, S. V. Malhotra. Synthesis-Stuttgart
pyridine (15 ml) at 0 °C, dry (CH CO) O (3.0 ml, 32 mmol) was
added slowly. The reaction was stirred for 5 h and monitored by
the formation of fluorescent spots on TLC (5% MeOH in CH Cl ).
After the reaction was complete, the solvent was removed under
reduced pressure. A white solid crystallized from ethanol (95%)
was collected by filtration and dried on an oil vacuum pump to
give the title compound (1.16 g, 95.9%), mp 52–53 °C.
3
2
2
009, 3957–3962.
[21] F. Yang, Y. G. Zhu, B. A. Yu. Chem. Commun. 2012, 48, 7097–7099.
[22] S. Standara, K. Malinakova, R. Marek, J. Marek, M. Hocek, J. Vaara, M.
Straka. Phys. Chem. Chem. Phys. 2010, 12, 5126–5139.
2
2
[
23] G. W. M. Visser, R. E. Herder, P. Noordhuis, O. Zwaagstra, J. D. M.
Herscheid, F. J. J. Dekanter. J. Chem. Soc., Perkin Trans. 1 1988,
2
547–2554.
[24] Q. Wu, A. H. Xia, X. F. Lin. J. Mol. Catal. B: Enzym. 2008, 54, 76–82.
Magn. Reson. Chem. 2013, 51, 523–529
Copyright © 2013 John Wiley & Sons, Ltd.
wileyonlinelibrary.com/journal/mrc

 Zhang, Xiaohui
Zhang, Xiaohui
 Wang, Jian
Wang, Jian
 Xu, Yao-Zhong
Xu, Yao-Zhong