to improve the physiological half-lives of effective HIV
reverse-transcriptase inhibitors.12 The physiochemical
benefits associated with halogenation of hydrolysis-inert
analogues of pyrophosphate were initially proposed in-
dependently by Blackburn and McKenna.13,14 One of the
difficulties encountered in exploiting these probes for
mechanistic studies is that reported syntheses of non-
hydrolyzable nucleotide analogues are lengthy and often
low yielding. These reactions typically proceed by activa-
tion of a nucleoside monophosphate with subsequent
addition of a nucleophile. Typically there is a requirement
for a specific salt form of the nucleoside monophosphate
starting material, as well as the removal of the water of
crystallization, through repeated coevaporation with
pyridine. Lengthy reaction times for the phosphate
coupling are due to the poor nucleophilicity of (haloge-
nated) diphosphonates (>24 h), and often lengthy puri-
fications by ion-exchange chromatography are required
to furnish pure product.15
An Improved Method for the Synthesis of
Nucleoside Triphosphate Analogues
Samy Mohamady‡ and David L. Jakeman*,†,‡
College of Pharmacy, Dalhousie University, 5968 College
Street, Halifax, Nova Scotia, B3H 3J5, Canada, and
Department of Chemistry, Dalhousie University,
Halifax, Nova Scotia, B3H 4J3, Canada
Received September 5, 2005
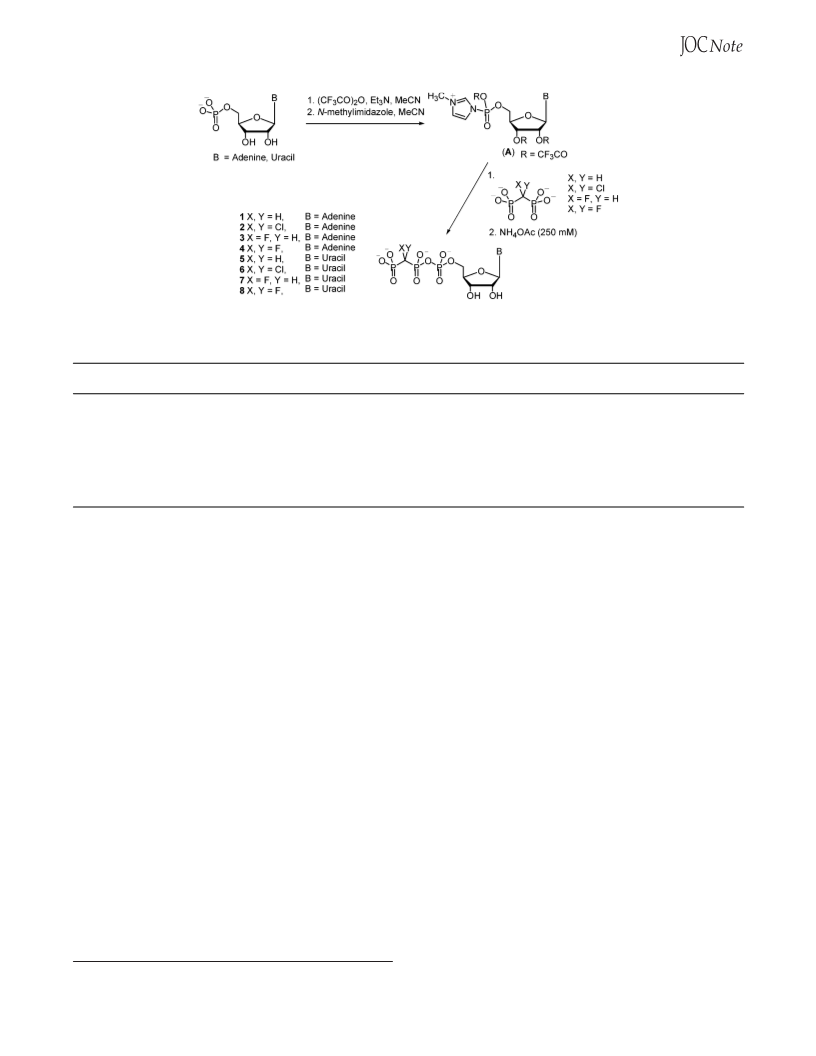
Herein, we report a significantly improved synthesis
and purification of substituted nucleoside 5′-â,γ-meth-
ylenetriphosphate analogues, as shown in Scheme 1. This
phosphonate-phosphate coupling procedure involves the
activation, and subsequent coupling, of purine and py-
rimidine nucleoside monophosphate-N-methylimidazo-
lides (A) with dichloromethylene-, difluoromethylene-,
fluoromethylene-, and methylenediphosphonic acids. This
reaction, which uses commercially available purine and
pyrimidine nucleoside monophosphates, proceeds without
the need to alter salt forms or remove moisture and is
complete within 2 h. Purification is facilated by the use
of ion-pair reversed-phase chromatography that is ame-
nable to scale-up.
Nucleoside monophosphates, when activated by trifluoro-
acetic anhydride and N-methylimidazole, efficiently couple
with a variety of electron-deficient diphosphonates in a
reproducible and efficient manner (<2 h, >72% isolated
yield). Unlike traditional methods for the preparation of
nucleoside 5′-â,γ-methylenetriphosphate analogues, there is
no requirement for predrying, or conversion to specific salt
forms, of commercially available nucleoside monophosphate
starting materials.
The first use of nucleoside monophosphate-N-meth-
ylimidazolides (A) as electrophilic reagents for phosphate
coupling was described by Bogachev for the synthesis of
deoxynucleoside 5′-triphosphates using activated deoxy-
nucleoside monophosphate-N-methylimidazolides and
pyrophosphate.16 Uridine monophosphate-N-methylimi-
dazolide was subsequently used by Kiessling for the
preparation of UDP-R-D-galactofuranose.17
Our coupling procedure for the synthesis of â,γ-
methylenenucleoside 5′-triphosphate analogues consists
of several facile steps. The electrophilic nucleoside 5′-
monophosphate-N-methylimidazolide (A) is formed by
the reaction of a nucleoside 5′-monophosphate, either as
the free acid, the monosodium, or the disodium salt form,
with an excess of trifluoroacetic anhydride in the pres-
ence of triethylamine in acetonitrile. The presence of
Mechanistic probes to investigate the roles of phos-
phate transfer in biological systems are of importance
in elucidating the mechanisms of fundamental enzyme-
catalyzed processes involving primary and secondary
metabolism.1 Non-hydrolyzable nucleotide analogues that
are isosteric and isoelectronic with nucleoside triphos-
phates are examples of analogues that have been used
successfully to probe phosphoryl transfer in enzyme-
catalyzed processes2-7 and receptor specificity.8-11 Re-
cently, such halophosphonate analogues have been shown
† College of Pharmacy.
‡ Department of Chemistry.
(1) Berkowitz, D. B.; Bose, M.; Pfannenstiel, T. J.; Doukov, T. J.
Org. Chem. 2000, 65, 4498-4508.
(2) Jakeman, D. L.; Ivory, A. J.; Blackburn, G. M.; Williamson, M.
P. J. Biol. Chem. 2003, 278, 10957-10962.
(3) Bystrom, C. E.; Pettigrew, D. W.; Branchaud, B. P.; O’Brien, P.;
Remington, S. J. Biochemistry 1999, 38, 3508-3518.
(4) Liu, X. H.; Brenner, C.; Guranowski, A.; Starzynska, E.; Black-
burn, G. M. Angew. Chem., Int. Ed. 1999, 38, 1244-1247.
(5) Shirokova, E. A.; Shipitsin, A. V.; Victorova, L. S.; Dyatkina, N.
B.; Goryunova, L. E.; Beabealashvilli, R. S.; Hamilton, C. J.; Roberts,
S. M.; Krayevsky, A. A. Nucleosides Nucleotides 1999, 18, 1027-1028.
(6) Bernstein, B. E.; Williams, D. M.; Bressi, J. C.; Kuhn, P.; Gelb,
M. H.; Blackburn, G. M.; Hol, W. G. J. J. Mol. Biol. 1998, 279, 1137-
1148.
(10) Hoffenberg, S.; Shannon, T. M.; Noonan, T. P.; Liu, S. B.; Daniel,
S.; Fishman, J. B.; Rubins, J. B.; Misra, H. K.; Wright, G. E.; Dickey,
B. F. Mol. Pharmacol. 1996, 49, 156-164.
(11) Arabshahi, L.; Khan, N. N.; Butler, M.; Noonan, T.; Brown, N.
C.; Wright, G. E. Biochemistry 1990, 29, 6820-6826.
(12) Wang, G.; Boyle, N.; Chen, F.; Rajappan, V.; Fagan, P.; Brooks,
J. L.; Hurd, T.; Leeds, J. M.; Rajwanshi, V. K.; Jin, Y.; Prhavc, M.;
Bruice, T. W.; Cook, P. D. J. Med. Chem. 2004, 47, 6902-6913.
(13) Blackburn, G. M. Chem. Ind. 1981, 134-138.
(14) McKenna, C. E.; Shen, P. J. Org. Chem. 1981, 46, 4573-4576.
(15) Blackburn, G. M.; England, D. A.; Kolkmann, F. J. Chem. Soc.,
Chem. Commun. 1981, 930-932.
(16) Bogachev, V. S. Russ. J. Bioorg. Chem. 1996, 22, 599-604.
(17) Marlow, A. L.; Kiessling, L. L. Org. Lett. 2001, 3, 2517-2519.
(7) Bystrom, C. E.; Pettigrew, D. W.; Remington, S. J.; Branchaud,
B. P. Bioorg. Med. Chem. Lett. 1997, 7, 2613-2616.
(8) Alvarado-Castillo, C.; Harden, T. K.; Boyer, J. L. Mol. Pharmacol.
2005, 67, 114-122.
(9) Spelta, V.; Mekhalfia, A.; Rejman, D.; Thompson, M.; Blackburn,
G. M.; North, R. A. Br. J. Pharmacol. 2003, 140, 1027-1034.
10.1021/jo0518598 CCC: $30.25 © 2005 American Chemical Society
Published on Web 10/29/2005
10588
J. Org. Chem. 2005, 70, 10588-10591

 Mohamady, Samy
Mohamady, Samy
 Jakeman, David L.
Jakeman, David L.