Journal of the American Chemical Society
Communication
M.; Hetterscheid, D. G. H.; Smits, J. M. M.; de Bruin, B.; Budzelaar, P. H.
M.; Gal, A. W. J. Mol. Catal. A: Chem. 2005, 232, 151.
(7) Monfette, S.; Turner, Z. R.; Semproni, S. P.; Chirik, P. J. J. Am.
Chem. Soc. 2012, 134, 4561.
(8) Yu, R. P.; Darmon, J. M.; Milsmann, C.; Margulieux, G. W.; Stieber,
S. C. E.; DeBeer, S.; Chirik, P. J. J. Am. Chem. Soc. 2013, 135, 13168.
(9) (a) Zhang, G.; Scott, B. L.; Hanson, S. K. Angew. Chem., Int. Ed.
2012, 51, 12102. (b) Zhang, G.; Hanson, S. K. Org. Lett. 2013, 15, 650.
(c) Zhang, G.; Vasudevan, K. V.; Scott, B. L.; Hanson, S. K. J. Am. Chem.
Soc. 2013, 135, 8668.
importantly no combustible gas was collected with a Toepler
pump (200 Torr expected).
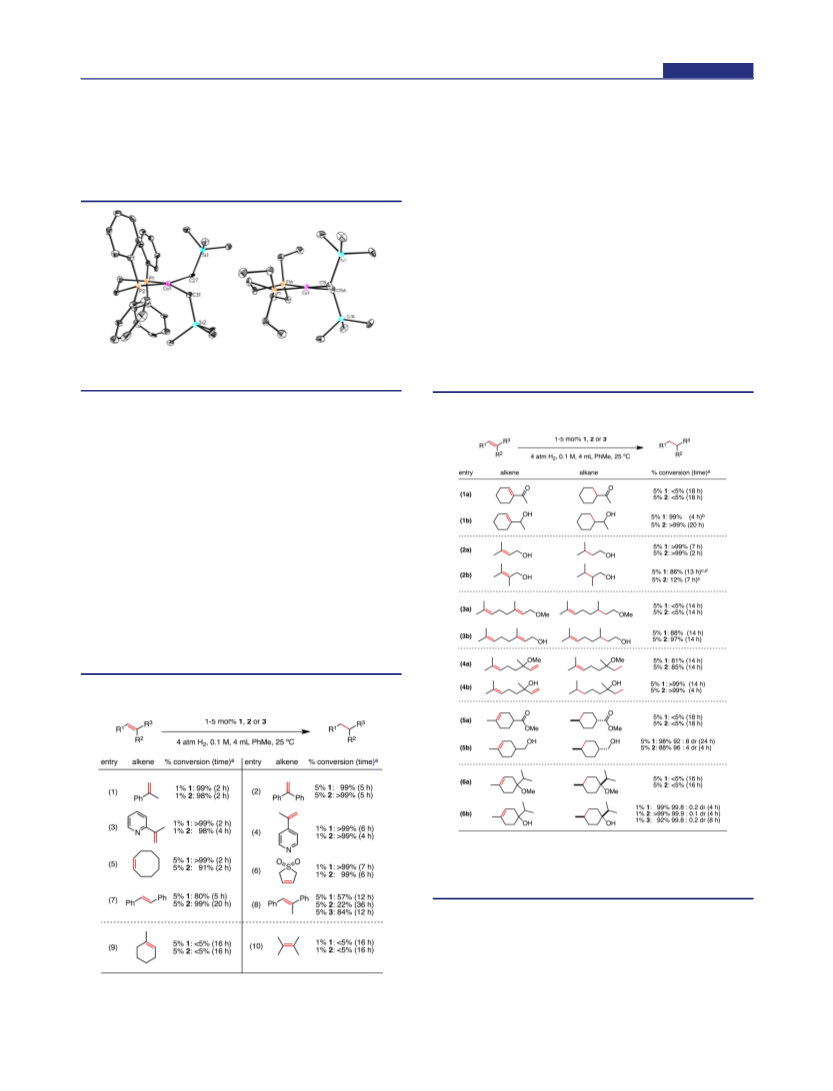
The competency of (dppe)Co(COD) as a precatalyst for
alkene hydrogenation was evaluated with α-methylstyrene and
terpinen-4-ol as representative substrates. Complete conversion
with the former was observed in 6 h under the standard
conditions, while for the latter only 34% conversion was obtained
after 2 h likely due to the presence of coordinating diene.
Notably, a 99.1:0.9 diastereomeric ratio was observed suggesting
catalyst activation similar to that for 1 (entry 6b, Table 2).
The mechanism for alkene hydrogenation by bis(phosphine)-
cobalt(II) alkyl complexes involves initial hydrogenolysis of the
alkyl groups to form SiMe4 (observed quantitatively by NMR
spectroscopy) and a thus far unobserved cobalt(II) dihydride.
Fryzuk et al. have reported a related phosphine-ligated cobalt
hydride, [(iPr2P(CH2)3PiPr2)Co]2(H)(μ-H)3,25 from addition
of H2 to the corresponding Co(I) allyl. Subsequent olefin
insertion and reductive elimination of alkane generates a putative
Co(0) species that coordinates alkene. Support for the existence
of such a species is provided by the isolation and catalytic activity
of (dppe)Co(COD). These observations and understanding
provide a platform for future optimization of base metal alkene
hydrogenation catalysts with high diastereo- and enantioselec-
tivity.
(10) Hopmann, K. H. Organometallics 2013, 32, 6388.
(11) Friedfeld, M. R.; Shevlin, M.; Hoyt, J. M.; Krska, S. W.; Tudge, M.
T.; Chirik, P. J. Science 2013, 342, 1076.
(12) For early reports of phosphine-ligand cobalt hydrogenation
catalysts, see: (a) Bleeke, J. R.; Muetterties, E. L. J. Am. Chem. Soc. 1981,
103, 556. (b) DuBois, D. L.; Meek, D. W. Inorg. Chim. Acta 1976, 19,
L29. (c) Hendriske, J. L.; Coenen, J. W. E. J. Catal. 1973, 30, 72.
(d) Hendrikse, J. L.; Kaspersma, J. H.; Coenen, J. W. E. Int. J. Chem.
Kinet. 1975, 7, 557. (e) Hidai, M.; Kuse, T.; Hikita, T.; Uchida, Y.;
Misono, A. Tetrahedron Lett. 1970, 11, 1715. (f) Yamamoto, A.;
Kitazume, S.; Pu, L. S.; Ikeda, S. J. Am. Chem. Soc. 1971, 93, 371.
(13) Camacho-Bunquin, J.; Ferguson, M. J.; Stryker, J. M. J. Am. Chem.
Soc. 2013, 135, 5537.
(14) Zhu, D.; Janssen, F. F. B. J.; Budzelaar, P. H. M. Organometallics
2010, 29, 1897.
(15) Imamura, Y.; Mizuta, T.; Miyoshi, K.; Yorimitsu, H.; Oshima, K.
Chem. Lett. 2006, 35, 260.
(16) Semproni, S. P.; Milsmann, C.; Chirik, P. J. J. Am. Chem. Soc. 2014,
136, 9211.
ASSOCIATED CONTENT
* Supporting Information
Experimental details, substrate and product characterization, and
crystallographic data for 1−4 in cif format. This material is
■
S
(17) (a) Maki, A. H.; Edelstein, N.; Davison, A.; Holm, R. H. J. Am.
Chem. Soc. 1964, 86, 4580. (b) Nishida, Y.; Sumita, A.; Kida, S. Bull.
Chem. Soc. Jpn. 1977, 50, 759. (c) Nishida, Y.; Kida, S. Bull. Chem. Soc.
Jpn. 1978, 51, 143. (d) Nishida, Y.; Hayashi, K.; Sumita, A.; Kida, S. Bull.
Chem. Soc. Jpn. 1980, 53, 271. (e) Przyojski, J. A.; Arman, H. D.;
Tonzetich, Z. J. Organometallics 2013, 32, 723.
AUTHOR INFORMATION
Corresponding Author
Notes
■
(18) Trovitch, R. J.; Lobkovsky, E.; Bouwkamp, M. W.; Chirik, P. J.
Organometallics 2008, 27, 6264.
(19) Crabtree, R. H.; Davis, M. W. J. Org. Chem. 1986, 51, 2655.
(20) (a) Loginov, D. A.; Pronin, A. A.; Starikova, Z. A.; Petrovskii, P.
V.; Kudinov, A. R. Koord. Khim. (Russ. J. Coord. Chem.) 2010, 36, 795.
(b) Hung-Low, F.; Krogman, J. P.; Tye, J. W.; Bradley, C. A. Chem.
Commun. 2012, 48, 368. (b) Hung-Low, F.; Bradley, C. A. Inorg. Chem.
2013, 52, 2446.
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
We thank the U.S. National Science Foundation (NSF) for a
Grant Opportunities for Academic Liaison with Industry
(GOALI) grant (CHE-1265988) between Princeton and
Merck. M.R.F. thanks the NSF for a Graduate Research
Fellowship (DGE-1148900). We also thank Z. R. Turner and
S. P. Semproni for assistance with X-ray crystallography, Prof. C.
Milsmann for assistance with EPR spectroscopy, and R. P. Yu for
assistance with Toepler pump experiments.
(21) (a) Jonas, K.; Mynott, R.; Kruger, C.; Sekutowski, J. C.; Tsay, Y.-
̈
H. Angew. Chem., Int. Ed. 1976, 15, 767. (b) Jonas, K. Angew. Chem., Int.
Ed. 1985, 24, 295. (c) Wang, J.-Q.; Fassler, T. F. Z. Naturforsch. 2009,
̈
64b, 985. (d) Brennessel, W. W.; Ellis, J. E. Inorg. Chem. 2012, 51, 9076.
(22) Klein, H.-F.; Fabry, L.; Witty, H.; Schubert, U.; Lueken, H.;
Stamm, U. Inorg. Chem. 1985, 24, 683.
(23) Drexler, H.-J.; Zhang, S.; Sun, A.; Spannenberg, A.; Arrieta, A.;
Preetz, A.; Heller, D. Tetrahedron: Asymmetry 2004, 15, 2139.
(24) (a) Deblon, S.; Liesum, L.; Harmer, J.; Schonberg, H.; Schweiger,
̈
A.; Grutzmacher, H. Chem.Eur. J. 2002, 8, 601. (b) Laporte, C.;
̈
REFERENCES
■
Breher, F.; Geier, J.; Harmer, J.; Schweiger, A.; Grutzmacher, H. Angew.
Chem., Int. Ed. 2004, 43, 2567. (c) Schonberg, H.; Boulmaaz, S.; Worle,
M.; Liesum, L.; Schweiger, A.; Grutzmacher, H. Angew. Chem., Int. Ed.
1998, 37, 1423. (d) de Bruin, B.; Russcher, J. C.; Grutzmacher, H. J.
̈
(1) Yamagishi, T. In The Handbook of Homogeneous Hydrogenation; de
Vries, J. G., Elsevier, C. J., Eds.; Wiley-VCH: Weinheim, 2008; Chapter
21.
(2) Brown, J. M. Angew. Chem., Int. Ed. Engl. 1987, 26, 190.
(3) Hoveyda, A. H.; Evans, D. A.; Fu, G. C. Chem. Rev. 1993, 93, 1307.
(4) For selected examples in natural product synthesis, see:
(a) Uwamori, M.; Saito, A.; Nakada, M. J. Org. Chem. 2012, 77, 5098.
(b) Yang, Q.; Njardarson, J. T.; Draghici, C.; Li, F. Angew. Chem., Int. Ed.
2013, 52, 8648. (c) DeCamp, A. E.; Verhoeven, T. R.; Shinkai, I. J. Org.
Chem. 1989, 54, 3207.
̂
̈
̈
̈
̈
Organomet. Chem. 2007, 692, 3167.
(25) Fryzuk, M. D.; Ng, J. B.; Rettig, S. J.; Huffman, J. C.; Jonas, K.
Inorg. Chem. 1991, 30, 2437.
(5) Chirik, P. J.; Weighardt, K. Science 2010, 327, 5967.
(6) (a) Kooistra, T. M.; Knijnenburg, Q.; Smits, J. M. M.; Horton, A.
D.; Budzelaar, P. H. M.; Gal, A. W. Angew. Chem., Int. Ed. 2001, 40, 4719.
(b) Gibson, V. C.; Humphries, M. J.; Tellmann, K. P.; Wass, D. F.;
White, A. J. P.; Williams, D. J. Chem. Commun. 2001, 2252.
(c) Knijnenberg, Q.; Horton, A. D.; van der Heijden, H.; Kooistra, T.
13181
dx.doi.org/10.1021/ja507902z | J. Am. Chem. Soc. 2014, 136, 13178−13181

 Friedfeld, Max R.
Friedfeld, Max R.
 Margulieux, Grant W.
Margulieux, Grant W.
 Schaefer, Brian A.
Schaefer, Brian A.
 Chirik, Paul J.
Chirik, Paul J.