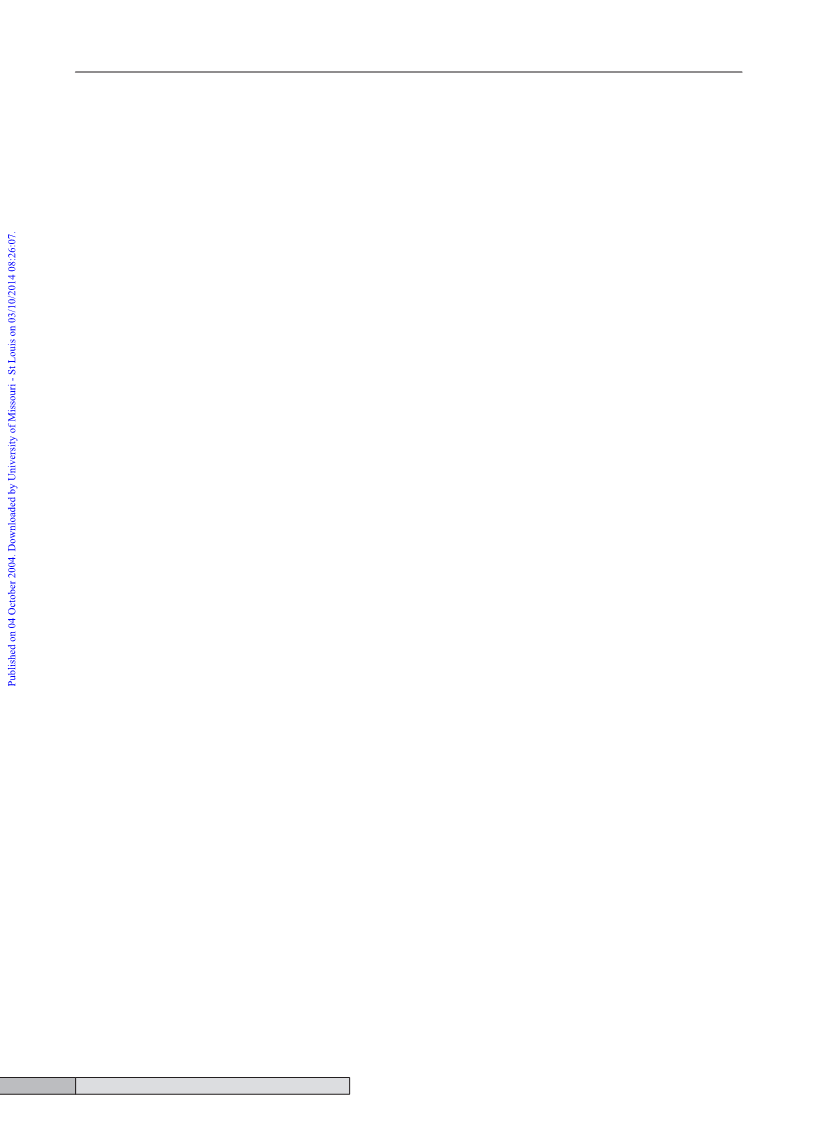
3J(HH) = 6.8 Hz, CH(CH3)2), 2.07 (s, 3H, CH3), 2.61 (sept, 1H,
3J(HH) = 6.8 Hz, CH(CH3)2), 4.37 (br, 12H, CH2), 6.20 and 6.56
(d, 2H each, J(HH) = 3.8 Hz, CH of p-cymene); dC (CD3OD)
D. W. Bruce, Coord. Chem. Rev., 2003, 241, 1; (q) T. V. RajanBabu,
Y.-Y. Yan and S. Shin, Curr. Org. Chem., 2003, 7, 1759.
3 For reviews on catalytic allylic alcohols isomerization, see:
(a) R. Uma, C. Crévisy and R. Grée, Chem. Rev., 2003, 103, 27;
(b) R. C. van der Drift, E. Bouwman and E. Drent, J. Organomet.
Chem., 2002, 650, 1.
3
16.34 (s, CH3), 19.58 (s, CH(CH3)2), 29.76 (s, CH(CH3)2), 55.77
(m, CH2), 86.44 and 92.88 (s, CH of p-cymene), 92.88 and
124.17 (s, C of p-cymene). 3c: Yield: 0.518 g, 89% (Found: C,
37.33; H, 6.09. RuC18H36O6Cl2P2 requires C, 37.12; H, 6.23%);
conductivity (MeOH, 20 °C) 99 X−1 cm2 mol−1; IR (KBr)/cm−1
460w, 547w, 576m, 668w, 721w, 758m, 798w, 878s, 1013s, 1037s,
1177m, 1294w, 1388s, 1441s, 1636m, 2906w, 2925m, 2972w,
3059m, 3263s; dP (CD3OD) 30.29 (s); dH (CD3OD) 2.26 (s, 18H,
CH3), 4.35 (br, 12H, CH2); dC (CD3OD) 16.33 (s, CH3), 57.22 (m,
CH2), 106.08 (t, 2J(CP) = 1.5 Hz, C6Me6).
4 The following catalytic systems have been used for the isomerization
of allylic alcohols into carbonyl compounds in water or mixtures of
water/organicsolvent:(a) [Rh(CO)2(l-Cl)]2:H. AlperandK. Hachem,
J. Org. Chem., 1980, 45, 2269; (b) [Ru(H2O)6][Tos]2 (Tos = p-
toluenesulfonate): D. V. McGrath, R. H. Grubbs and J. W. Ziller, J.
Am. Chem. Soc., 1991, 113, 3611; D. V. McGrath and R. H. Grubbs,
Organometallics, 1994, 13, 224; (c) [Ni(COD)2]/DPPBTS (COD =
1,5-cyclooctadiene; DPPBTS = tetrasulfonated 1,4-bis(diphenyl-
phosphino)butane): H. Bricout, E. Monflier, J.-F. Carpentier and
A. Mortreux, Eur. J. Inorg. Chem., 1998, 1739; (d) [Na][RhCl-
(COD)(PAr3)]/onium salt (PAr3 = carboxylated triarylphosphines):
H. Schumann, V. Ravindar, L. Meltser, W. Baidossi, Y. Sasson
and J. Blum, J. Mol. Catal. A, 1997, 118, 55; (e) RhCl3/TPPTS
and Rh2(SO4)3/TPPTS (TPPTS = P(m-C6H4SO3Na)3): C. de
Bellefon, S. Caravieilhes and E. G. Kuntz, C. R. Acad. Sci., Ser. IIc:
Chim., 2000, 3, 607; C. de Bellefon, N. Tanchoux, S. Caravieilhes,
P. Grenouillet and V. Hessel, Angew. Chem., Int. Ed., 2000, 39,
General procedure for the catalytic isomerization of allylic
alcohols under biphasic conditions. In a Schlenk tube, the
ruthenium catalyst precursor (0.04 mmol) and Cs2CO3 (0.026 g,
0.08 mmol) were dissolved, under inert atmosphere, in water
(10 cm3). A solution of the corresponding allylic alcohol
(4 mmol) in n-heptane (10 cm3) was then added and the reac-
tion mixture stirred at 75 °C for the indicated time. The course
of the reaction was monitored by regular sampling and analysis
of the organic phase by gas chromatography. The identity of
the resulting saturated carbonyl compound was assessed by
comparison with commercially available pure samples and by
their fragmentation in GC/MS. Catalyst recycling: the reaction
mixture was allowed to reach room temperature, the aqueous
phase was then separated under inert atmosphere and washed
twice with 5 cm3 of n-heptane. To the aqueous phase a new solu-
tion of the corresponding allylic alcohol (4 mmol) in n-heptane
(10 cm3) was added.
3442;
(f) [Rh(COD)(SULPHOS)]
(SULPHOS = −O3S(C6H4)-
CH2C(CH2PPh2)3): C. Bianchini, A. Meli and W. Oberhauser,
New J. Chem., 2001, 25, 11; (g) mer-[RuCl3(DMSO)(phen)] and
cis,cis-[RuCl2(DMSO)2(phen)]
(DMSO = dimethyl
sulfoxide;
phen = 1,10-phenanthroline): R. C. van der Drift, J. W. Sprengers,
E. Bouwman, W. P. Mul, H. Kooijman, A. L. Spek and E. Drent,
Eur. J. Inorg. Chem., 2002, 2147; (h) [Rh(COD)(TPPMP)2][BF4]
(TPPMP = Ph2P(p-C6H4PO3Na2)): D. A. Knight and T. L. Schull,
Synth. Commun., 2003, 33, 827; (i) [Ru(g3 :g2 :g3-C12H18)Cl2]
(C12H18 = dodeca-2,6,10-triene-1,12-yl):
V. Cadierno,
S. E.
García-Garrido and J. Gimeno, Chem. Commun., 2004, 232.
5 Tamdem isomerization–aldol and Mannich type reactions of allylic
alcohols with aldehydes and imines, respectively, catalyzed by
[RuCl2(PPh3)3] in water have been reported. See, for example: X.-F.
Yang, M. Wang, R. S. Varma and C.-J. Li, J. Mol. Catal. A, 2004,
214, 147, and references therein.
6 (a) Polystyryldiphenylphosphine-anchored [RuCl2(PPh3)3] has been
used as a recyclable catalyst for the isomerization of allylic alcohols
in organic solvents: A. Zoran, Y. Sasson and J. Blum, J. Org. Chem.,
1981, 46, 255; (b) A recycable Pd-catalyzed isomerization of allylic
alcohols in molten tetrabutylammonium bromide has been recently
reported: B. Ganchegui, S. Bouquillon, F. Hénin and J. Muzart,
J. Mol. Catal. A, 2004, 214, 65.
7 We note that the highest TON and TOF values reported to date
for allylic alcohols isomerization have been attained using the
ruthenium complexes [Ru(g3 :g2 :g3-C12H18)Cl2] (TON up to 106,
TOF up to 50000 h−1), [Ru(g5-C5H5)(PCy3)(NCMe)2][PF6] (TON up
to 3000, TOF up to 36000 h−1) and [RuCl(g5-C5H5)(PPh3)2] (TON
up to 6000, TOF up to 200000 h−1). See ref. 4i and (a) C. Slugovc,
E. Rüba, R. Schmid and K. Kirchner, Organometallics, 1999, 18,
4230; (b) R. C. van der Drift, M. Vailati, E. Bouwman and E. Drent,
J. Mol. Catal. A, 2000, 159, 163.
General procedure for the catalytic hydration of terminal
alkynes. A solution of the corresponding alkyne (1 mmol) and
the appropriate ruthenium precursor (0.05 mmol) in a mixture
of propan-2-ol and water (3 cm3/1 cm3) was heated, under inert
atmosphere at 90 °C in a sealed tube, for the indicated time. The
course of the reaction was monitored by gas chromatography.
The identity of the resulting ketones and aldehydes was assessed
by comparison with commercially available pure samples and by
their fragmentation in GC/MS.
Acknowledgements
We are indebted to the Ministerio de Ciencia y Tecnología
(MCyT) of Spain (Projects BQU2000-0227 and BQU2003-
00255) and the Gobierno del Principado de Asturias (Project
PR-01-GE-6) for financial support. S. E. G.-G. thanks the
MCyT for the award of a PhD grant. V. C. and P. C. thank also
the MCyT for “Ramón y Cajal” contracts.
8 A short account on platinum metals complexes containing
P(CH2OH)3 has been published: P. G. Pringle and M. B. Smith,
Platinum Met. Rev., 1990, 34, 74.
9 For reviews on the chemistry of [{Ru(g6-arene)(l-Cl)Cl}2] dimers,
see: (a) H. Le Bozec, D. Touchard and P. H. Dixneuf, Adv.
Organomet. Chem., 1989, 29, 163; (b) M. A. Bennett, in Compre-
hensive Organometallic Chemistry II, ed. E. W. Abel, F. G. A. Stone
and G. Wilkinson, Pergamon Press, Oxford, 1995, vol. 7, p. 549;
(c) M. A. Bennett, Coord. Chem. Rev., 1997, 166, 225; (d) F. C. Pigge
and J. J. Coniglio, Curr. Org. Chem., 2001, 5, 757.
10 For recent reviews, see: (a) T. Naota, H. Takaya and S.-I. Murahashi,
Chem. Rev., 1998, 98, 2599; (b) B. M. Trost, F. D. Toste and
A. B. Pinkerton, Chem. Rev., 2001, 101, 2067; (c) V. Ritleng, C. Sirlin
and M. Pfeffer, Chem. Rev., 2002, 102, 1731.
11 Hydrogenation of sorbic acid: B. Drießen-Hölscher and J. Heinen,
J. Organomet. Chem., 1998, 570, 141.
12 (a) M. Bianchi, P. Frediani, A. Salvini, L. Rosi, L. Pistolesi,
F. Piacenti, S. Ianelli and M. Nardelli, Organometallics, 1997, 16,
482; (b) Hydrogenation of carboxylic acids: A. Salvini, P. Frediani,
M. Bianchi, F. Piacenti, L. Pistolesi and L. Rosi, J. Organomet.
Chem., 1999, 582, 218.
References
1 (a) F. Joó, in Aqueous Organometallic Catalysis, Kluwer, Dordrecht,
2001; (b) Aqueous-Phase Organometallic Catalysis. Concepts and
Applications, ed. B. Cornelis and W. A. Herrmann, Wiley-VCH,
Weinheim, 1998; (c) Aqueous Organometallic Chemistry and
Catalysis (NATO ASI 3/5), ed. I. T. Horváth and F. Joó, Kluwer,
Dordrecht, 1995.
2 For specific reviews and personal accounts, see: (a) T. Kiji and
T. Okano, TrendsOrg. Chem., 1991, 2, 41;(b) P. KalckandF. Monteil,
Adv. Organomet. Chem., 1992, 34, 219; (c) W. A. Herrmann and
C. W. Kohlpaintner, Angew. Chem., Int. Ed. Engl., 1993, 32, 1524;
(d) G. Papadogianakis and R. A. Sheldon, New J. Chem., 1996, 20,
175; (e) G. Papadogianakis and R. A. Sheldon, Catalysis, 1997,
13, 114; (f) B. E. Hanson, Coord. Chem. Rev., 1999, 185–186, 795;
(g) D. Sinou, Top. Curr. Chem., 1999, 206, 41; (h) F. Fringuelli,
O. Piermatti, F. Pizzo and L. Vaccaro, Eur. J. Org. Chem., 2001, 439;
(i) U. M. Lindström, Chem. Rev., 2002, 102, 2751; (j) F. Joó, Acc.
Chem. Res., 2002, 35, 738; (k) S. Kobayashi and K. Manabe, Acc.
Chem. Res., 2002, 35, 209; (l) R. A. Sheldon, I. W. C. E. Arends,
G. ten Brink and A. Dijksman, Acc. Chem. Res., 2002, 35, 209;
(m) K. Manabe and S. Kobayashi, Chem. Eur. J., 2002, 8, 4094;
(n) T. Dwars and G. Oehme, Adv. Synth. Catal., 2002, 344, 239;
(o) D. Sinou, Adv. Synth. Catal., 2002, 344, 221; (p) N. Pinault and
13 (a) L. Higham, A. K. Powell, M. K. Whittlesey, S. Wocadlo and
P. T. Wood, Chem. Commun., 1998, 1107; (b) Hydrogenation of
supercritical carbon dioxide: Y. Kayaki, T. Suzuki and T. Ikariya,
Chem. Lett., 2001, 1016.
14 We note that P(CH2OH)3 has been used for the removal of residual
ruthenium from RCM reaction products: H. D. Maynard and
R. H. Grubbs, Tetrahedron Lett., 1999, 40, 4137.
3 6 4 0
D a l t o n T r a n s . , 2 0 0 4 , 3 6 3 5 – 3 6 4 1

 Cadierno, Victorio
Cadierno, Victorio
 Crochet, Pascale
Crochet, Pascale
 Garcia-Garrido, Sergio E.
Garcia-Garrido, Sergio E.
 Gimeno, Jose
Gimeno, Jose