Paper
RSC Advances
unreacted nitrosobenzene and phenylhydroxylamine. The 12 R. J. Kalbasi, A. A. Nourbakhsh and F. Babaknezhad, Catal.
absence of nitrosobenzene and phenylhydroxylamine in the Commun., 2011, 12, 955.
reaction mixture signies that they get very strongly adsorbed 13 J. N. Solanki and Z. V. P. Murthy, Ind. Eng. Chem. Res., 2011,
59
onto the catalyst surface and react before desorption. From
these results, it is concluded that the condensation route is 14 H. Wena, K. Yao, Y. Zhang, Z. Zhou and A. Kirschning, Catal.
disfavored over the direct route. Indeed, intermediates of the Commun., 2009, 19, 1207.
direct route are completely reduced in a shorter time, and the 15 J. F. Quinn, C. E. Bryant, K. C. Golden and B. T. Gregg,
high rate of reduction of nitrosobenzene and phenylhydroxyl- Tetrahedron Lett., 2010, 51, 786.
amine could also explain the absence of condensation between 16 R. J. Rahaim, J. Robert and E. Maleczka, Org. Lett., 2005, 7,
50, 7338.
these two products to give azoxybenzene.
5087.
1
7 P. Veerakumara, M. Velayudhamb, K. L. Lub and
S. Rajagopala, Appl. Catal., A, 2012, 439–440, 197.
8 A. Rajapakse and K. S. Gates, J. Org. Chem., 2012, 77, 3531.
9 M. B. Gawande, H. Guo, A. K. Rathi, P. S. Branco, Y. Chen,
R. S. Varmad and D. L. Peng, RSC Adv., 2013, 3, 1050.
0 M. A. Harrad, B. Boualy, L. El Firdoussi, A. Mehdi, C. Santi,
S. Giovagnoli, M. Nocchetti and M. A. Ali, Catal. Commun.,
Conclusion
1
1
In conclusion, as a hierarchical zeolite, mZSM-5 was success-
fully synthesized using KIT-6 as a mesoporous silica source,
modied with Ni nanoparticles and exhibited good catalytic
performance in reduction of nitro aromatic compounds at room
temperature in an aqueous media.
2
2
013, 32, 92.
2
1 R. J. Kalbasi, N. Mosaddegh and A. Abbaspourrad, Appl.
Catal., A, 2012, 423–424, 78.
2 R. J. Kalbasi, N. Mosaddegh and A. Abbaspourrad,
Tetrahedron Lett., 2012, 53, 3763.
3 D. shah and H. Kaur, J. Mol. Catal. A: Chem., 2014, 381, 70.
4 M. Jacquin, D. J. Jones, J. Roziere, S. Albertazzi, A. Vaccari,
M. Lenarda, L. Storaro and R. Ganzerla, Appl. Catal., A,
N
2
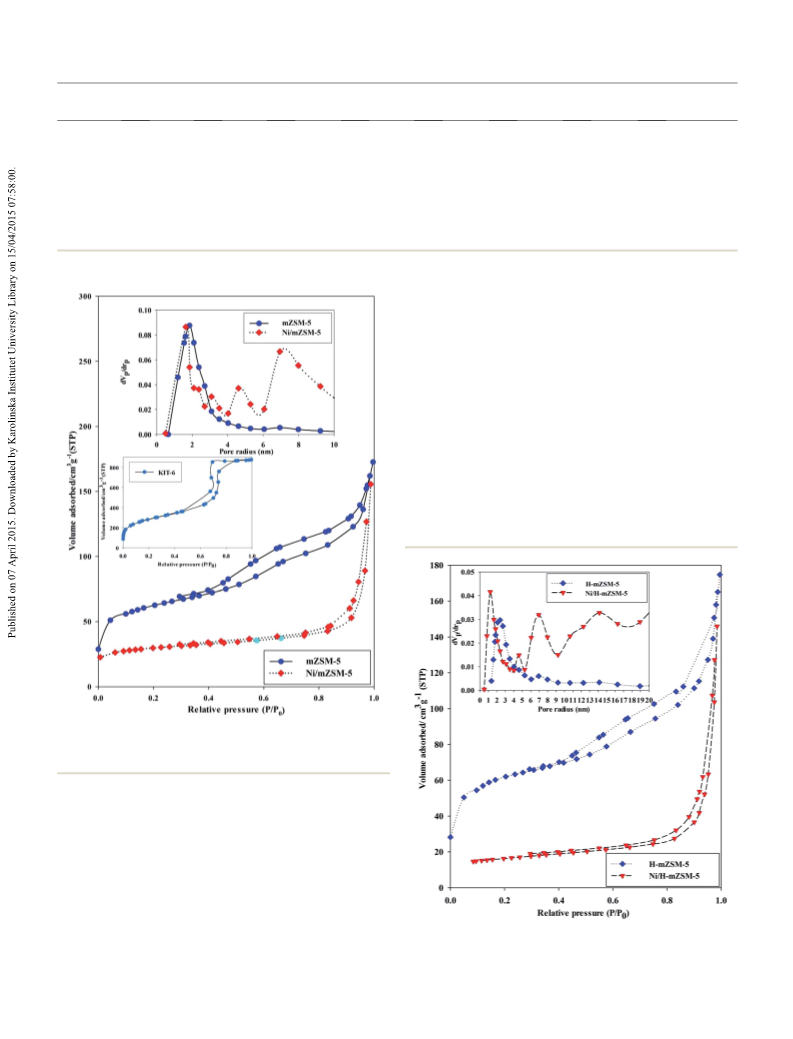
adsorption–desorption isotherms showed that Ni/mZSM-
5
has a mesoporous-microporous structure. The existence of the
2
mesopores in the zeolite structure accelerates the molecular
transportation and also causes high accessibility of acid sites of
the zeolite. These features increase the activity/stability of the
catalyst. A comparison between Ni/mZSM-5 and Ni/H-mZSM-5
showed superior activities of Ni/H-mZSM-5 catalyst, which is
possibly due to the existence of more acidic sites in the Ni/H-
mZSM-5 structure rather than Ni/mZSM-5. Additionally, upon
the completion of the reactions, the catalysts could be removed
from the reaction medium easily and reused for several cycles
without any signicant decrease in the catalytic activity/
stability. These unique features open new perspectives for the
application of these types of hierarchical zeolites in other
organic reactions instead of using traditional zeolites.
2
2
2
003, 251, 131.
5 A. Hakki, R. Dillert and D. W. Bahnemann, ACS Catal., 2013,
, 565.
2
2
2
3
6 R. J. Kalbasi, A. R. Massah and A. Shaei, J. Mol. Catal. A:
Chem., 2011, 335, 51.
7 F. Ocampoa, J. A. Cunhab, M. R. de Lima Santosb,
J. P. Tessonnierc, M. M. Pereirab and B. Louis, Appl. Catal.,
A, 2010, 390, 102.
2
8 Y. Tao, H. Kanoh and K. Kaneko, J. Am. Chem. Soc., 2003, 125,
6
044.
9 K. Na, M. Choi and R. Ryoo, Microporous Mesoporous Mater.,
013, 166, 3.
2
Notes and references
2
1
2
3
4
D. M. Dotzauer, S. Bhattacharjee, Y. Wen and M. L. Bruening, 30 K. Egeblad, C. H. Christensen, M. Kustova and
Langmuir, 2009, 25, 1865. C. H. Christensen, Chem. Mater., 2008, 20, 946.
D. Cantillo, M. M. Moghaddam and C. O. Kappe, J. Org. 31 J. C. Groen, T. Bach, U. Ziese, A. M. Paulaime-van Donk,
Chem., 2013, 78, 4530.
K. P. de Jong, J. A. Moulijn and J. P ´e rez-Ram ´ı rez, J. Am.
Chem. Soc., 2005, 127, 10792.
G. W. Lamb, F. A. Al-Badran, J. M. J. Williams and
S. T. Kolaczkowski, Chem. Eng. Res. Des., 2010, 88, 1533.
J. Zhou, J. Dai, G. Q. Bian and C. Y. Li, Coord. Chem. Rev.,
32 R. Chal, R. Cacciaguerra, S. van Donk and C. Ge-radin, Chem.
Commun., 2010, 46, 7840.
2
009, 253, 1221.
33 K. Na and G. A. Somorjai, Catal. Lett., 2015, 145, 193.
34 J. Perez-Ramırez, C. H. Christensen, K. Egeblad,
C. H. Christensen and J. C. Groen, Chem. Soc. Rev., 2008,
37, 2530.
5
6
R. Shen and S. A. Andrews, Water Res., 2013, 47, 2446.
A. Kumar, D. Nepak and D. Srinivas, Catal. Commun., 2013,
3
7, 36.
7
8
9
X. F. Wu, A. Petrosyan, T. V. Ghochikyan, A. S. Saghyan and 35 S. L. Orozco, A. Inayat, A. Schwab, T. Selvam and
P. Langer, Tetrahedron Lett., 2013, 54, 3158.
W. Schwieger, Adv. Mater., 2011, 23, 2602.
F. C. Lizana, S. G. Quero and M. A. Keane, Catal. Commun., 36 J. Zheng, X. Zhang, Y. Zhang, J. Maa and R. Li, Microporous
2008, 9, 475.
Mesoporous Mater., 2009, 122, 264.
A. Saha and B. Ranu, J. Org. Chem., 2008, 73, 6867.
37 M. S. Holm, E. Taarning, K. Egeblad and C. H. Christensen,
Catal. Today, 2011, 168, 3.
1
0 S. Farhadi and F. Siadatnasab, J. Mol. Catal. A: Chem., 2011,
39, 108.
1 S. Gowda, B. K. K. Gowda and D. C. Gowda, Synth. Commun.,
003, 33, 281.
3
38 D. P. Serrano, R. A. Garcia, G. Vicente, M. Linares,
D. Prochazkova and J. Cejka, J. Catal., 2011, 279, 366.
1
2
This journal is © The Royal Society of Chemistry 2015
RSC Adv., 2015, 5, 34398–34414 | 34413

 Mazaheri, Omid
Mazaheri, Omid
 Kalbasi, Roozbeh Javad
Kalbasi, Roozbeh Javad