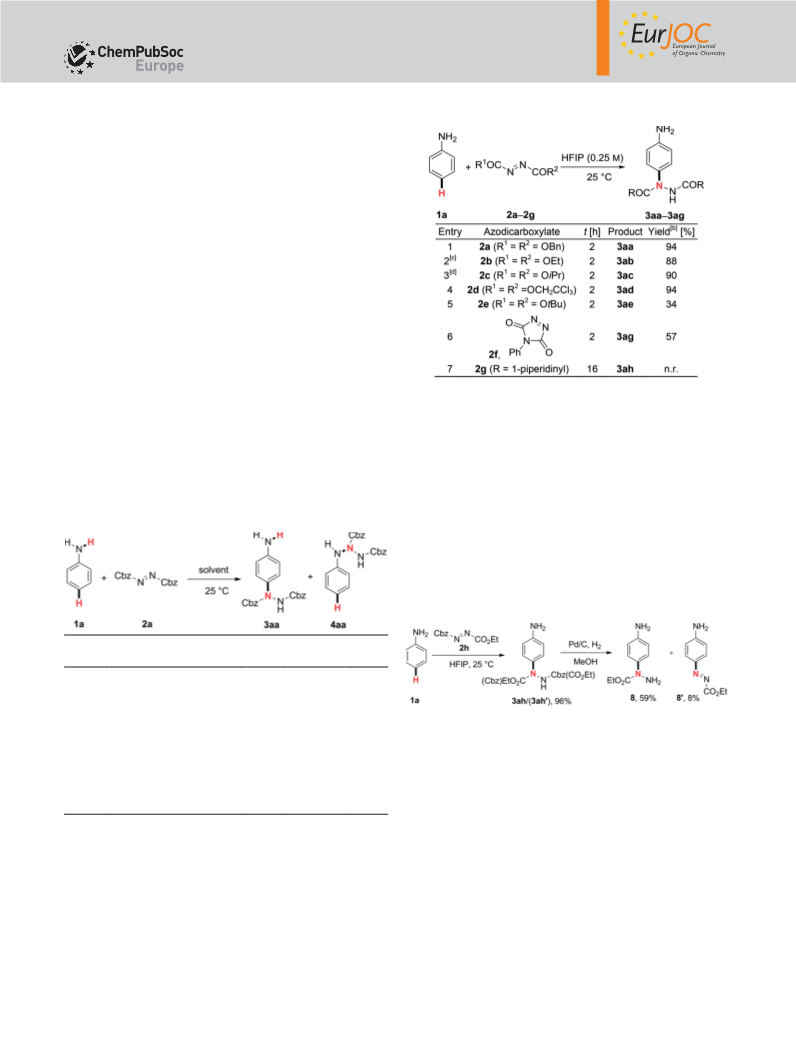
Communication
was obtained in a reasonable yield of 57 % over three steps. desired product 3aa obtained smoothly in 86 % yield, but also
[
15d]
Always in one pot, a Michael addition
was performed with 17 mL of HFIP was recovered after distillation directly from the
methyl acrylate to afford the monoalkylated product 12 in reaction.
moderate yield.
Conclusions
We developed an effective, mild, and clean method for C–H
amination between azodicarboxylate derivatives and a wide
range of anilines in 1,1,1,3,3,3-hexafluoro-2-propanol. The de-
sired products were formed smoothly at room temperature
without any additional catalyst or reagent in short times. This
reaction was highly regioselective, provided para-substituted
anilines, and was shown to have a broad substrate scope. The
compatibility of a free amino group in this procedure is of high
synthetic value. Besides, because of its low boiling point (b.p.
5
9 °C) and low viscosity, HFIP can be easily recovered and re-
used.
Experimental Section
General Procedure: Azodicarboxylate 2a (0.55 mmol, 1.1 equiv.)
was added to a stirred solution of aniline (1a; 0.5 mmol) in HFIP
(2 mL) at 25 °C. The mixture was stirred at this temperature for 2 h.
Upon completion of the reaction (TLC monitoring), the mixture was
concentrated under reduce pressure to give a crude product. Then,
the crude product was purified by column chromatography (silica
gel; cyclohexane/diethyl ether, 2:1) to afford 3aa as a white solid.
Scheme 4. Synthetic utilities of this reaction. PTSA = para-toluenesulfonic
acid.
[
9e,10c,10e,16]
Acknowledgments
On the basis of previous literature reports
studies,
and our
[
13,15d]
a plausible H-bond activation of the azodicarbox-
Central Glass Co. Ltd. is gratefully acknowledged for kindly pro-
viding HFIP. R.-J. T. is grateful for a doctoral fellowship from the
Chinese Scholarship Council.
ylate by HFIP is proposed (Scheme 5). Then, nucleophilic attack
of aniline (1a) on 2a generates intermediate 13, which subse-
quently undergoes hydrogen transfer to afford the expected
product 3aa. Owing to HFIP–azodicarboxylate association, anil-
ine can only react in its para position, which is sterically less
hindered than the ortho position.
Keywords: C–H activation · Amination · Regioselectivity ·
Azodicarboxylates · Fluorine
[
1] a) D. N. Zalatan, J. D. Bois, Org. Process Res. Dev. 2009, 292, 347–378; b)
T. G. Driver, Org. Biomol. Chem. 2010, 8, 3831–3846; c) J. D. Bois, Org.
Process Res. Dev. 2011, 15, 758–762; d) J. L. Roizen, M. E. Harvey, J. D.
Bois, Acc. Chem. Res. 2012, 45, 911; e) M. L. Louillat, F. W. Patureau, Chem.
Soc. Rev. 2014, 43, 901–910; f) K. Shin, H. Kim, S. Chang, Acc. Chem. Res.
2
015, 48, 1040–1052; g) G. He, B. Wang, W. A. Nack, G. Chen, Acc. Chem.
Res. 2016, 49, 635–645; h) J. Jiao, K. Murakami, K. Itami, ACS Catal. 2016,
, 610–633; i) X. Dong, Q. Liu, Y. Dong, H. Liu, Chem. Eur. J. 2017, 23,
6
Scheme 5. Proposed mechanism.
2481–2511; j) Y. Park, Y. Kim, S. Chang, Chem. Rev. 2017, 117, 9247–9301.
2] a) R.-J. Tang, C.-P. Luo, L. Yang, C.-J. Li, Adv. Synth. Catal. 2013, 355, 869–
[
8
73; b) H. J. Kim, M. J. Ajitha, Y. Lee, J. Ryu, H. Kim, Y. Lee, Y. Jung, S.
A gram-scale reaction was also evaluated (Scheme 6). The
para-selective amination of 1a with 2a was performed on an
1.0 mmol scale in 20 mL of HFIP for 2 h. Not only was the
Chang, J. Am. Chem. Soc. 2014, 136, 1132–1140; c) T. Matsubara, S. Asako,
L. Ilies, E. Nakamura, J. Am. Chem. Soc. 2014, 136, 646–649; d) D. Zhu, G.
Yang, J. He, L. Chu, G. Chen, W. Gong, K. Chen, M. D. Eastgate, J. Q. Yu,
Angew. Chem. Int. Ed. 2015, 54, 2497–2500; Angew. Chem. 2015, 127,
1
2
5
527–2530; e) X.-H. Hu, X.-F. Yang, T.-P. Loh, ACS Catal. 2016, 6, 5930–
934; f) Z. Dong, G. Dong, J. Am. Chem. Soc. 2013, 135, 18350–18353; g)
P. Wang, G. C. Li, P. Jain, M. E. Farmer, J. He, P. X. Shen, J. Q. Yu, J. Am.
Chem. Soc. 2016, 138, 14092–14099; h) K. Sun, Y. Li, T. Xiong, J. Zhang,
Q. Zhang, J. Am. Chem. Soc. 2011, 133, 1694–1697; i) B. Berzina, I. Soko-
lovs, E. Suna, ACS Catal. 2015, 5, 7008–7014.
[
3] a) J. S. Arnold, H. M. Nguyen, J. Am. Chem. Soc. 2012, 134, 8380–8383;
b) X. Li, X. Li, N. Jiao, J. Am. Chem. Soc. 2015, 137, 9246–9249; c) M. S.
Mayo, X. Yu, X. Zhou, X. Feng, Y. Yamamoto, M. Bao, Org. Lett. 2014, 16,
Scheme 6. Gram-scale reaction.
Eur. J. Org. Chem. 2017, 4753–4757
www.eurjoc.org
4756
© 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Tang, Ren-Jin
Tang, Ren-Jin
 Milcent, Thierry
Milcent, Thierry
 Crousse, Benoit
Crousse, Benoit