10.1002/cplu.201900377
ChemPlusChem
FULL PAPER
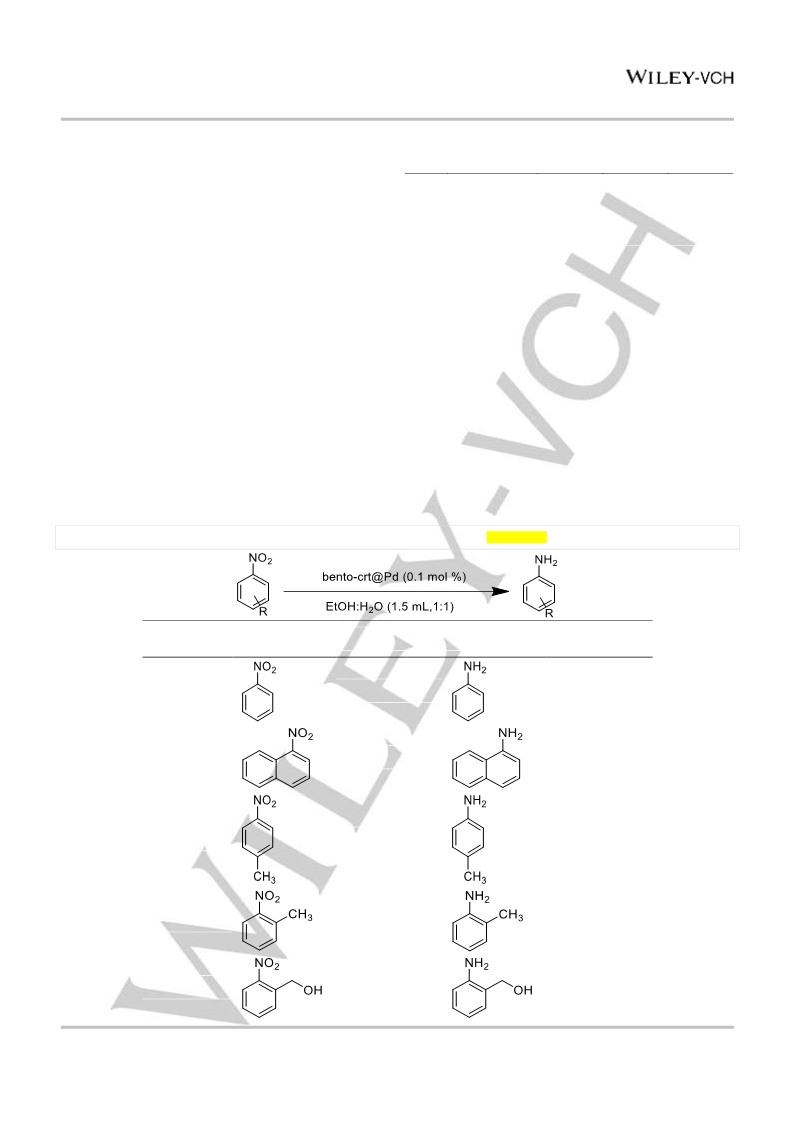
EtOH (1.5 mL) were added. The reaction mixture was stirred at room
temperature for the appropriate reaction times, and the progress of the
reactions was monitored by GC, TLC and or 1H NMR. After completion of
the reaction, the crude product was extracted with ethyl acetate (3×5 mL)
and further purified using column or plate chromatography.
[6]
[7]
[8]
a) A. Ghaderi, M. Gholinejad, H. Firouzabadi, Curr. Org. Chem. 2016, 20,
327-348; b) O. Ayodele, B. Hameed, J. Ind. Eng. Chem. 2013, 19, 966-
974; c) G. Nagendrappa, Appl. Clay Sci. 2011, 53, 106-138; d) A. Gil, S.
Korili, M. Vicente, Catal. Rev. 2008, 50, 153-221; e) R. B. Achma, A.
Ghorbel, A. Dafinov, F. Medina, Appl. Catal., A 2008, 349, 20-28.
a) M. Nasrollahzadeh, S. M. Sajadi, M. Maham, I. Kohsari, Microporous
and Mesoporous Mater. 2018, 271, 128-137; b) M. Moosavi, Iran. J.
Public Health 2017, 46, 1176; c) D. D. Eisenhour, R. K. Brown, Elements.
2009, 5, 83-88; d) J. W. Hosterman, Bentonite and Fuller's earth
resources of the United States,1985.
Acknowledgements
The authors are grateful to Institute for Advanced Studies in Basic
Sciences (IASBS) Research Council and Iran National Science
Foundation (INSF-Grant number of 97021804) for support of this work. We
also thankful to the Spanish Ministerio de Economía, Industria
Competitividad, AgenciaEstatal de Investigación (AEI) and Fondo
Europeo de Desarrollo Regional (FEDER, EU) (projects CTQ2016-76782-
a) Y. Jiang, T. Huang, Y. Xu, X. Li, Z. Qin, H. Ji, Chem. Eng. Technol.
2018, 41, 175-181; b) M. Gholinejad, R. Bonyasi, C. Najera, F. Saadati,
M. Bahrami, N. Dasvarz, ChemPlusChem 2018, 83, 431 – 438; c) L.Yuan,
Pol. J. Environ. Stud. 2017, 26, 2355-2361; d) V. M. Abbasov, H. C.
Ibrahimov, G. S. Mukhtarova, M. I. Rustamov, E. Abdullayev, Energy
Fuels 2017, 31, 5840-5843; e) Y. Jiang, X. Li, Z. Qin, H. Ji, Chin. J ,
Chem. Eng. 2016, 24, 1195-1200; f) H. Gaffour, M. Mokhtari, Res. Chem.
Intermed. 2016, 42, 6025-6038.
y
P
and
CTQ2016-81797-REDC),
the
Generalitat
Valenciana
(PROMETEOII/2014/017) and the University of Alicante for financial
support.
[9]
a) D. Gangopadhyay, S. K. Singh, P. Sharma, H. Mishra, V. Unnikrishnan,
B. Singh, R. K. Singh, Spectrochim. Acta, Part A 2016, 154, 200-206; b)
J. Dulinska, Z. Setkowicz, K. Janeczko, C. Sandt, P. Dumas, L. Uram, K.
Gzielo-Jurek, J. Chwiej, Anal. Bioanal. Chem. 2012, 402, 2267-2274.
Keywords: Amino acids • clays • heterogeneous catalysis • palladium • nitro
reduction
[1]
S. Nishimura, Handbook of heterogeneous catalytic hydrogenation for
organic synthesis. Wiley New York etc, 2001.
[10] a) D. Huang, Z. L. Wu, W. Liu, N. Hu, H. Z. Li, Chem. Eng. Process. 2016,
104, 13-21; b) I. Pischel, T. Gastner, Creatine–its chemical synthesis,
chemistry, and legal status, 2007; pp 291-307.
[2]
[3]
Bechamp, A., Ann. Chim. Phys. 1854, 42, 140.
a) B. Zeynizadeh, Z. Shokri, M. Hasanpour Galehban, Appl. Organomet.
Chem. 2019, e4771; b) M. Gholinejad, N. Dasvarz, M. Shojafar, J. M.
Sansano, Inorganica Chim. Acta 2019, 495, 118965; c) J. Feng, S.
Handa, F. Gallou, B. H.Lipshutz, Angew. Chem. 2016, 128, 9125-9129;
d) I. Pogorelić, M. Filipan-Litvić, S. Merkaš, G. Ljubić, I. Cepanec, M.
Litvić, J. Mol. Catal. A: Chem. 2007, 274, 202-207; e) K. Nomura, J. Mol.
Catal. A: Chem. 1995, 95, 203-210; f) M. Miura, M. Shinohara, M.
Nomura, J. Mol. Catal. 1988, 45, 151-153; g) R. Adams, F. Cohen, O.
Rees, J. Am. Chem. Soc. 1927, 49, 1093-1099.
[11] a) H. Firouzabadi, N. Iranpoor, A. Ghaderi, M. Gholinejad, S. Rahimi, S.
Jokar, RSC Adv. 2014, 4, 27674-27682; b) K. Datta, M. Eswaramoorthy,
C. Rao, J. Mater. Chem. 2007, 17, 613-615.
[12] a) Z. Issaabadi, M. Nasrollahzadeh, S. M. Sajadi, J. Cleaner Prod. 2017,
142, 3584-3591; b) G. Ding, W. Wang, T. Jiang, B. Han, Green Chem.
2013, 15, 3396-3403.
[13] M. Gholinejad, M. Afrasi, N. Nikfarjam, C. Nájera, Appl. Catal., A 2018,
563, 185-195.
[14] C. Nithya, S. B. Devi, S. Gopukumar, J. Mater. Sci. 2012, 47, 6784-6791.
[15] Y. Zhang, L. Chen, J.-J. Zhao, H.-B. Chen, M.-X. He, Y.-P. Ni, J.-Q. Zhai,
X.-L. Wang, Y.-Z. Wang, Polym. Chem. 2014, 5, 1982-1991.
[16] Q. Liu, J. Li, Z. Zhou, J. Xie, J. Y. Lee, Sci. Rep. 2016, 6, 19593.
[17] a) V. Dalmoro, J. H. dos Santos, I. M. Baibich, I. S. Butler, E. Armelin, C.
Alemán, D. S. Azambuja, Prog. Org. Coat. 2015, 80, 49-58; b) Y. Zhang,
X. He, J. Ouyang, H. Yang, Sci. Rep. 2013, 3, 2948.
[4]
a) Z. Li, D. Zhao, J. Luo, Inorg. Chem. Commun. 2019, 100, 1-5; b) H.
Pang, F. Gallou, H. Sohn, J. Camacho-Bunquin, M. Delferro, B. H.
Lipshutz, Green Chem. 2018, 20, 130-135; c) M. Gholinejad, F. Zareh,
C. Nájera, Appl Organometal Chem. 2017, e3984; d) B. S. Kumar, A. J.
Amali, K. Pitchumani, J. Mol. Catal. A: Chem. 2016, 423, 511-519; e) A.
B. Dongil, L. Pastor-Pérez, J. L. G. Fierro, N. Escalona, A. Sepúlveda-
Escribano, Appl. Catal., A 2016, 513, 89-97; f) M. Shokouhimehr,
Catalysts 2015, 5, 534-560; g) H. K. Kadam, S. G. Tilve, RSC Adv. 2015,
5, 83391-83407; h) T. Jiang, S. Du, T. Jafari, W. Zhong, Y. Sun, W. Song,
Z. Luo, W. A. Hines, S. L. Suib, Appl. Catal., A 2015, 502, 105-113; i) B.
Karimi, F. Mansouri, H. Vali, ChemPlusChem 2015, 80, 1750-1759; j) A.
Kumar, K. Purkait, S. K. Dey, A. Sarkar, A. Mukherjee, RSC Adv. 2014,
4, 35233-35237; k) P. Wang, H. Liu, J. Niu, R. Li, J. Ma, Catal. Sci.
Technol. 2014, 4, 1333-1339; l) M. Baron, E. Metay, M. Lemaire, F.
Popowycz, Green Chem. 2013, 15, 1006-1015; m) M. L. Kantam, R.
Chakravarti, U. Pal, B. Sreedhar, S. Bhargava, Adv. Synth. Catal. 2008,
350, 822-827.
[18] A. Kumar, S. Samanta, S. Latha, A. Debnath, A. Singh, K. Muthe, H. C.
Barshilia, RSC Adv. 2017, 7, 4135-4143.
[19] S. Yuan, J. Gu, Y. Zheng, W. Jiang, B. Liang, S. O. Pehkonen, J. Mater.
Chem. A 2015, 3, 4620-4636.
[20] a) T. Lu, R. Zhang, C. Hu, F. Chen, S. Duo, Q. Hu, Phys. Chem. Chem.
Phys. 2013, 15, 12963-12970; b) A. Lopez-Santiago, H. R. Grant, P.
Gangopadhyay, R. Voorakaranam, R. A. Norwood, N. Peyghambarian,
Opt. Mater. Express 2012, 2, 978-986.
[21] a) Y. Zhang, Z. Xie, Z. Wang, X. Feng, Y. Wang, A. Wu, Dalton Trans.
2016, 45, 12653-12660; b) E. Mazzotta, S. Rella, A. Turco, C. Malitesta,
RSC Adv. 2015, 5, 83164-83186.
[5]
C. E. Garrett, K. Prasad, Adv. Synth. Catal. 2004, 346, 889-900.
[22] M. Gholinejad, M. Bahrami, C. Nájera, Mol. Catal. 2017, 433, 12-19.
This article is protected by copyright. All rights reserved.

 Gholinejad, Mohammad
Gholinejad, Mohammad
 Rasouli, Zahra
Rasouli, Zahra
 Najera, Carmen
Najera, Carmen
 Sansano, José M.
Sansano, José M.