2
N. Yılmaz Baran / Journal of Molecular Structure 1220 (2020) 128697
capability, large surface area, and a large number of active sites
[14,15]. Although use of MNPs in catalytic systems has recently
attracted tremendous interest of researchers, agglomeration and
size control of nanoparticles drawbacks encountered in fabrication
of MNPs have pushed researchers to be in search of new solutions
[12]. Selection of suitable support material for the immobilization
of MNPs has been stated as one of the best ways to overcome these
problems [16]. Support material affects many catalyst properties
such as activity, recoverability, reusability, and ease of removal of
the catalyst from the reaction media. Moreover, suitable support
material decreases the agglomeration of NPs [17,18]. Thus, it is
highly desirable to generate an efficient support material for the
immobilization of MNPs.
Polyazomethines which are prepared by oxidative poly-
condensation reactions of Schiff base monomers may be effective
and ideal support materials for reduction of nitroarenes. Their high
thermal and mechanical stability, outstanding optic, electro-
chemical, semiconductive, efficient catalyst and antibacterial,
anticancer, and antitumor properties make them highly attractive
as support material [19e23].
The purpose of the present research was to reveal that - a novel
polyazomethine could be an efficient and suitable support material
for immobilization of MNPs and - to fabricate Pd NPs as an effective
and reusable heterogeneous catalyst for easy removal of toxic
nitroarenes from wastewaters. Catalytic tests showed that the
designed Pd NPs were a highly effective, fast, recurrently reusable,
and easily recoverable nanocatalyst for removal of hazardous
nitroarenes from wastewaters.
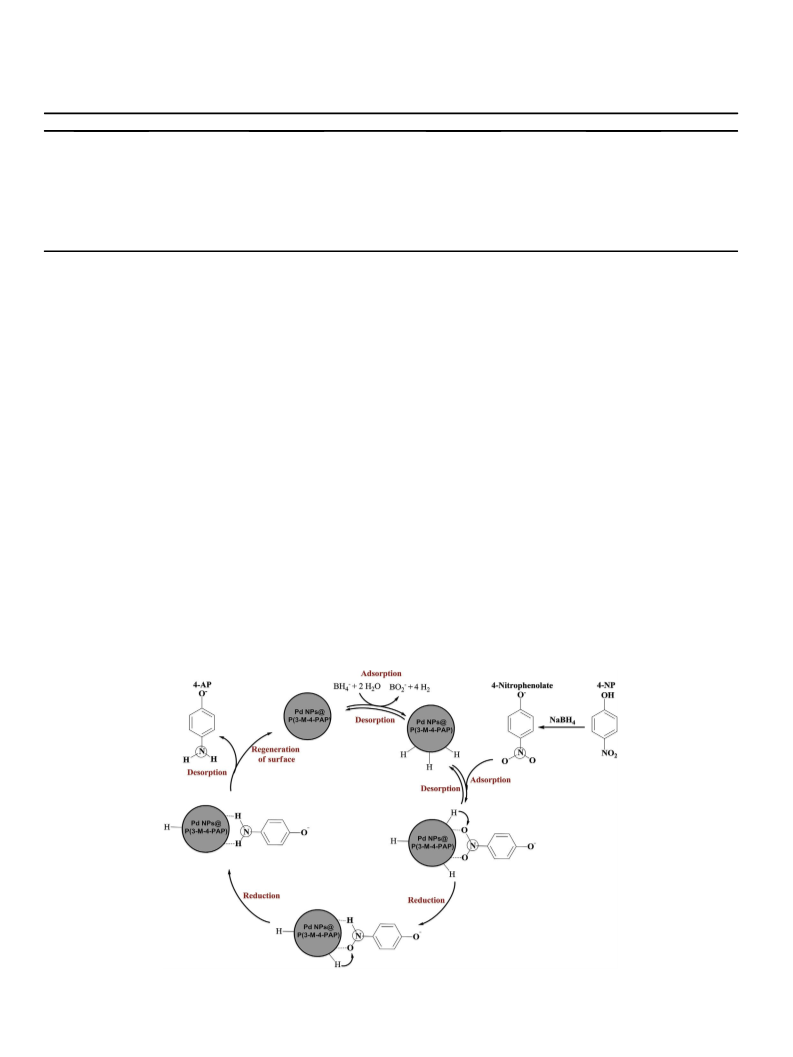
Scheme 1. Preparation of 3-M-4-PAP, P(3-M-4-PAP) and Pd NPs@P(3-M-4-PAP)
catalyst.
2.2.3. Generation of Pd NPs@P(3-M-4-PAP) catalyst
The immobilization of Pd NPs on P(3-M-4-PAP) was carried out
without use of any reducing agent. Briefly, P(3-M-4-PAP) (0.3g) and
PdCl2 (0.1g) were introduced into 25 mL ethanol and refluxed at
70 ꢀC for 24 h. Finally, the nanocatalyst was filtered, washed with
2.2.4. Typical procedure for reduction of nitroarenes
0.1 mL of 0.05 M aqueous NaBH4 solution which was freshly
prepared was added on 1 mL of 1.0 ꢁ 10ꢂ4 M aqueous nitroarene
solution in a tube. After the prepared solution was stirred for 1 min,
2 mg of the fabricated nanocatalyst was added in the produced
solution. The final yellow mixture was stirred until its color became
colorless and this process was also followed by UVeVis analysis.
Then, the nanocatalyst was filtered, rinsed with water (3 ꢁ 50 mL)
and dried.
2. Experimental
2.1. Materials
3-pyridinecarboxaldehyde, 4-amino-3-methylphenol, PdCl2,
NaBH4, 3-nitrophenol, 4-nitrophenol, 2-nitroaniline, 4-nitroaniline,
4-nitro-o-phenylenediamine, 4-nitrobenzoic acid, 3-nitro benzal-
dehyde and 4-nitro benzaldehyde, ethyl acetate, methanol, ethanol,
DMF, and DMSO were obtained from Sigma-Aldrich. NaOCl (15%
aqu.) was also purchased from Pl-dess Chem. Co.
2.3. Characterization
Shimadzu Prominence Gel Permeation Chromatography
attached Nucleogel GPC 103-5 VA300/7.7 column for GPC analysis
was used to determine number average molecular weight (Mn),
weight average molecular weight (Mw) and polydispersity index
(PDI) values of the polymer (eluent: DMF, analysis temperature:
40 ꢀC, flow rate: 0.5 mL/min, detector: refractive index detector
(RID)). Shimadzu UV-1700 PharmaSpec UVeVisible Spectropho-
tometer was used to obtain UVeVis spectra of the generated ma-
terials and nitroarenes. FTIR spectra of the fabricated products were
attained by PerkinElmer FTIR Spectrometer (wavenumber range:
2.2. Method
2.2.1. Synthesis of 3-M-4-PAP
3-M-4-PAP was prepared by stirring 3-pyridinecarboxaldehyde
(0. 96 mL, 10 mmol) and 4-amino-3-methylphenol (1.23 g,
10 mmol) in methanol (25 mL) at 60 ꢀC for 24 h using DeaneStark
apparatus. Additionally, the completion of Schiff base reaction was
followed by FT-IR and TLC techniques. After the reaction, 3-M-4-
PAP was filtered, washed with methanol and it was crystallized
from ethyl acetate twice.
4000-650 cmꢂ1). 1H NMR spectra of the products were recorded by
ꢀ
Bruker Avance 500 MHz NMR in DMSO at 25 C. TGA curves were
obtained via EXSTAR S11 7300 Thermal Analyzer. Crystalline
structure of the designed nanocatalyst was displayed by Rigaku
Smart Lab X-Ray Diffractometer. SEM images and EDS analysis of
the samples were acquired by FEI Quanta 450 FEG FE-ESEM-EDS.
TEM images of Pd NPs@P(3-M-4-PAP) catalyst were attained by
TEM JEOL JEM-2100 (UHR).
2.2.2. Fabrication of P(3-M-4-PAP)
P(3-M-4-PAP) was prepared by oxidative polycondensation re-
action of 3-M-4-PAP in the presence of NaOCl oxidant in an aqueous
alkaline medium (Scheme 1). 1 mmol (0.212 g) of 3-M-4-PAP was
added in 0.2 M 10 mL KOH solution and it was dissolved under
nitrogen atmosphere by refluxing. Then, NaOCl (3 mmol) was
added in the monomer solution for 15 min after the planned
polymerization temperature was reached. The yellow colored
monomer solution was turned into dark brown as soon as NaOCl
was added. After the planned polymerization time was over, the
polymer in the obtained solution was precipitated with 1 M HCl.
Then, the attained polymer was filtered, washed with hot methanol
and water and dried at 80 ꢀC.
3. Results and discussion
3.1. Determination of optimum polymerization conditions
The effects of NaOCl concentration, polymerization time and
temperature on the polymer yield were investigated and the ob-
tained data were given in Fig. S1 (Please refer to Supplementary
data). It was determined that the polymer yield (92.6%) increased

 Y?lmaz Baran, Nuray
Y?lmaz Baran, Nuray