T.B. Ogunbayo, T. Nyokong / Journal of Molecular Catalysis A: Chemical 350 (2011) 49–55
55
4. Conclusions
OH
O
This work showed that PdODPc is capable of both homogeneous
and heterogeneous photo-oxidation of 4-chlorophenol and pen-
tachlorophenol. The oxidation of both substrates was faster under
homogeneous conditions and gave higher percentage conversion
than for heterogeneous catalysis. It was confirmed that singlet oxy-
gen played a role in the phototransformation of both substrates.
Direct oxidation of 4-chlorophenol appeared to result in the for-
mation of benzoquinone while hydroquinone seemed to be the
product from Type I reaction pathway.
1O2
Cl
O
4-chlorophenol
Benzoquinone
radicals
OH
Acknowledgements
This work was supported by the Department of Science and
Technology (DST) and National Research Foundation (NRF) of South
Africa through DST/NRF South African Research Chairs Initiative
for Professor of Medicinal Chemistry and Nanotechnology and
Rhodes University. T.O. thanks African Laser centre for graduate
bursary.
OH
Hydroquinone
Scheme 1. Proposed mechanism for the phototransformation of 4-CP on ads-
PdODPc–SWCNT–COOH.
References
[1] A. Mylonas, E. Papaconstantinou, J. Photochem. Photobiol. A Chem. 94 (1996)
77–82.
presence of oxygen
presence of argon
presence of sodium azide
[2] H. Roques, Chemical Water Treatment, VCH Verlag, Weinheim, Germany,
1996.
[3] A. Mylonas, E. Papaconstantinou, Polyhedron 15 (1996) 3211–3217.
[4] A. Sorokin, B. Meunier, Chem. Eur. J. 2 (1996) 1308–1317.
[5] A. Sorokin, J.-L. Seˇıris, B. Meunier, Science 268 (1995) 1163–1165.
1.20
1.00
0.80
0.60
0.40
0.20
0.00
[6] A. Sorokin, L. Fraisse, A. Rabion, B. Meunier, J. Mol. Catal.
103–114.
A 117 (1997)
[7] S. Rismayani, M. Fukushima, A. Sawada, H. Ichikawa, K. Tatsumi, J. Mol. Catal.
A Chem. 217 (2004) 13–19.
[8] M. Fukushima, K. Tatsumi, Biores. Tech. 97 (2006) 1605–1611.
[9] M. Fukushima, K. Tatsumi, Toxicol. Environ. Chem. 85 (2003) 39–49.
[10] M. Fukushima, K. Tatsumi, Environ. Sci. Technol. 35 (2001) 1771–1778.
[11] N. Nensala, T. Nyokong, J. Mol. Catal. A 164 (2000) 69–76.
[12] E. Chamarro, A. Marco, S. Esplugas, Water. Res. 35 (2001) 1047–1051.
[13] B. Meunier, A. Sorokin, Acc. Chem. Res. 30 (1997) 470–476.
[14] M. Alvaro, E. Carbonell, M. Esplá, H. Garcia, Appl. Catal. B Environ. 57 (2005)
37–42.
[15] V. Iliev, V. Alexiev, L. Bilyarska, J. Mol. Catal. A Chem. 137 (1999) 15–26.
[16] T.B. Ogunbayo, T. Nyokong, J. Mol. Struc. 973 (2010) 96–103.
[17] A. Sun, Z. Xiong, Y. Xu, J. Hazard. Mater. 152 (2008) 191–195.
[18] X. Xue, Y. Xu, J. Mol. Catal. A Chem. 276 (2007) 80–85.
[19] T.B. Ogunbayo, E. Antunes, T. Nyokong, J. Mol. Catal. A Chem. 334 (2011)
123–129.
0
10
20
30
40
50
60
70
Irradiaꢀon ꢀme (min)
Fig. 10. Demonstration of the effect of sodium azide and argon on 4-CP phototrans-
formation rate using ads-PdODPc–SWCNT–COOH.
O
OH
[20] T.B. Ogunbayo, T. Nyokong, J. Mol. Catal. A Chem. 337 (2011) 68–76.
[21] J. Liu, A.G. Rinzler, H. Dai, J.H. Hafner, R.K. Bradley, P.J. Boul, A. Lu, T. Iverson, K.
Shelimov, C.B. Huffman, F. Rodriguez-Macias, T.Y.-S. Shon, R. Lee, D.T. Colbert,
R.E. Smalley, Science 280 (1998) 1253–1256.
[22] T.B. Ogunbayo, A. Ogunsipe, T. Nyokong, Dyes Pigm. 82 (2009) 422–426.
[23] W. Spiller, H. Kliesch, D. Worhle, S. Hackbarth, B. Roder, G. Schnurpfield, J.
Porphyr. Phthalocyan. 2 (1998) 145–158.
1O2
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
O
Cl
[24] K. Ozoemena, N. Kuznetsova, T. Nyokong, J. Mol. Catal. A Chem. 176 (2001)
29–40.
tetrachlorobenzoquinone
pentachlorophenol
[25] F. Wilkinson, J.G. Brummer, J. Phys. Chem. Ref. Data 10 (1981) 825.
[26] H. Al-Ekabi, N. Serpone, J. Phys. Chem. 92 (1988) 5726–5731.
[27] D.D. Dionysiou, A.P. Khodadoust, A.M. Kern, M.T. Suidan, I. Baudin, J.-M. Laîné,
Appl. Catal. B Environ. 24 (2000) 139–155.
Scheme 2. Proposed mechanism for the phototransformation of PCP on ads-
PdODPc–SWCNT–COOH.
[28] K.J. Laider, J.H. Meiser, B.C. Sanctuary, Physical Chemistry, fourth ed., Houghton
Mifflin Company, Boston, 2003, p. 933.
[29] M. Terrones, W.K. Hsu, A. Schwoerer, K. Prassides, H.W. Kroto, D.R. Walton,
Appl. Phys. A 66 (1998) 307–317.
[30] Y. Zhang, X. Sun, L. Pan, H. Li, Z. Sun, C. Sun, B.K. Tay, J. Alloys Compd. 480 (2009)
L17–L19.
[31] K. Wang, J.-J. Xu, K.-S. Tang, H.-Y. Chen, Talanta 67 (2005) 798–805.
[32] Z. Xu, H. Li, K.-Z. Li, Y. Kuang, Y. Wang, Q. Fu, Z. Cao, W. Li, Cryst. Growth Design
9 (2009) 4136–4141.
[33] B.K. Price, J.M. Tour, J. Am. Chem. Soc. 128 (2006) 12899–12904.
[34] J. Jiang, R. Saito, A. Grüneis, S.G. Chou, Ge.G. Samsonidze, A. Jorio, G. Dresselhaus,
M.S. Dresselhaus, Phys. Rev. B 71 (2005), 045417/25.
[35] M.D. Ellison, P.J. Gasda, J. Phys. Chem. C 112 (2008) 738–740.
[36] M. Barbeni, E. Pramauro, E. Pelizzeti, Chemosphere 14 (1985) 195–208.
[37] Z. Ezˇerskis, Z. Jusys, Pure Appl. Chem. 73 (2001) 1929–1940.
[38] R. Li, P.E. Savage, D. Szmukler, AIChE J. 39 (1993) 178–187.
[39] Z. Ezˇerskis, Z. Jusys, J. Appl. Electrochem. 31 (2001) 1117–1124.
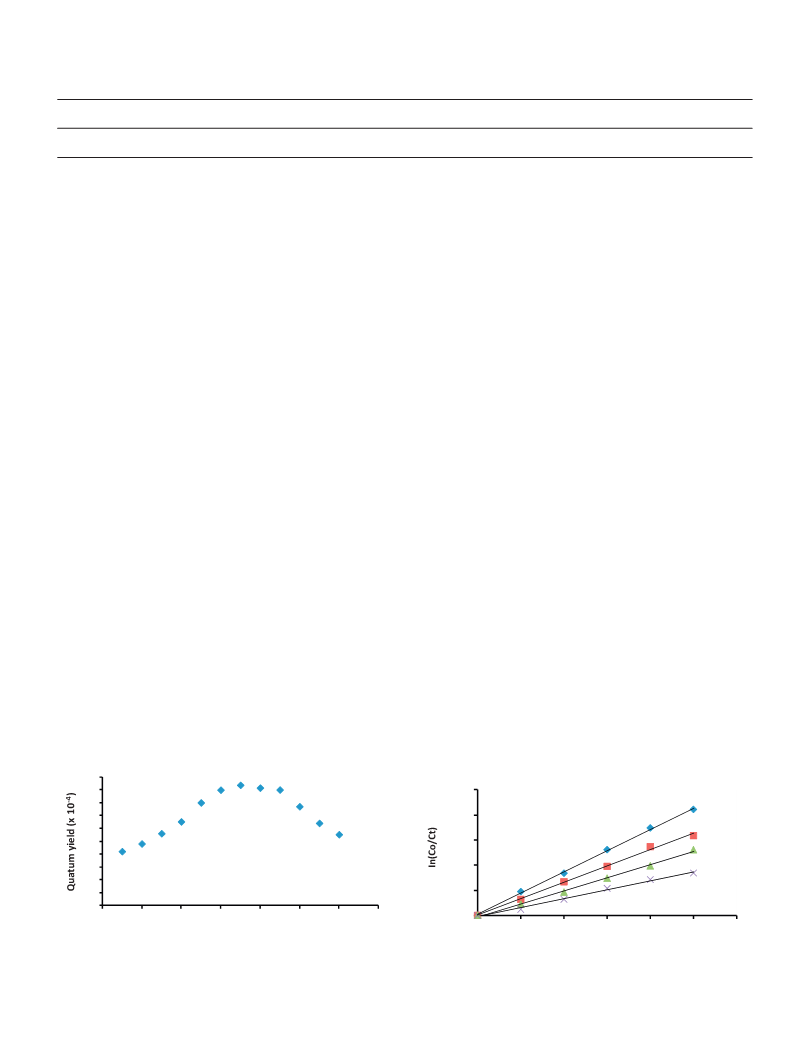
a higher proportion of hydroquinone (70% of products) though rate
of reaction (4.00 × 10−7 M min−1) was slower than when oxygen
was bubbled (8.00 × 10−7 M min−1) (Fig. 10). Photolysis was also
reaction rate was similar to when argon was bubbled with a rate
of 3.00 × 10−7 M min−1. Both homogeneous and heterogeneous
photosensitization for pentachlorophenol gave 2,3,5,6-tetrachloro-
1,4-benzoquinone (Scheme 2) with homogeneous reaction giving
70% conversion as compared with 30% for heterogeneous reaction.
Introduction of singlet oxygen scavenger halted the reaction sug-
gesting that the only pathway available for this transformation is
the Type II pathway.

 Ogunbayo, Taofeek B.
Ogunbayo, Taofeek B.
 Nyokong, Tebello
Nyokong, Tebello