Angewandte
Communications
Chemie
also cause peptide cleavage as demonstrated in hypervelocity
À1 [34]
impacts (2.4–5.8 kms ), which might explain the relatively
short peptides formed under harsh mechanical conditions.
In synthetic chemistry, one efficient way to carry out
productive chemical transformations enabled by mechanical
activation involves implementing ball milling techniques,
[35–40]
often in the absence of a liquid media.
Unlike hyper-
velocity tests, which involve impacts at speeds in the range of
kilometers per second, collisions in standard mixer ball mills
[
41]
occur in the range of few meters per second, making the
[
41]
mechanical load transferred by ball milling,
and the
[
42]
reached temperatures more suitable for chemical synthesis,
[
41b]
hence rarely observing sample decomposition.
In the prebiotic context, ball milling has been applied for
synthesizing relevant building blocks such as a-aminoni-
[43,44]
[45,46]
triles
and monosaccharides
under plausible early
Earth conditions. Furthermore, chemical syntheses of nucle-
[
47]
[48]
[49]
otides,
DNA fragments,
and lipid derivatives
have
been developed in ball mills, and solid-state approaches have
led to DNA-specific recognition and self-assembly of model
[
50]
nucleobases. Additionally, the use of other setups such as
flow chemistry photoreactors has proven helpful to recreate
[51]
prebiotic environments for chemical transformations.
These previous studies highlighted the potential of i) mechan-
[52]
ical energy to drive prebiotically relevant transformations,
and ii) the usefulness of enabling technologies to mimic and
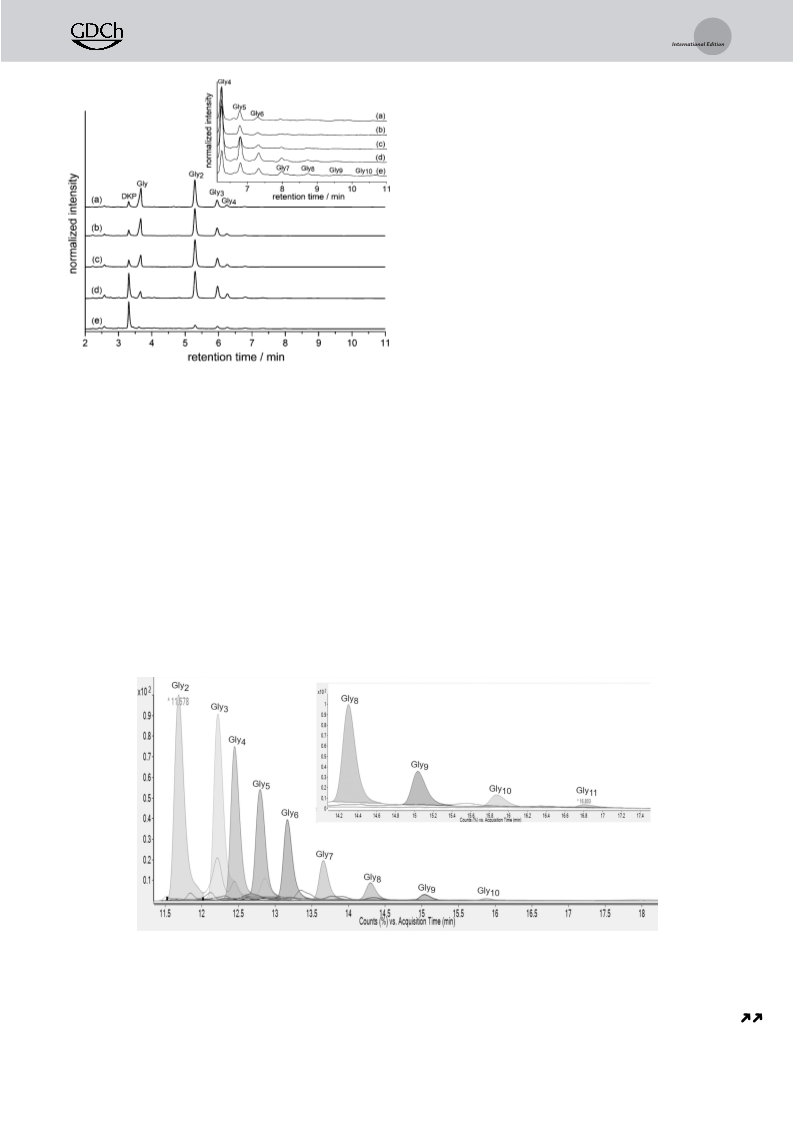
Figure 1. IP-HPLC chromatograms using a UV/Vis detector set to
record the absorbance at 195 nm, for: a) pristine Gly milled at RT and
b) Gly and TiO mixture milled at RT; c) Gly standard; d) Gly
2 2
[
20,51,53]
study prebiotic conditions.
Consequently, we wondered
standard; e) Gly standard; f) DKP standard. The inset shows the
3
if the controllable and moderate mechanical activation by
grinding, mixing, impact, and shear achieved by ball milling
zoomed region and reveals the presence of Gly and Gly in (a) and
2
3
(
b). Note: the solubility of DKP, Gly, and Gly standards in the mobile
n
(
Scheme 1c) could enable prebiotic peptide bond formation
phase was verified, longer Gly oligomers have also been analyzed
under similar HPLC conditions.
[
54]
[20,58]
from inactivated amino acids (Scheme 1d).
Here, we
present the first results of mechanical oligomerization of
pristine glycine (Gly) as a model amino acid. The choice to
use Gly is based on its presence as a product in simulated
[
61]
the known high adsorption of Gly onto the mineral, but we
also tested other minerals in the course of the study.
[
9,55]
prebiotic experiments,
its abundance in studied meteor-
[
56]
[57]
ites, its expected prevalence in the earliest proteins, and
the availability of analytical methods to determine the
products and extent of Gly oligomerization.
In general, milling at moderately elevated temperatures
increases the total yield of Gly converted to linear oligomers
(Gly ) (Table 1; for quantification details, see the Supporting
ꢀ
2
As an initial experiment, we milled pristine Gly at room
temperature (RT) and subsequently analyzed the reaction
mixture applying the modified IP-HPLC (ion-pair high-
performance liquid chromatography) method developed by
Information), and leads to the formation of higher oligomers
(Figure 2 and Table 1). The maximum total yield for milling at
1008C is 10.2% (Figure 2d). The selectivity of the oligome-
rization process changes at higher temperatures; despite the
the Bracher group, which has proven useful to resolve Gly
longest oligomer (Gly ) observed after milling at 1308C,
DKP was the dominant component of the product mixture
(Figure 2e).
n
10
[
58]
mixtures that are typically difficult to dissolve.
The
chromatographic analysis of the white powder collected
after the mechanochemical treatment revealed DKP forma-
tion and traces of Gly and Gly (Figure 1a).
2
3
Table 1: Effect of the milling temperature on the mechanochemical
To study the effect of the temperature on the peptide bond
formation, we applied a recently developed temperature-
controlled ball milling protocol. For example, in a standard
experiment, Gly (60 mg, 0.80 mmol) was milled with a five-
oligomerization of Gly with TiO2.
[
a]
[b]
Milling temperature [8C]
Longest detected oligomer
Yield [%]
[
59]
RT
Gly6
6.7
6.7
10.1
10.2
8.5
4
7
1
1
0
0
00
30
Gly6
Gly7
Gly8
Gly10
fold molar excess of TiO (anatase) at 30 Hz for 18 h where
2
[c]
the temperature was varied from RT to 1308C, thus respect-
ing practical thermal limits for prebiotic peptide bond
[d]
[
20,21]
formation,
while acknowledging the expected dilution
[
a] Based on IP-HPLC analysis using a UV/Vis detector set to record the
absorbance at 195 nm. [b] The combined yield of all linear oligomers of
glycine Gly . [c] The presence of Gly was detected by the UPLC-MS
of amino acids on mineral surfaces under prebiotic conditions
[
30,56]
(
Scheme 1d).
Among the oxide minerals considered for
ꢀ2
8
[
60]
prebiotic peptide bond formation, TiO was chosen due to
2
method (Supporting Information, Figure S12). [d] The presence of Gly11
was detected by UPLC-MS (Figure 3).
2
ꢀ 2021 Wiley-VCH GmbH
Angew. Chem. Int. Ed. 2021, 60, 1 – 6
These are not the final page numbers!

 Cindro, Nikola
Cindro, Nikola
 Grube?i?, Sa?a
Grube?i?, Sa?a
 Hernández, José G.
Hernández, José G.
 Me?trovi?, Ernest
Me?trovi?, Ernest
 Stolar, Tomislav
Stolar, Tomislav
 U?arevi?, Krunoslav
U?arevi?, Krunoslav