2
Y. Sasanuma et al. / Polymer xxx (2014) 1e13
silk fibroins, can also effectively degrade PLA. This may be partly
because of the similarity in chemical structure between alanine and
lactic acid. (5) The melting point of the polyesters is closely related
to the enzymatic degradability.
This study has also aimed to interpret the above microbiological
findings in terms of polymer physicochemistry.
removed by filtration. The filtrate (2 mL) underwent extraction
with ethyl acetate (100 mL) and water (100 mL), and this extraction
was repeated four times. The organic layer was condensed and
subjected to the column chromatography (Rf of the product ~ 0.6,
yield 26%).
2.1.6. Butylene glycol diacetate-13C (BGDA-13C)
2. Methods
Acetyl chloride (5.0 mL, 70 mmol), 1,4-butanediol (7.6 g,
84 mmol), and pyridine (6.7 g, 85 mmol) were treated in the same
manner as described in Section 2.1.5 to yield butylene glycol
monoacetate (Rf of the product ~ 0.3, yield 25%).
2.1. Sample preparation
Commercial available chemicals were used as received: ethylene
glycol, 1,4-butanediol, acetyl chloride, diethyl ether, 1,4-dioxane,
ethyl acetate, n-hexane, silica gel (Wako Pure Chemical Industries,
Acetyl chloride-13C (0.25 g, 3.1 mmol) was added to butylene
glycol monoacetate (0.51 g, 3.9 mmol) and pyridine (0.30 g,
3.8 mmol), and the mixture was stirred at 0 ꢀC for 3 h and purified
as described in Section 2.1.2 to yield BGDA-13C (64%).
Tokyo, Japan); succinyl chloride, acetyl chloride-13
C (Sigma-
eAldrich Japan, Tokyo, Japan).
All syntheses here employed the following experimental
setup: a four-necked flask (under a stream of dry nitrogen)
equipped with a mechanical or a magnetic stirrer and a Dimroth
condenser connected to a calcium chloride drying tube. In silica
gel column chromatography, a mixed eluting solvent (ethyl ace-
tate:n-hexane ¼ 1:1) was used. All products were identified by
NMR.
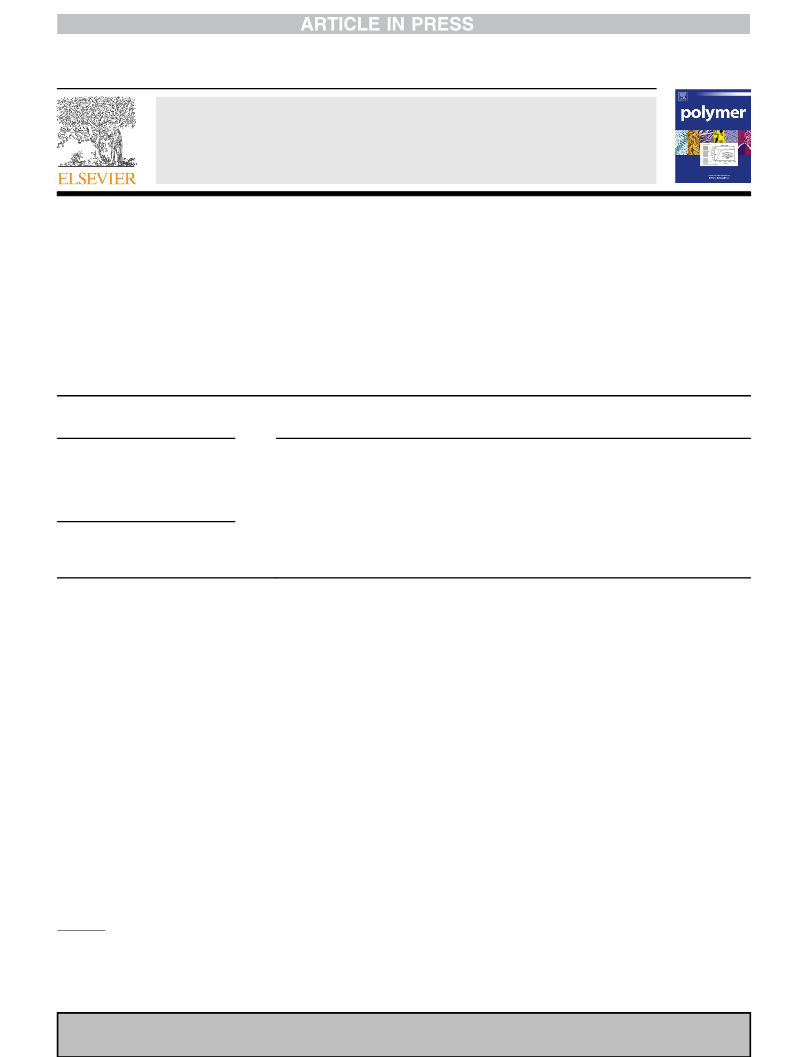
2.2. NMR measurements
1H (13C) NMR spectra were recorded at 500 MHz (125.7 MHz) on
a JEOL JNM-ECA500 spectrometer equipped with a variable tem-
perature controller in the Center for Analytical Instrumentation of
Chiba University. The measurement temperatures were 15, 25, 35,
45, and 55 ꢀC and maintained within 0.1 ꢀC fluctuations. Free
induction decays (FIDs) were accumulated 32 (256) times. The
pulse width, data acquisition time, and recycle delay were 5.6
(5.0)
s, 3.3 (2.0) s, and 3.7 (2.0) s, respectively. In the 13C NMR
experiments, the gated decoupling technique was employed under
the conditions given in the above parentheses. The solvents were
cyclohexane-d12 (C2D12), benzene-d6 (C6D6), chloroform-d (CDCl3),
methanol-d4 (CD3OD), and dimethyl-d6 sulfoxide ((CD3)2SO), and
the solute concentration was approximately 5 vol%. The NMR
spectra were simulated with the gNMR program [17] to yield
chemical shifts and coupling constants.
p/2
2.1.1. Dimethyl succinate (DMS)
Methanol (2.7 mL, 67 mmol) was added to succinyl chloride
(3.0 mL, 27 mmol) and stirred for 1 h. After methanol (1.0 mL,
25 mmol) was superadded, the mixture was gradually heated up
to 70 ꢀC, kept there for 1 h to remove generated hydrogen chlo-
ride, and then cooled down to ambient temperature. After pyri-
dine (1.1 mL, 14 mmol) was added, the reaction mixture was
stirred, filtrated, and condensed on a rotary evaporator to yield
DMS.
m
2.1.2. Ethylene glycol diacetate (EGDA)
2.3. MO calculations on model compounds
Acetyl chloride (4.5 mL, 63 mmol) was added to ethylene glycol
(1.97 g, 32 mmol) and pyridine (5.03 g, 64 mmol) stirred in the four-
necked flask, and then the mixture was stirred for 3 h with the flask
bathed in ice water. Diethyl ether was added into the flask, and the
mixture was filtrated to remove white precipitate. The filtrate was
subjected to the silica gel column chromatography (Rf of the
product ~ 0.6) and condensed to yield EGDA (yield 32%).
Density functional and ab initio MO calculations were carried
out with the Gaussian09 program [18] installed on an HPC Sys-
tems 5000-Z800 computer. For each conformer of model com-
pounds, the geometrical parameters were fully optimized at the
B3LYP/6-311þG(2d,p) level, and the thermal-correction term to
the Gibbs free energy (at 25 ꢀC) was calculated at the same level.
Bond lengths, bond angles, and dihedral angles used in the
refined RIS computations were chosen from the optimized
geometrical parameters (see Tables S4 and S5, Supplementary
data). All the self-consistent field calculations were conducted
under the tight convergence. With the optimized geometry, the
electronic energy was computed at the MP2/6-311þG(2d,p) and
M062X/6-311þG(2d,p) level [19]. The Gibbs free energy was
evaluated from the electronic energy and the thermal-correction
term, being given here as the difference from that of the all-trans
2.1.3. Ethylene glycol diacetate-13C (EGDA-13C)
Acetyl chloride (30 mL, 0.42 mol) was added to ethylene glycol
(31.4 g, 0.51 mol) dissolved in 1,4-dioxane (52 mL, 0.61 mol) stirred
in the four-necked flask. The mixture was refluxed for 2 h with the
flask heated at 120 ꢀC in an oil bath [15]. After cooled down to
ambient temperature, the reaction mixture was condensed and
purified by the column chromatography (Rf of the product ~ 0.3) to
yield ethylene glycol monoacetate (yield 42%).
Acetyl chloride-13C (0.25 g, 3.1 mmol) was added to a mixture of
ethylene glycol monoacetate (0.43 g, 4.1 mmol) and pyridine
(0.32 g, 4.1 mmol) and stirred for 3 h. The handling described in
Section 2.1.2 was employed to yield EGDA-13C (56%).
conformer and denoted as D
G. Vicinal 1He1H and 13Ce1H
coupling constants used to determine bond conformations of
model compounds were calculated at the B3LYP/6-
311þþG(3df,3pd) level [20]. The
DG values of the model com-
pounds in benzene and chloroform at 25 ꢀC were also calculated
at the MP2/6-311þG(2d,p) and M062X/6-311þG(2d,p) level with
the integral equation formalism of the polarizable continuum
model [21].
2.1.4. Ethylene glycol di(methyl succinate) (EGDMS)
This compound was prepared according to van der Brand et al.
[16].
Herein the dihedral angle is defined according to the tradition in
2.1.5. Butylene glycol diacetate (BGDA)
polymer science [7]: traꢁns (t) ~ 0ꢀ, cis (c) ~ 180ꢀ, gaucheþ (gþ)
Acetyl chloride (5.0 mL, 70 mmol) was added to 1,4-butanediol
(3.8 g, 42 mol) and pyridine (6.7 g, 85 mmol) in the flask kept at
0 ꢀC, and then the mixture was stirred at 0 ꢀC for 3 h. Diethyl ether
was added into the flask, and yellow solid was precipitated and
~þ120ꢀ, and gaucheꢁ (g ) ~ꢁ120ꢀ. The dihedral angle (
f) can be
converted to that (F) recommended by IUPAC [22] according to
F
¼ ꢁsign(
f
)(180 ꢁ j
f
j), where the function, sign(
f), returns the
¼ ꢁsign( )(180ꢁj j). Non-SI units are
sign of
f
, and vice versa:
f
F
F
Please cite this article in press as: Sasanuma Y, et al., Conformational characteristics and configurational properties of poly(ethylene succinate)
and poly(butylene succinate) and structureepropertyefunction relationships of representative biodegradable polyesters, Polymer (2014),

 Sasanuma, Yuji
Sasanuma, Yuji
 Nonaka, Yuta
Nonaka, Yuta
 Yamaguchi, Yuki
Yamaguchi, Yuki