1
036
Chemistry Letters Vol.37, No.10 (2008)
An Environmentally Friendly Electrolytic System Based on the Acid–Base Reaction
between Water and Solid-supported Bases
ꢀ1
2
2
Toshiki Tajima, Satomi Ishino, and Hitoshi Kurihara
Global Edge Institute, Tokyo Institute of Technology, Nagatsuta, Midori-ku, Yokohama 226-8502
Department of Electronic Chemistry, Tokyo Institute of Technology, Nagatsuta, Midori-ku, Yokohama 226-8502
1
2
(Received July 8, 2008; CL-080673; E-mail: tajima.t.ac@m.titech.ac.jp)
We have developed an environmentally friendly electrolytic
-4
(a)
b)
(c)
1
1
5
.5x10
(
system based on the acid–base reaction between water and solid-
supported bases. Furthermore, we have successfully applied the
electrolytic system to a flow cell.
-
4
.0x10
.0x10
-5
0
.0
Since its beginning in the early 1990s, green chemistry has
grown into a significant internationally engaged area within
chemistry. Over the past two decades, the design of environ-
-5
-
5.0x10
1
-4
-1.0x10
1.5x10
mentally benign solvents has been one of the leading research
areas of green chemistry. Water, ionic liquids, fluorous solvents,
and supercritical fluids have emerged as alternative reaction me-
dia for organic synthesis. In particular, organic synthesis in water
without the use of any harmful organic solvents has long been a
-4
-
-
4
-2.0x10
-
3
-2
-1
0
1
2
3
Potential / V vs. SCE
2
major object in green chemistry, because water is a safe, cheap,
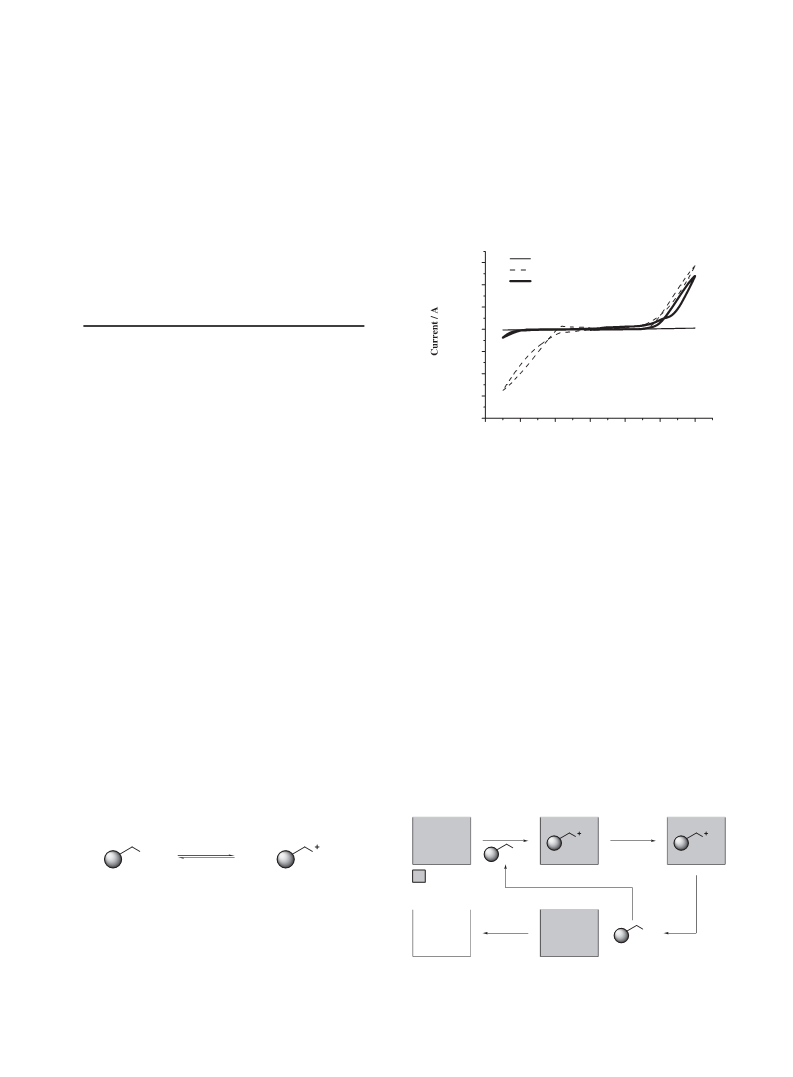
Figure 1. Cyclic voltammograms of (a) water using a Pt disk
electrode (ꢀ ¼ 1 mm), (b) 0.1 M Si–NH2/water using a Pt disk
easily available, and environmentally benign solvent. Therefore,
it is of much importance to develop radical new methods for
using water as a reaction medium in organic synthesis.
electrode (ꢀ ¼ 1 mm), and (c) 0.1 M Si–NH2/water using a
GC disk electrode (ꢀ ¼ 1 mm). The scan rate was 100 mV sꢁ
1
.
Organic electrosynthesis has recently attracted much atten-
tion as one of the most promising green processes in organic
synthesis, because it is possible to oxidize and reduce organic
compounds without the use of any conventional redox reagents.
However, it generally requires large amounts of supporting elec-
trolytes and organic solvents to provide sufficient ionic conduc-
tivity and solubilizing ability to the electrolytic media. In order
to remove the need for supporting electrolytes, we have recently
ꢁ
þ
current for OH and the reduction current for H were clearly
observed in the presence of Si–NH2 as shown in Figure 1b. In
addition, the solution had a pH of 9.7. These results indicate that
the acid–base reaction between water and Si–NH2 occurs (eq 1;
1
2
R, R , and R ¼ H), and the conjugate acid–base pair seems to
play the role of supporting electrolytes. On the other hand, the
cathodic potential window was extended to the negative direc-
tion up to ca. ꢁ2:0 V vs. SCE by using a glassy carbon (GC) disk
electrode as shown in Figure 1c. This finding suggests that the
electrolytic system based on the acid–base reaction between
water and Si–NH2 is available for electroreduction of organic
compounds.
We then investigated electroreduction of dimethyl maleate
1) as a model reaction using solid-supported bases in water.
The typical experimental procedure is illustrated in Figure 2.
As shown in Table 1, electroreduction of 1 proceeded smoothly
to provide the corresponding hydrogenation product 2 in high to
quantitative yields with the use of relatively weak bases (Entries
developed a novel electrolytic system using solid-supported
bases.3,4 The system is based on the acid–base reactions between
3
4
protic solvents or carboxylic acid substrates and solid-support-
ed bases, and the conjugate acid–base pairs act as supporting
electrolytes (eq 1). With the use of solid-supported bases,
it is possible to carry out organic electrosynthesis without any
additional supporting electrolytes. On the other hand, solid-sup-
ported bases may also be applied to the electrolytic system based
on the acid–base reaction between water and solid-supported
bases. It would enable us to achieve organic electrosynthesis
without the use of any additional supporting electrolytes and
organic solvents. Thus, we herein report an environmentally
friendly electrolytic system based on the acid–base reaction
between water and solid-supported bases.
5
(
6
1
–3). While silica gel-supported pyridine, amine (Si–NH2), and
substrate 1
product 2
NH3
+ OH-
substrate 1
electrolysis
NH3
Si
+ OH-
H2O
Si
NR R2
1
RO-
NHR R
1
2
NH2
Si
ROH +
+
ð1Þ
R = Me, Ac, R'CO
: water
reuse
First of all, in order to confirm the acid–base reaction be-
tween water and solid-supported bases, we measured the cyclic
voltammograms of water in the absence and presence of silica
gel-supported amine (Si–NH2) using a platinum disk electrode.
As shown in Figure 1a, ionic conductivity was hardly observed
in the absence of Si–NH2. In sharp contrast, both the oxidation
product 2
separation
filtration
product 2
+
Si
NH2
H2O
Figure 2. Experimental procedure.
Copyright Ó 2008 The Chemical Society of Japan

 Tajima, Toshiki
Tajima, Toshiki
 Ishino, Satomi
Ishino, Satomi
 Kurihara, Hitoshi
Kurihara, Hitoshi