1322
L. Rosi et al. / Journal of Organometallic Chemistry 695 (2010) 1314–1322
were prepared in a Schlenk tube under nitrogen atmosphere, and
transferred into the autoclave by suction. The autoclave was pres-
surized at room temperature with hydrogen, then stirred and
heated at the prefixed temperature for the established time. At
the end of the reaction the autoclave was rapidly cooled to room
temperature, the gas vented and the solution collected and trans-
ferred into a 50 ml Schlenk tube. A sample of the solution was ta-
ken for the MS and GC analyses. Each test was repeated three
times, the mean value was reported.
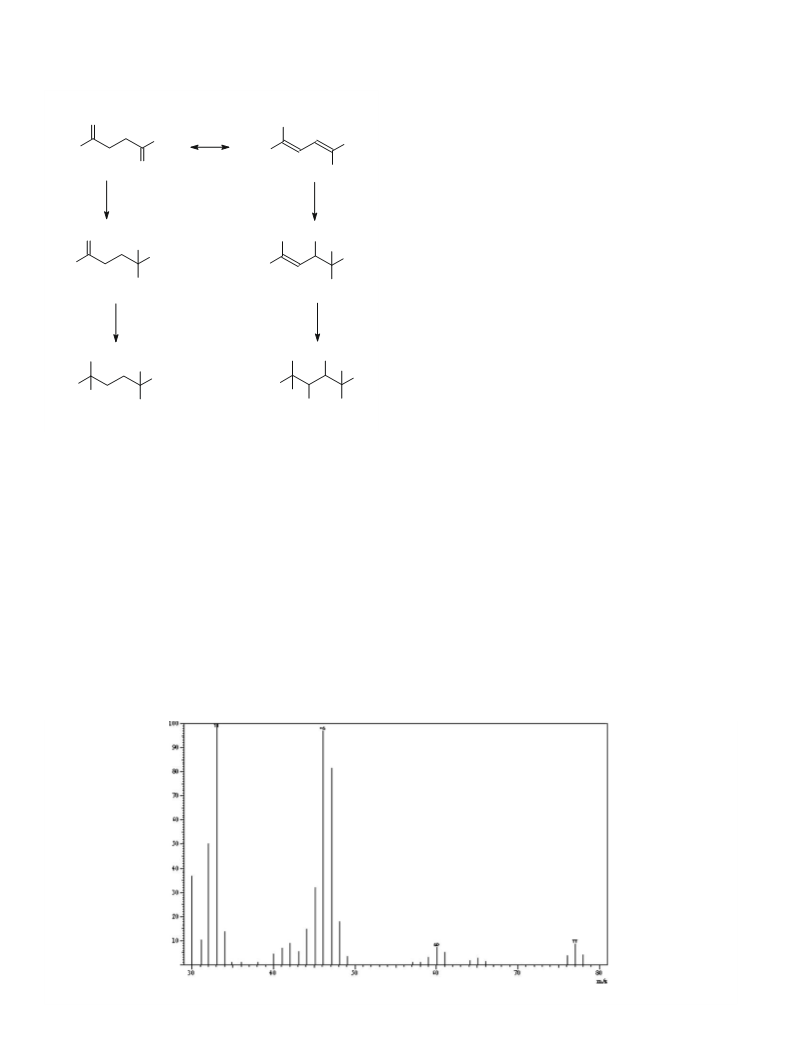
MS: The MS spectrum of 1,4-BD-d8 is reported in Fig. 1, show-
ing: [CD2CDCHD]+Å as base peak at 46 m/z. Interpretation of the
other peaks present is reported in the text.
Acknowledgements
The authors thank the University of Florence, the Ministero del-
la Industria, Università e Ricerca (MIUR), Programmi di Ricerca Sci-
entifica di Notevole Interesse Nazionale, Cofinanziamento MIUR
2008–09, the Regione Toscana – POR FESR 2007–2013 – Project Te-
Con@BC for financial support, and the Ente Cassa di Risparmio –
Firenze for the gift to acquire the Varian Mercury 400 NMR
instrument.
4.3.2. Preparation of 1,4-BD from fumaric or succinic acid
About 0.303 mg of Zn (4.6 ꢁ 10ꢂ3 mmol) was placed in the
autoclave, then the vessel was closed, and air was evacuated.
206.0 mg (1.77 mmol) of fumaric acid or 209.0 mg (1.77 mmol)
of succinic acid, 8.338 mg (2.09 ꢁ 10ꢂ2 mmol) of Ru(acac)3,
17.95 mg (0.029 mmol) of triphos were dissolved at room temper-
ature in 12 ml of solvent (CH3OH or THF) in a Schlenk tube. The
mixture was introduced in the autoclave by suction, then the ves-
sel was pressurized with hydrogen at 80 bar. The mixture was stir-
red at the settled temperature for the prefixed time. At the end of
the reaction the autoclave was rapidly cooled, the gas vented out
and the crude analyzed by GC and GC–MS techniques.
References
[1] (a) Y. Pouilloux, F. Autin, J. Atmrault, Catal. Today 63 (2000) 87. and reference
therein cited;
(b) N.W. Cant, D.L. Trimm, T. Turek, Catal. Rev. Sci. Eng. 36 (1994)
645.
[2] (a) U. Matteoli, M. Bianchi, G. Menchi, P. Frediani, F. Piacenti, J. Mol. Catal. 22
(1984) 353;
(b) U. Matteoli, M. Bianchi, G. Menchi, P. Frediani, F. Piacenti, J. Mol. Catal. 29
(1985) 269;
(c) U. Matteoli, G. Menchi, M. Bianchi, F. Piacenti, J. Organomet. Chem. 299
(1986) 233;
(d) U. Matteoli, G. Menchi, M. Bianchi, F. Piacenti, J. Mol. Catal. 44 (1988) 347;
(e) U. Matteoli, G. Menchi, M. Bianchi, F. Piacenti, J. Mol. Catal. 64 (1991) 257;
(f) U. Matteoli, G. Menchi, M. Bianchi, F. Piacenti, S. Ianelli, M. Nardelli, J.
Organomet. Chem. 498 (1995) 177.
4.3.3. Preparation of 1,4-BD from GBL
About 0.303 mg of Zn (4.6 ꢁ 10ꢂ3 mmol) was placed in the
autoclave, then the autoclave was closed, and air was evacuated
from the vessel. And 140 ll (1.77 mmol) of GBL, 8.575 mg
[3] R.A. Grey, G.P. Pez, A. Wallo, J. Am. Chem. Soc. 103 (1981) 7536.
[4] (a) H. Teunissen, C.J. Elsevier, Chem. Commun. (1997) 667;
(b) M.C. van Engelen, H.T. Teunissen, J.G. de Vries, C.J. Elsevier, J. Mol. Catal. A:
Chem. 206 (2003) 185.
[5] H. Teunissen, C.J. Elsevier, Chem. Commun. (1998) 1367.
[6] K. Nomura, H. Ogura, Y. Imanishi, J. Mol. Catal. A: Chem. 178 (2002) 105.
[7] B. Boardman, M.J. Hanton, H. van Rensburg, R.P. Tooze, Chem. Commun. (2006)
2289.
(2.15 ꢁ 10ꢂ2 mmol) of Ru(acac)3, 18.16 mg (0.029 mmol) of triphos
were dissolved at room temperature in 12 ml of solvent (CH3OH or
THF). The mixture was introduced in the autoclave by suction, then
the vessel was pressurized with hydrogen at 80 bar. The mixture
was stirred at the settled temperature for the prefixed time. After-
wards the autoclave was rapidly cooled, the gas vented out and the
crude analyzed by GC and GC–MS techniques.
[8] J. Zhang, G. Leitus, Y. Ben-David, D. Milstein, Angew. Chem., Int. Ed. 45 (2006)
1113.
[9] P. Frediani, L. Rosi, M. Frediani, G. Bartolucci, M. Bambagiotti-Alberti, J. Agric.
Food Chem. 55 (2007) 3877.
[10] C. Delhomme, D. Weuster-Botz, F.E. Kühn, Green Chem. 11 (2009) 13.
[11] C. Yin, Z. Xu, S.-Y. Yang, S.M. Ng, K.Y. Wong, Z. Lin, C.P. Lau, Organometallics 20
(2001) 1222.
[12] G. Horváth, J. Kuszmann, Org. Mass Spectrom. 12 (1977) 45.
[13] C.C. Van de Sande, F.W. McLafferty, J. Am. Chem. Soc. 97 (1975) 4613.
[14] M.C. van Engelen, Ph.D. Thesis, University von Amsterdam, 2003, p. 99, <http://
[15] V.I. Backhmutov, E.V. Bakhmutov, N.V. Belkova, C. Bianchini, L.M. Epstein, D.
Masi, M. Peruzzini, E.S. Shubina, E.V. Vorontsov, F. Zanobini, Can. J. Chem. 79
(2001) 479.
4.3.4. Preparation of 1,4-BD-d8
The amount of reactants and the operating procedure were the
same for the hydrogenation of fumaric acid: 206.0 mg (1.77 mmol)
of fumaric acid, 8.338 mg (2.09 ꢁ 10ꢂ2 mmol) of Ru(acac)3,
17.95 mg (0.029 mmol) of MeC(CH2PPh2)3, 0.303 mg (4.6 ꢁ
10ꢂ3 mmol) of Zn and 12 ml of CD3OD. The reaction was carried
out for 72 h under a D2 pressure of 80 bar, and the resulting solu-
tion was analyzed by GC, GC–MS, 1H and 13C NMR techniques.
1H NMR (400 MHz, (CD3OD)), d: 1.541 ppm (singlet, 2H, –CHD–).
13C NMR (400 MHz, (CD3OD)), d: 60.416 ppm (quintet, –CD2–,
1J: 21.43 Hz), 28.670 ppm (triplet, –CHD–, 1J: 19.11 Hz).
[16] W.L.F. Armarego, D.D. Perrin, Purification of Laboratory Chemicals, IV ed.,
Pergamon Press, UK, 1996.
[17] G. Braca, G. Sbrana, P. Pino, Chim. Ind. (Milan) 50 (1968) 121.

 Rosi, Luca
Rosi, Luca
 Frediani, Marco
Frediani, Marco
 Frediani, Piero
Frediani, Piero