Synthesis of Valsartan via Decarboxylative Biaryl
Coupling
Lukas J. Goossen* and Bettina Melzer
Institut fu¨r Organische Chemie, TU Kaiserslautern,
Erwin-Schro¨dinger-Strasse, Building 54, D-67663
Kaiserslautern, Germany
FIGURE 1. Angiotensin-II-receptor antagonists.
ReceiVed June 27, 2007
Their common structural element, a biphenyl unit, is essential
for the binding affinity to the receptor and for the oral
bioavailability. The formation of its aryl-aryl bond represents
the key step in the synthesis of sartans: while for the synthesis
of losartan,2b the uses of Negishi6 and Ullmann7 couplings is
described in the literature, the published methods for the
preparation of valsartan make use of Suzuki-Miyaura cou-
plings.8,9 The principal synthetic pathways leading to valsartan
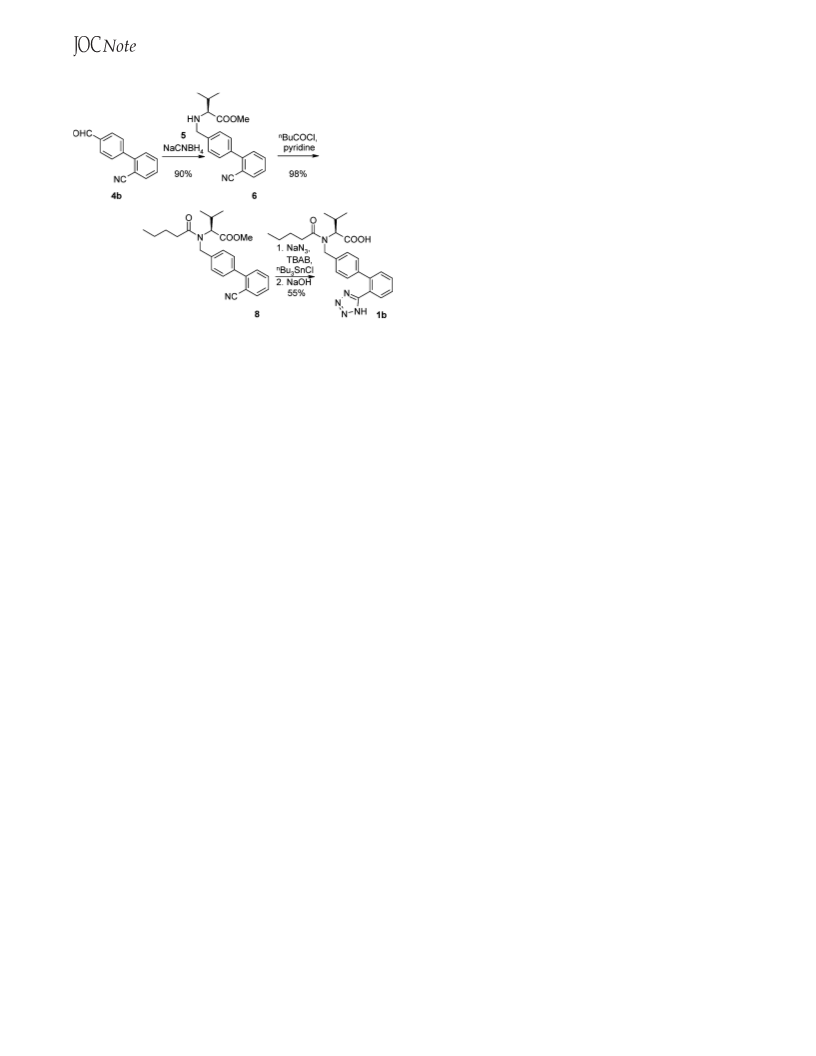
are depicted in Scheme 1. In route A, 2-chlorobenzonitrile (2a)
and 4-tolylboronic acid (3) are coupled to give 2-cyano-4′-
methylbiphenyl (4a), which is then brominated and reacted with
L-valine methyl ester to give N-[(2′-cyanobiphenyl-4-yl)methyl]-
L-valine methyl ester (6). Alternatively, 6 can be obtained via
the coupling of 4-bromobenzaldehyde (7b) with a boronic acid
derivative (e.g., 2b), followed by reductive amination with
L-valine methyl ester (route B). Route C results from a
combination of both approaches, in which the sensitive formyl
group in biaryl 4b is generated by oxidation of the more robust
derivative 4a.
The main shortcoming common to these syntheses originates
from the use of expensive boronic acid substrates in the cross-
coupling step. We believed that we could overcome this
weakness with the biaryl synthesis recently developed in our
group, which instead draws on carboxylic acid salts as a stable,
inexpensive, and widely available source of the aryl nucleo-
phile.10 In this method, these salts are decarboxylated by a
copper/phenanthroline system, and the resulting aryl-copper
species are coupled in situ with aryl halides by a palladium
cocatalyst. Other decarboxylative couplings of the Heck type
An efficient synthesis of the angiotensin II inhibitor valsartan
(Diovan) is presented. Two routes were evaluated, both
making use of an advanced version of our decarboxylative
coupling for the construction of the biaryl moiety. Thus, in
the presence of a catalyst system consisting of copper(II)
oxide, 1,10-phenanthroline, and palladium(II) bromide, 2-cy-
anocarboxylic acid was coupled with 1-bromo(4-dimethoxy-
methyl)benzene in 80% yield and with 4-bromotoluene in
71% yield. The valsartan synthesis using 1-bromo(4-di-
methoxymethyl)benzene was completed in four steps overall
with a total yield of 39%, via a novel route that presents
substantial economical and ecological advantages over the
literature process, as it is more concise and stoichiometric
amounts of expensive organometallic reagents are avoided.
Hypertension is one of the most prevalent diseases in
developed countries with an estimated 1 billion cases world-
wide,1 conferring its treatment an enormous social and economic
importance. The therapeutic standard was significantly improved
in the 1980s by the introduction of losartan (1a; Lorzaar, Merck)
(Figure 1)2 as the first nonpeptidic angiotensin-II-receptor
antagonist. An entire therapeutic class, the sartans, has since
been developed, among which valsartan (1b; Diovan, Novar-
tis: US $4.2 billion sales in 2006) (Figure 1) currently holds
the largest market share.3-5
(4) For published syntheses, see: (a) Bu¨hlmayer, P.; Ostermayer, F.;
Schmidlin, T. Eur. Pat. Appl. EP443983, 1991. (b) Bu¨hlmayer, P.;
Ostermayer, F.; Schmidlin, T. U.S. Patent 5,339,578, 1995. (c) Bu¨hlmayer,
P.; Furet, P.; Criscione, L.; de Gasparo, M.; Whitebread, S.; Schmidlin, T.;
Lattmann, R.; Wood, J. Bioorg. Med. Chem. Lett. 1994, 4, 29-34.
(5) For clinical trial results, see: (a) Novartis Pharma Schweiz, AG.
Health & Science 2005, 2, 8-9. (b) McMurry, J.; Solomon, S.; Pieper, K.;
Reed, S.; Rouleau, J.; Velazquez, E.; White, H.; Howlett, J.; Swedberg,
K.; Maggioni, A.; Køber, L.; Van de Werf, F.; Califf, R.; Pfeffer, M. J.
Am. Coll. Cardiol. 2006, 47, 726-733.
(6) (a) Negishi, E.; King, A. O.; Okukado, N. J. Org. Chem. 1977, 42,
1821-1823. (b) Nicolaou, K. C.; Bulger, P. G.; Sarlah, D. Angew. Chem.,
Int. Ed. 2005, 44, 4442-4489.
(1) U.S. Department of Health and Human Services; National Heart,
Lung, and Blood Institute; National High Blood Pressure Education
Program, NIH Publication No. 03-5233, December 2003.
(2) (a) Duncia, J.; Chiu, A.; Carini, D.; Georgory, G.; Johnson, A.; Price,
W.; Wells, G.; Wong, P.; Calabrese, J.; Timmermans, P. J. Med. Chem.
1990, 33, 1312-1329. (b) Carini, D.; Duncia, J.; Aldrich, P.; Chiu, A.;
Johnson, A.; Pierce, M.; Price, W.; Santella, J., III; Wells, G.; Wexler, R.;
Wong, P.; Yoo, S. E.; Timmermans, P. J. Med. Chem. 1991, 34, 2525-
2547.
(7) Fanta, P. E. Chem. ReV. 1964, 64, 613-632.
(8) Miyaura, N.; Suzuki, A. Chem. ReV. 1995, 95, 2457-2483.
(9) For state-of-the-art Suzuki couplings, see: (a) Littke, A. F.; Fu, G.
C. Angew. Chem., Int. Ed. 2002, 41, 4176-4211. (b) Kristensen, J.; Lyse´n,
M.; Vedsø, P.; Begtrup, M. Org. Lett. 2001, 3, 1435-1437.
(10) (a) Goossen, L. J.; Deng, G.; Levy, L. M. Science 2006, 313, 662-
664. (b) Goossen, L. J.; Rodr´ıguez, N.; Melzer, B.; Linder, C.; Deng, G.;
Levy, L. M. J. Am. Chem. Soc. 2007, 12, 4824-4833. (c) Goossen, L. J.;
Thiel, W. R.; Rodr´ıguez, N.; Linder, C.; Melzer, B. AdV. Synth. Catal., in
press.
(3) For sales figures, see: NoVartis Annual Report 2006; Novartis
International AG: Basel, Switzerland, 2007.
10.1021/jo701391q CCC: $37.00 © 2007 American Chemical Society
Published on Web 08/23/2007
J. Org. Chem. 2007, 72, 7473-7476
7473

 Goossen, Lukas J.
Goossen, Lukas J.
 Melzer, Bettina
Melzer, Bettina