CATALYTIC SYNTHESIS OF DIALKYL SULFIDES FROM DIALKYL DISULFIDES
407
were obtained in the conversion of diethyl disulfide
over γ-Al2O3 at 350°C and a residence time of τ = 0.5 s:
X, y, %
100
1
М
2
Yield, mol %
0.5
1
3
4
2
3
ΣMe2S + MeSEt
ΣMeSH + EtSH + H2S
13 30 54 64 66
66 50 26 15 12
60
20
Clearly, as М is increased, the total dialkyl sulfide
yield increases and the by-product yield decreases. At
М > 2, the process is somewhat stabilized. The above
data concerning the reactions in disulfide–methanol
mixtures (Tables 2–6) were obtained at М = 2.5–3.2
and a temperature of 350°C.
4
0.1
0.3
0.5
0.7
τ, s
The reactions of the disulfides with methanol
occur over all of the catalysts examined. The products
of the reactions are alkanethiols, dialkyl sulfides,
hydrogen sulfide, carbon oxides, and dimethyl ether.
Over Na/Al2O3, a catalyst with strong BS’s and weak
acid sites, the individual disulfides are converted at a
fairly high rate. All methanethiol disulfides turn into
alkanethiols, and the reactions of the disulfides con-
taining an ethyl or propyl group yield ethanethiol and
propanethiol as well. The dialkyl sulfide yield is
extremely low. The catalyst containing only weak PS’s
and medium-strength BS’s (SiO2) shows a low activity
in the reactions of the disulfides with methanol, and
the products consist mainly of alkanethiols. The reac-
tion over the catalyst having strong PS’s, weak L-sites,
and medium-strength BS’s (Cr/SiO2) occurs in a sim-
ilar way, but its rate is higher than in the case of silica.
The presence of strong acid sites on the catalyst sur-
face (as in HZSM-5 and γ-Al2O3) markedly increases
the reaction rate and favors the formation of dialkyl
sulfides in high yield. The reaction over the zeolite is
complicated by alkanethiol formation, and the reac-
tion over γ-Al2O3 practically yields only sulfides.
Dimethyl sulfide forms from all of the disulfides, and
the reactions of diethyl disulfide and methyl propyl
disulfide yield sulfides with R = Me, Et, and Pr as well
(see, e.g., Fig. 3). The dimethyl sulfide yield decreases
in the MeSSPr > Et2S2 > Me2S2 order. This is likely
due to the fact that the DMDS homologues undergo
cracking more readily to release hydrogen sulfide,
which then reacts with methanol, yielding dimethyl
sulfide. The reactions of methanol with the disulfides
contained in DSO occur in the same way as the reac-
tions of the individual disulfides, but the rate of con-
version of the petroleum disulfides mixed with meth-
anol is 1.5–2 lower than the rate of their conversion in
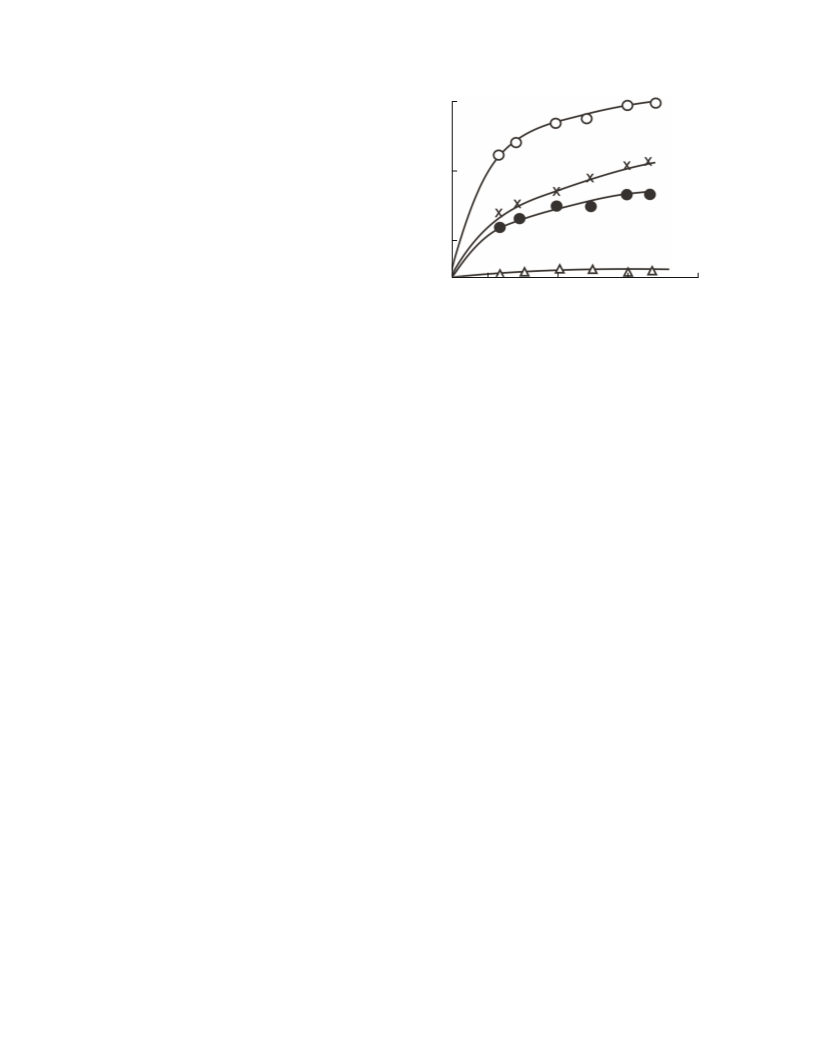
Fig. 3. Residence time effect on the (1) methyl propyl
disulfide conversion, (2) methyl propyl sulfide yield,
(3) dimethyl sulfide yield, and (4) hydrogen sulfide yield in
the reaction of the methyl propyl disulfide–methanol mix-
ture over the γ-Al O catalyst at 350°C.
2
3
Therefore, for the formation of dialkyl sulfides
from dialkyl disulfides and methanol, it is necessary
that the catalyst surface contain high concentrations of
strong L-sites and medium-strength BS’s and some
quantity of strong PS’s. High concentrations of the
latter enhance the formation of alkanethiols. The most
efficient catalyst for the synthesis of dialkyl sulfides
from methanol and dialkyl disulfides is alumina.
Methanol adsorption on the solid acid catalysts
leads to the release of surface-bound СН3 fragments
[5, 11, 12]; disulfide adsorption, to the formation of
RS fragments. The interaction of these fragments
yields reaction products. Dimethyl, methyl ethyl, and
methyl propyl sulfides result from the interaction of
MeS, EtS, and PrS groups with the СН3 fragment. In
addition, part of the dialkyl disulfides and resulting
thiols and sulfides decomposes to release hydrocar-
bons and hydrogen sulfide. The reaction between
hydrogen sulfide and methanol produces methan-
ethiol and dimethyl sulfide. The mechanism of the
catalytic reaction producing dialkyl sulfides from dial-
kyl disulfides will be the subject of our further studies.
Using diethyl disulfide as an example, we investi-
gated the interaction of the disulfides with methanol
over alumina samples differing in their phase compo-
sition, porosity, specific surface area, and granule size.
It was found that, for uncrushed catalyst granules, the
observed rate of the reaction is independent of the gas
hourly space velocity when the latter is not lower than
a helium atmosphere. In the reactions catalyzed by 0.8 h–1, but it increases markedly with an increasing
Na/Al2O3, SiO2, or Cr/SiO2, the main products are temperature. Therefore, there are no external diffu-
sion limitations in the process. The rate of the reaction
over pure γ-Al2O3 is 1.4–3 times higher than over
γ-Al2O3 containing χ- and η-Аl2O3.
alkanethiols and the dialkyl sulfide selectivity is low.
However, the selectivity is much higher in the reac-
tions catalyzed by zeolite HZSM-5 or γ-Al2O3.
KINETICS AND CATALYSIS Vol. 58 No. 4 2017

 Mashkina
Mashkina
 Khairulina
Khairulina