
Journal of Physical Chemistry p. 1674 - 1678 (1982)
Update date:2022-08-17
Topics:
 Kumar, Anil
Kumar, Anil
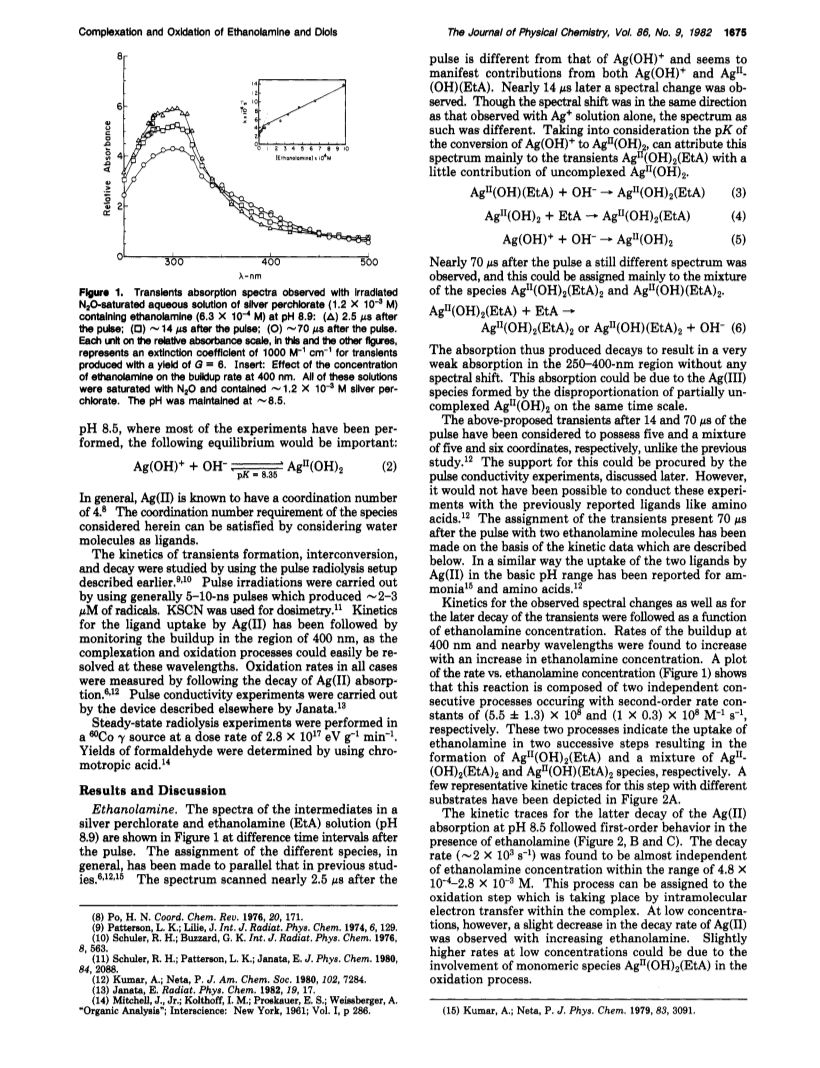
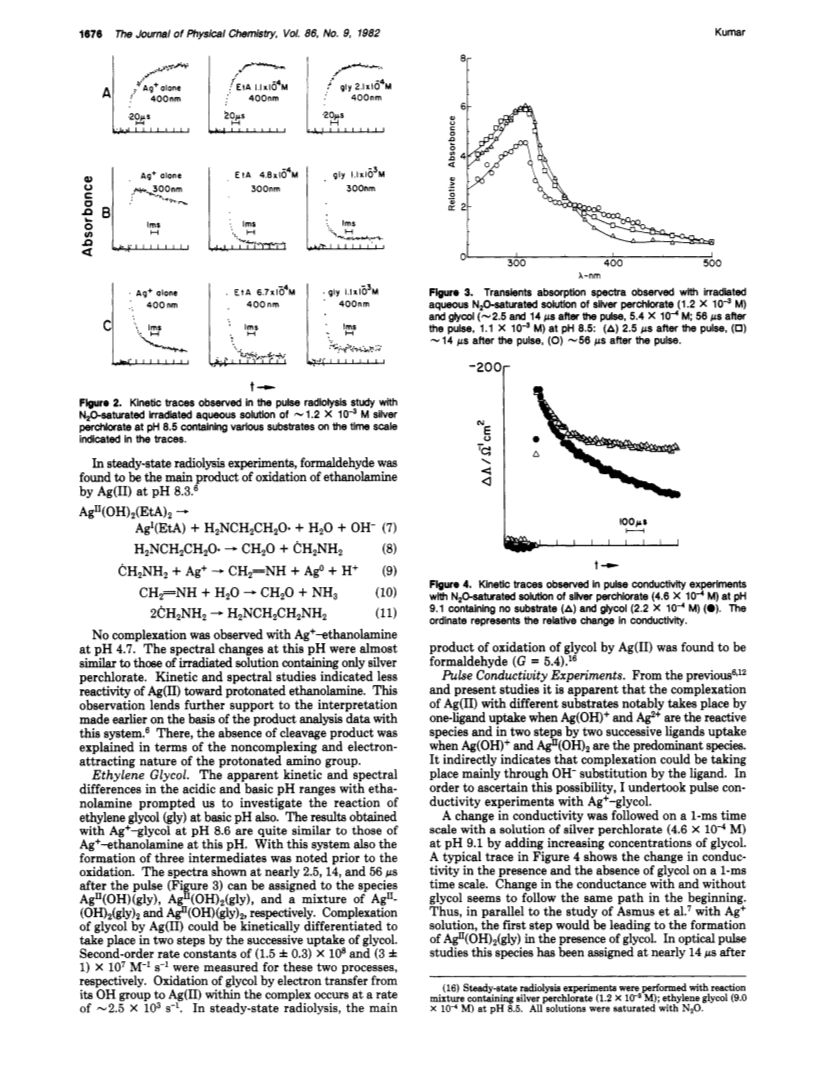
The oxidation of ethanolamine (EtA), ethylene glycol, and several other diols by Ag(II) has been studied at pH ca. 8.5.In the basic pH range, complexation of the substrate by Ag(II) has been found to take place in two steps by successive ligand uptake.Complexation rates are higher 1 order of magnitude in the basic pH range as compared to the acidic pH range.Oxidation then takes place through intramolecular electron transfer from substrate to Ag(II) within the complex.Oxidation rates for cis- and trans-1,2-cyclohexanediols are quite similar.
View More


Doi:10.3390/molecules23112905
(2018)Doi:10.1021/ol301849z
(2012)Doi:10.1016/j.bmcl.2014.11.042
(2015)Doi:10.5560/ZNB.2012-0249
(2012)Doi:10.1007/BF00497875
()Doi:10.1002/ejic.201402992
(2015)