
RSC Advances p. 45894 - 45906 (2016)
Update date:2022-08-11
Topics:
 Li, Fengjiao
Li, Fengjiao
 Wang, Liguo
Wang, Liguo
 Han, Xiao
Han, Xiao
 He, Peng
He, Peng
 Cao, Yan
Cao, Yan
 Li, Huiquan
Li, Huiquan
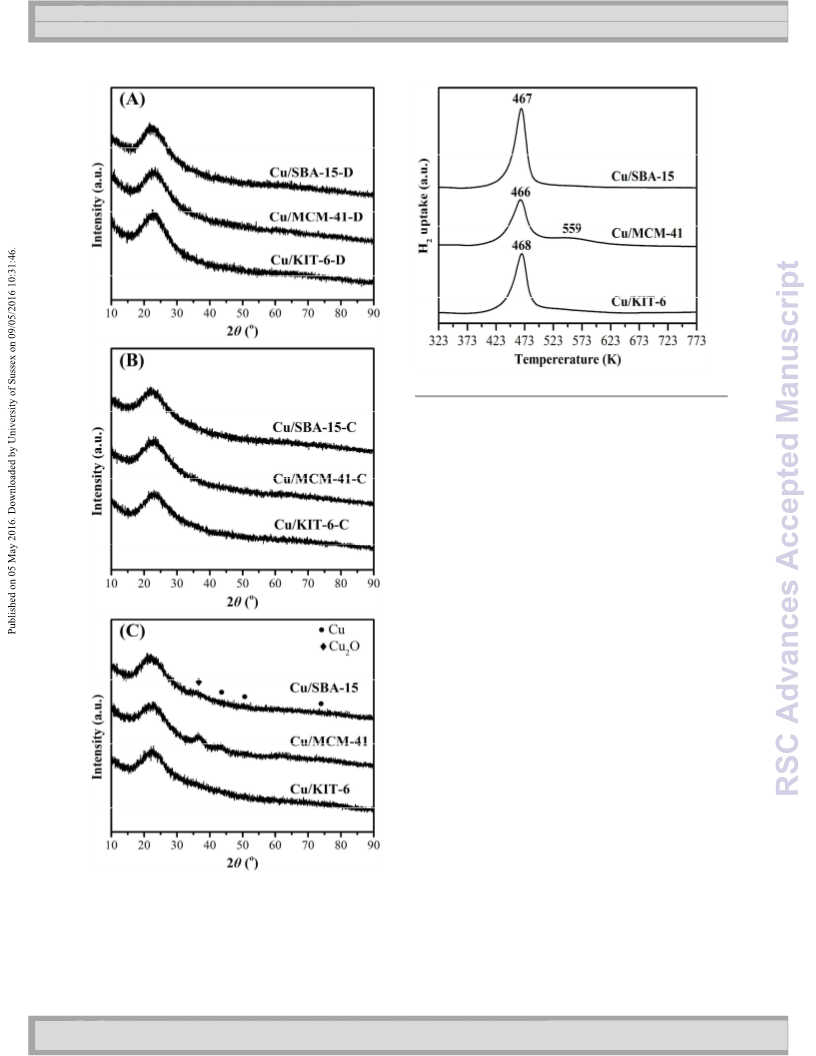
Hydrogenation of ethylene carbonate (EC) is an attractive and promising approach for indirect hydrogenation of CO2 to co-produce ethylene glycol (EG) and methanol (MeOH). In this work, thermodynamics of EC hydrogenation was firstly calculated, and the results disclosed that EC hydrogenation is exothermic (ΔrHθm = -71.59 kJ mol-1) and thermodynamically favorable (ΔrGθm = -25.62 kJ mol-1). The mesoporous silica supported copper catalysts were successfully prepared by facile ammonia evaporation method using KIT-6, MCM-41 and SBA-15 as support materials. The as-prepared Cu/KIT-6, Cu/MCM-41 and Cu/SBA-15 were thoroughly characterized by N2 physisorption, ICP-AES, N2O titration, FT-IR, XRD, H2-TPR, TEM, XPS and XAES. It was found that copper nanoparticles were well dispersed on these supports. Interestingly, a larger metallic Cu surface area with higher dispersion was obtained on SBA-15 when compared with KIT-6 and MCM-41. Furthermore, Cu0 and Cu+ species in various ratios were verified to co-exist on the surfaces of the three catalysts, which originated from CuO and copper phyllosilicate, respectively. The catalytic performances showed that Cu/SBA-15 exhibited a superior catalytic activity with a TON of 22.0 and 11.4 (mol mol-1) towards EG and MeOH, respectively. Importantly, 100% of EC conversion, 94.7% of EG yield and 62.3% of MeOH yield were achieved within a short reaction time of 4 h over Cu/SBA-15 under optimized conditions. The synergetic effect between Cu0 and Cu+ species, in which it was proposed that Cu0 species dissociated H2, while Cu+ species absorbed the carbonyl group of EC, was responsible for the higher catalytic activity of Cu/SBA-15.
View More












Taizhou Huading Chemical Co.,Ltd
Contact:+86-576-88583898
Address:Economic&Technology development zone,Taizhou City,Zhejiang Province,China
Contact:+31-24-3886056
Address:Binderskampweg 29 Unit 36
EASYBUYER Co.,LTD.( Chen-Chen )
Contact:+86-21-51816013/51816067
Address:1306,Bldg 8 Hi-Shanghai Center,No.950 Dalian Rd
Shanghai SMEC Trading Co., Ltd.
website:http://www.shanghaismec.com
Contact:+86-21-68811293
Address:ROOM 1101,NO.800 SHANGCHENG RD,SHANGHAI,CHINA
Contact:
Address:ROOM 1715, No#345 Jin Xiang Road, Pudong District
Doi:10.14233/ajchem.2017.20545
(2017)Doi:10.1021/jo01267a091
(1968)Doi:10.1016/0022-328X(93)83206-B
(1993)Doi:10.1016/S0957-4166(97)00330-3
(1997)Doi:10.1016/S0040-4020(01)82749-6
(1969)Doi:10.1080/15257779508014667
(1995)