After production of I~, however, HOI will carry most of the
oxidation through the rapid equilibrium reaction (M2). Reac-
tions (M9), (M10) and (M11) show the step-wise oxidation of
AETSA to the sulfonic acid and sulfate. For these simulations
it was assumed that the cleavage of the SwS bond occurs
together with a two-electron oxidation of the inner sulfur
atom. Reactions (M12) and (M13) show the oxidation of
AETSA by the molecular iodine species. The rate constant,
ported by a grant from the National Science Foundation
(Grant Number CHEÈ9632592).
References
1
2
3
C. R. Chinake and R. H. Simoyi, J. Phys. Chem., 1996, 100, 1643.
E. Mambo and R. H. Simoyi, J. Phys. Chem., 1993, 97, 13662.
C. R. Chinake, E. Mambo and R. H. Simoyi, J. Phys. Chem.,
1
994, 95, 2908.
J. B. Jones, C. R. Chinake and R. H. Simoyi, J. Phys. Chem.,
995, 99, 1523.
S. Hashimoto and J. Sunamoto, Bull. Chem. Soc. Jpn., 1966, 39,
207.
k
, for the oxidation of AETSA by I ~ was evaluated by this
11
3
4
5
6
study and was low enough to account for the autoinhibition.
All the oxyiodineÈAETSA reactions were taken as irreversible.
The 13 rate equations in Table 1 were numerically inte-
grated using a semi-implicit Runge Kutta method.35 There
was good agreement between the experimental data and the
computer simulations. The mechanism was tested for the
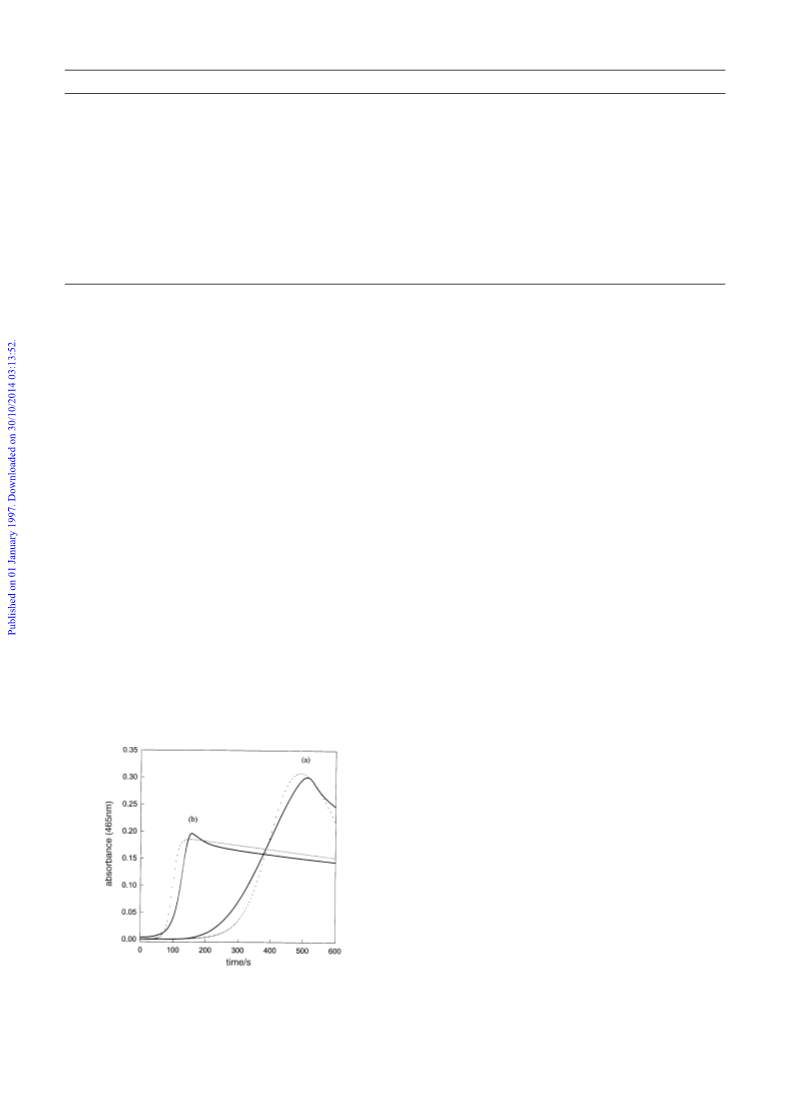
e†ects of acid, iodide and iodate. Fig. 9 shows the Ðt to the
data for the e†ect of acid. The slight deviations obtained were
as a result of the other species which have slight absorptivities
at 465 nm. Our simulations follow strictly the concentrations
1
1
Y. Luo, M. Orban, K. Kustin and I. R. Epstein, J. Am. Chem.
Soc., 1989, 111, 4541.
7
8
9
0
I. R. Wilson and G. M. Harris, J. Am. Chem. Soc., 1960, 82, 4515.
I. R. Wilson and G. M. Harris, J. Am. Chem. Soc., 1961, 84, 1101.
M. R. Ho†man, Environ. Eng. Sci., 1977, 11, 61.
1
R. J. Field, E. Koros and R. M. Noyes, J. Am. Chem. Soc., 1972,
94, 8649.
11 R. M. Noyes, Ber. Bunsen-Ges. Phys. Chem., 1980, 84, 295.
12 B. P. Belousov, Sb. Ref. Radiat. Med., 1959, 145, 1958.
of I only.
2
1
1
3
4
A. M. Zhabotinsky, Dokl. Akad. Nauk SSSR., 1959, 157, 392.
W. C. Bray, J. Am. Chem. Soc., 1921, 43, 1262.
Conclusion
15 G. Nicolis and J. Portnow, Chem. Rev., 1973, 73, 371.
1
6 T. C. Briggs and W. C. Rauscher, J. Chem. Educ., 1973, 50, 496.
Our proposed mechanism is the most concise that can be gen-
erated and yet still fully explain the observed reaction
dynamics. An important subset of reactions completely
ignored in this mechanism is the group of sulfurÈsulfur reac-
tions. These can help, especially, to explain what happens to
the intermediate sulfur species in excess reductant. Through
autooxidations and disproportionations, the sulfur centres will
eventually end up in the preferred oxidation states of [2, 0
and ]6. By maintaining HOI as the sole important oxidant in
solution, errors that might arise from omitting sulfurÈsulfur
reactions have been minimized. For example, two sulÐnic acid
centres can disproportionate:
17 R. M. Noyes, J. Am. Chem. Soc., 1980, 102, 4644.
18 R. J. Field and R. M. Noyes, J. Chem. Phys., 1974, 60, 1877.
19 D. Banard, J. Chem. Soc., 1957, 4675.
20
21
22
G. Rabai and M. Beck, J. Chem. Soc., Dalton T rans., 1985, 1669.
S. J. Dushman, J. Phys. Chem., 1904, 8, 453.
A. D. Autrey and R. E. Connick, J. Am. Chem. Soc., 1953, 73,
1
842.
23 D. A. Skoog and D. M. West, Analytical Chemistry: An Intro-
duction, Saunders College Publishing, San Francisco 4th edn.,
1
986, p. 83.
2
2
4
5
H. Landolt, Ber. Dtsch. Chem. Ges., 1886, 19, 1313.
D. C. Harris, Quantitative Chemical Analysis, W. H. Freeman and
Co., San Francisco, 1982, p. 384.
2
6
P. Soupart, in Amino Acid Pools, ed. J. T. Holden, Elsevier, New
York, 1962, pp. 220È262.
SII ] SII ] SIV ] SO
(19)
2
2
7
8
J. G. Jacobson and L. H. Smith, Physiol. Rev., 1968, 48, 424.
In the oxidation of thiourea by Br (aq), for example, an increase
There is no quantitative way of measuring sulfate production
as the reaction proceeds, although experimental data shows
that sulfate production is not immediate (there is a time lag
2
in the reductant concentration (thiourea) increased the induction
period, see R. H. Simoyi and I. R. Epstein, J. Phys. Chem., 1987,
91, 5124.
between mixing of solution and production of BaSO
precipitate). If sulfate production can be properly monitored,
29 R. M. Noyes, L. V. Kalachev and R. J. Field, J. Phys. Chem.,
1995, 99, 3514.
4
3
3
0
1
M. Eigen and K. Kustin, J. Am. Chem. Soc., 1962, 84, 1355.
then it will be easier to predict the point of SwS bond cleav-
age.
The NMR data strongly suggest that the initial step is the
formation of the sulfone and not the cleavage of the SwS
bond. Cleavage of the SwS bond, it appears, occurs e†ectively
when each of the sulfur atoms is coordinately saturated.
T. R. Thomas and D. T. Pence, J. Inorg. Nucl. Chem., 1980, 42,
1
83.
3
2
3
S. D. Furrow and R. M. Noyes, J. Am. Chem. Soc., 1982, 104, 38.
3
L. Treindl and R. M. Noyes, J. Phys. Chem., 1993, 97, 11354.
34 M-F. Ruasse, J. Aubard, B. Galland and A. Adenier, J. Chem.
Phys., 1986, 90, 4382.
3
5 P. Kaps and P. Rentrop, Numer. Math., 1979, 23, 55.
We would like to acknowledge helpful discussions with Pro-
fessors Jonnalagadda and Martincigh. This work was sup-
Paper 6/06729H; Received 1st October, 1996
1550
J. Chem. Soc., Faraday T rans., 1997, V ol. 93

 Mundoma, Claudius
Mundoma, Claudius
 Simoyi, Reuben H.
Simoyi, Reuben H.