9838
J. Phys. Chem. A 1998, 102, 9838-9846
Antioxidant Chemistry: Hypotaurine-Taurine Oxidation by Chlorite1
Bice S. Martincigh
Department of Chemistry and Applied Chemistry, UniVersity of Natal, PriVate Bag X10,
Dalbridge 4014, Republic of South Africa
Claudius Mundoma and Reuben H. Simoyi*
Department of Chemistry, West Virginia UniVersity, Morgantown, West Virginia 26506-6045
ReceiVed: June 10, 1998; In Final Form: September 24, 1998
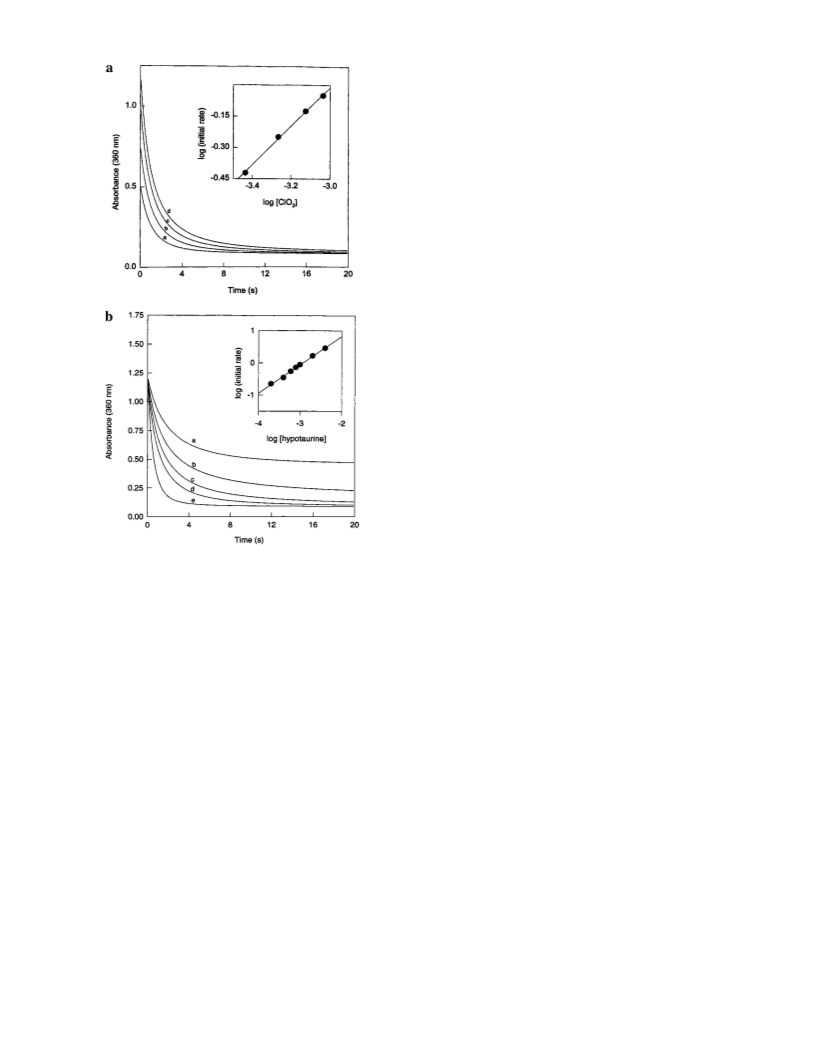
Extensive experimental data have been collected on the oxidation of hypotaurine, H2NCH2CH2SO2H, by
chlorite and chlorine dioxide. Hypotaurine is stable and reacts slowly with chlorite to give taurine, hypotaurine
sulfonic acid, and monochloro- and dichlorotaurine. However, it reacts rapidly with chlorine dioxide with a
second-order rate constant of 801 M-1 s-1 to give taurine. Oxidation occurs simultaneously at the sulfur
center (to give the sulfonic acid) and at the nitrogen center (to give the chloramines). The stoichiometry of
the reaction was experimentally determined to be ClO2- + H2NCH2CH2SO2H + H+ f ClHNCH2CH2SO3H
+ H2O. The formation of dichlorotaurine is favored only in high acid environments.
Introduction
be responsible for mopping up HOCl.12 Specific studies are
thus needed for each suspected antioxidant compound before
its mechanism for antioxidant activity can be deduced.
Although the physiological roles antioxidants play in the
human body have been studied2 extensively, in vitro antioxidant
chemistry studies are still in their infancy. There is a need to
study mechanistic details of how antioxidants protect tissue from
harmful oxidants. A good start would be to identify common
antioxidants and study their reactivity patterns in vitro.
Apart from its antioxidant activity, taurine has been implicated
in several other physiological roles.14 However, there is no
adequate mechanistic data on how it functions in these roles.
Most reports on mechanistic studies are speculative. For
example, it has been reported that in physiological concentra-
tions taurine can, in vitro, kill schistosoma mansoni.15 Also,
taurine has been shown to inhibit calcium-activated respiration
in small mammalian liver mitochondria,16 to inhibit nerve
impulses in nerve tissues,17 and to reduce cholesterol in
vertebrates.18
The biochemistry and clinical biology communities have
different definitions of an antioxidant.3 In this paper, we will
assume that an antioxidant is a substance that, when present in
low concentrations, can significantly delay oxidation of an
oxidizable substrate.4 Previous work suggests that hypotaurine
and taurine fall into this category. A number of reactive oxygen
species (•OH and O2•-) are produced in vivo, and they oxidize
and damage normal tissue.5 •OH can easily be produced in
biological systems by the metal-ion-dependent decomposition
of hydrogen peroxide.6 Antioxidants should be able to scavenge
all reactive oxygen species (ROS) and thus protect tissue from
damage and also to reduce inflammation.7
Taurine and hypotaurine are closely linked. Taurine, with
its special stability, is considered a metabolic end product of
methionine and cysteine.19 Most metabolic pathways give
hypotaurine as a precursor to taurine.19 Hypotaurine differs from
taurine only in the oxidation state of the sulfur center and the
resulting change in the acidity of the molecule.
In general, hypotaurine is much more reactive than taurine.
Recent experimental data have shown that hypotaurine reacts
rapidly and efficiently with the hydroxyl radical, superoxide
radical, and hydrogen peroxide.20 The ability to scavenge these
reactive oxygen species rapidly is a prerequisite for a molecule
to act as an antioxidant in vivo.21 The other metabolic pre-
cursors of taurine, cysteic acid and cysteamine, are also poor
scavengers of ROS as is taurine. Taurine also appears to be
inefficient in moderating HOCl toxicity because the N-chlor-
amine produced can still deactivate R1-antiproteinase.22
Some of our recent work examined the reactivity of taurine.23
We found taurine to be extremely inert and surprisingly inactive
to oxidation by the reactive radical oxyhalogen species ClO2
and by acidified bromate.23 It did appear that, whatever
physiological roles taurine might play, it does not seem to be a
strong and effective antioxidant.
The major antioxidants most commonly mentioned (cysteine,
glutathione, taurine, hypotaurine, vitamin E, ascorbic acid,
superoxide dismutase, and caeruloplasmin)8 all contain an amino
group. The formation of N-chloro- and N-bromomines has been
suggested as possible mechanisms by which the most common
harmful oxidants, HOCl and HOBr, can be scavenged.9 With
the high concentrations of Cl- (0.10 M) and Br- (0.001 M) in
the human body, HOCl and HOBr can be easily produced from
the myeloperoxidase-catalyzed peroxidation of chloride and
bromide ions, respectively.10 These neutrophil-derived prooxi-
dants, HOCl and HOBr, are important in bacterial killing but
also damage tissue at inflammation sites.11
A recent study showed that cephalosporins are scavengers
of HOCl.12 Even some cephalosporins without a primary amino
group (such as cefamandale) have been shown to have powerful
antioxidant activity with respect to HOCl, leading to a reduced
antibiotic activity of the cephalosporin.13 Cephalosporins
contain thioether groups: speculation is that these groups may
In this paper we report an extensive kinetics and mechanistic
study of the reaction of hypotaurine with chlorite and chlorine
10.1021/jp982575c CCC: $15.00 © 1998 American Chemical Society
Published on Web 11/05/1998

 Martincigh, Bice S.
Martincigh, Bice S.
 Mundoma, Claudius
Mundoma, Claudius
 Simoyi, Reuben H.
Simoyi, Reuben H.