Q. Chu et al.
Catalysis Communications 108 (2018) 46–50
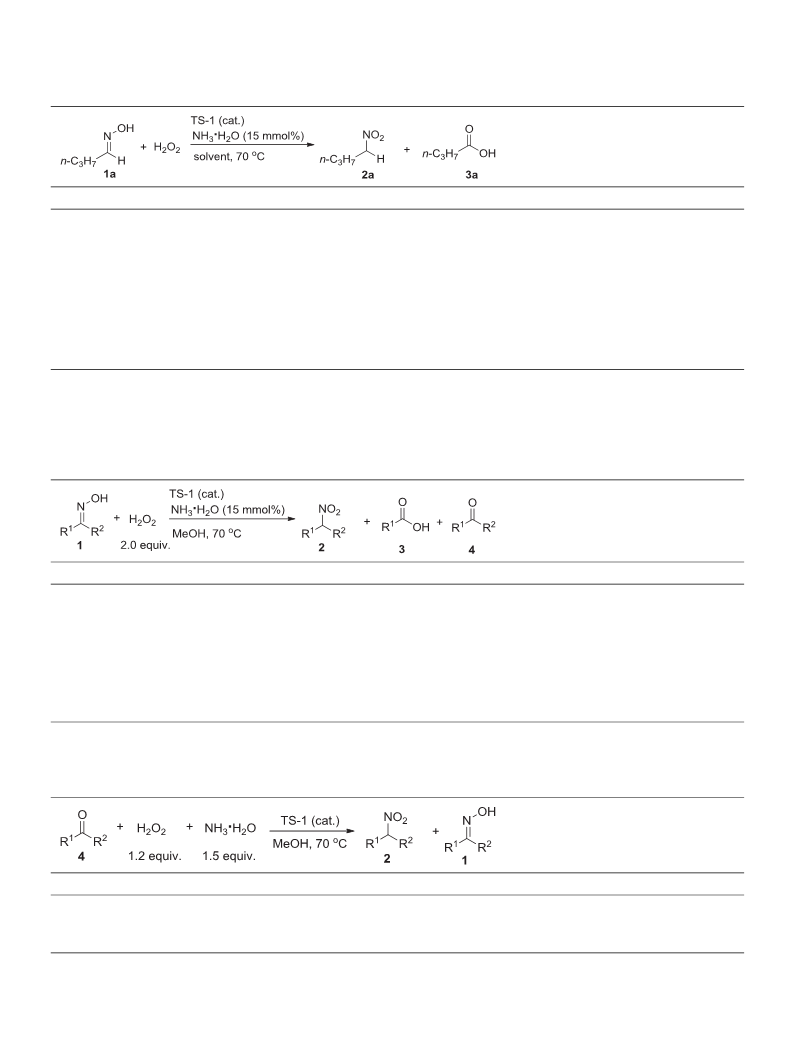
2.2. Synthesis of nitroalkanes
TS-1 catalyzed oxidation of ketones or oximes to prepare nitroalk-
anes was carried out in a reaction vessel. A solution of ketones or
oximes, TS-1 catalyst and NH ·H O in MeOH was mixed and heated at
0 °C. To this mixture was slowly injected H by a peristaltic pump
3
2
7
2 2
O
within 30 min. After stirring for an additional 1 h, the progress of the
reaction was monitored by Agilent 7820A GC System. Upon comple-
tion, the resulting mixture was cooled to r.t. and TS-1 catalyst filtered
off. Then, saturated NaHSO
3
aqueous solution was added to deal with
the unreacted H . After removing MeOH by distillation, the residual
2 2
O
aqueous phase was extracted with DCM. The combined organic layer
was concentrated in vacuum to give the nitroalkanes. All reactions were
performed in reaction vessels at atmospheric pressure.
3. Results and discussion
3.1. Catalyst characterization
The morphology analysis of TS-1 was shown in Fig. S1. It is ob-
Fig. 1. Py-IR spectra of TS-1.
served that the well-defined, uniform cubic crystallites are distributed
homogeneously with particle size of about 1.1 × 0.64 × 0.24 μm in the
SEM (Fig. S1a) and TEM images (Fig. S1b). And the lattice structures of
TS-1 can be well distinguished from the TEM images (Fig. S1c and S1d).
Moreover, the clear lattice fringes with two spacing intervals (1.1 nm
and 0.36 nm) correspond to the crystallographic planes of monoclinic
that Si existed in the TS-1 (Fig. S5b).
.2. Synthesis of nitroalkanes from oximes
3
o
Next, we tested the reactivity of TS-1 through the oxidation of n-
butanaldoxime (1a) with H in the presence of NH ·H O at 70 °C
Table 1). The experimental results showed that the amount of TS-1 had
TS-1, which are in well accord with XRD (Fig. S3) peaks of 7.8 and
o
2
O
2
3
2
2
4.4 . To gain insights into the distribution of TS-1, elemental map-
(
pings were taken (Fig. S1e). It could be seen that the three elements of
Ti, Si and O were evenly distributed in the prepared catalytic material.
Furthermore, the XRF analysis showed that the Si/Ti molar ratio of the
TS-1 was 38.
an important effect on the reaction. When 25 mg of TS-1 was used, the
reaction afforded the 1-nitrobutane (2a) in only less than 10% yield
with the 1:1 ratio of 2a/3a. And the yield could be dramatically in-
creased to 96% by adding 100 mg of TS-1 to the reaction (entry 3). In
fact, lowering the reaction temperature caused lower conversion rate
and inferior selectivity (entries 4–5). However, increasing the con-
The FT-IR spectrum of TS-1 sample showed that an obvious IR ab-
−
1
sorption peak at 983 cm
(Fig. S2), indicating the collective vibration
of Si-O-Ti or Si-O bond influenced by Ti(IV) of TS-1 framework [24].
The results confirmed that titanium was present in the TS-1 framework
2 2
centration of H O or reducing its amount did not maintain a sa-
tisfactory result (entries 6–8). Solvents screening experiments indicated
that MeOH was a better reaction medium than EtOH, i-PrOH, t-BuOH,
and MeCN (entry 3 vs. entries 9–12). No reaction occurred in the ab-
sence of catalyst (entry 13). Thus, the reaction efficiently proceeded
when 100 mg of TS-1 was used in combination with H
in MeOH at 70 °C (entry 3).
Having established the standard conditions for the TS-1 catalyzed
oxidative nitration (Table 1, entry 3), we next moved to the scope and
limitation studies using a variety of structurally diverse oximes
[
25]. The XRD peaks at 2θ = 7.8°, 8.8°, 23.2°, 23.8° and 24.3° showed
the MFI topological structure of TS-1 (Fig. S3) [26]. And the diffraction
peak at 2θ = 25.3° was not observed, which confirmed that antatase
TiO
2
is not formed in TS-1 [27].
2 2
O (2.0 equiv.)
As shown in the N
2
adsorption-desorption isotherms (Fig. S4), the
adsorption isotherm coincided well with the desorption isotherm from
relative pressure values between 0 and 0.47, indicating that the pre-
sence of micropores. A hysteresis loop was clearly observed from re-
lative pressures of 0.5 to 1.0, which showed the presence of mesopores.
And the upward tail of the N adsorption-desorption isotherm showed
2
the presence of macropores. The formation of mesopores and macro-
(
Table 2). The reactions of acetaldoxime, propionaldoxime, and n-
pentaldoxime with H proceeded smoothly and afforded the corre-
2 2
O
sponding 1-nitroalkanes in 83% to 97% yields with excellent selectiv-
ities (entries 2–4). However, the oxidation reactivity dropped sharply
with an increase of the carbon chain length of oxmies (entries 5–6), and
the reaction did not even work at all for decanaloxime (entry 7). This
was probably caused by the steric hindrance of substrates impeding the
interaction of oximes with the active sites of TS-1. Unexpectedly,
acetoxime (1 h) could also be oxidized to 2-nitropropane in 91% yield
with 93/7 selectivity under the standard conditions (entry 8). Similarly,
the yield and selectivity of the TS-1 catalyzed oxidation markedly de-
creased with the increase of the steric hindrance from alkyl group on
ketone oximes (entries 9–11).
pores is caused by TS-1 stacking. Further, TS-1’s BET specific surface
area, pore volume, and pore size are 328.23 m /g, 0.19 cm /g and
2
3
2.29 nm, respectively.
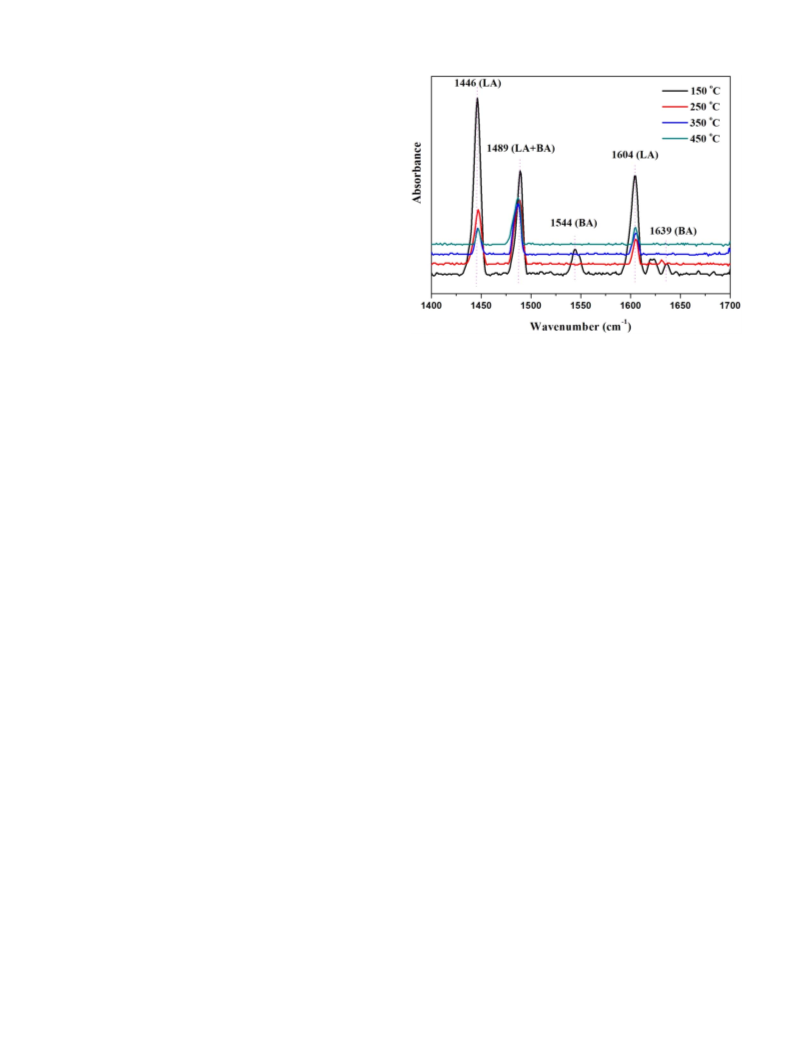
The Py-IR spectra (Fig. 1) were employed to evaluate the types and
strength of Brönsted and Lewis acid sites on the surface of prepared TS-
−
1
1
[28,29]. The bands of 1446 and 1604 cm , and the bands at 1544
−
1
and 1639 cm
demonstrated the existence of Lewis acid and Brönsted
acid sites, respectively. And the band at 1489 cm
−
1
was a combination
−
1
between two separate bands at 1446 and 1544 cm . In addition, the
changes of Brönsted and Lewis acidity were also observed at different
desorption temperatures (150, 250, 350 and 450 °C). As shown in
Fig. 1, the Brönsted acidity reduced more rapidly than the Lewis acidity
with the temperature rising, which indicated the acidic sites of Lewis
acidity are more than that of Brönsted acidity in TS-1 catalyst.
3.3. Synthesis of nitroalkanes from aldehydes/ketones
As shown in Fig. S5a, the binding energy values of Ti 2p3/2 and 2p1/2
were observed at 458.5 and 465.0 eV, respectively. And a 6.5 eV
splitting from Ti 2p3/2 to Ti 2p1/2 in the XPS spectrum indicated that all
Inspired by the oxidative results of oximes, we speculated whether
low-carbon aldehydes and ketones could also be directly oxidized to
corresponding nitroalkanes by the protocol. After many modifications
to the established standard conditions, we still could not observe the
4
+
titanium as octahedron Ti
existed in the TS-1 [30,31]. The binding
energy value of Si 2p was also observed at 103.4 eV, which indicated
2 2
oxidation from n-butanal to 1-nitrobutane in the presence of H O and
47

 Chu, Qingyan
Chu, Qingyan
 He, Guangke
He, Guangke
 Xi, Yang
Xi, Yang
 Wang, Ping
Wang, Ping
 Yu, Haoxuan
Yu, Haoxuan
 Liu, Rui
Liu, Rui
 Zhu, Hongjun
Zhu, Hongjun