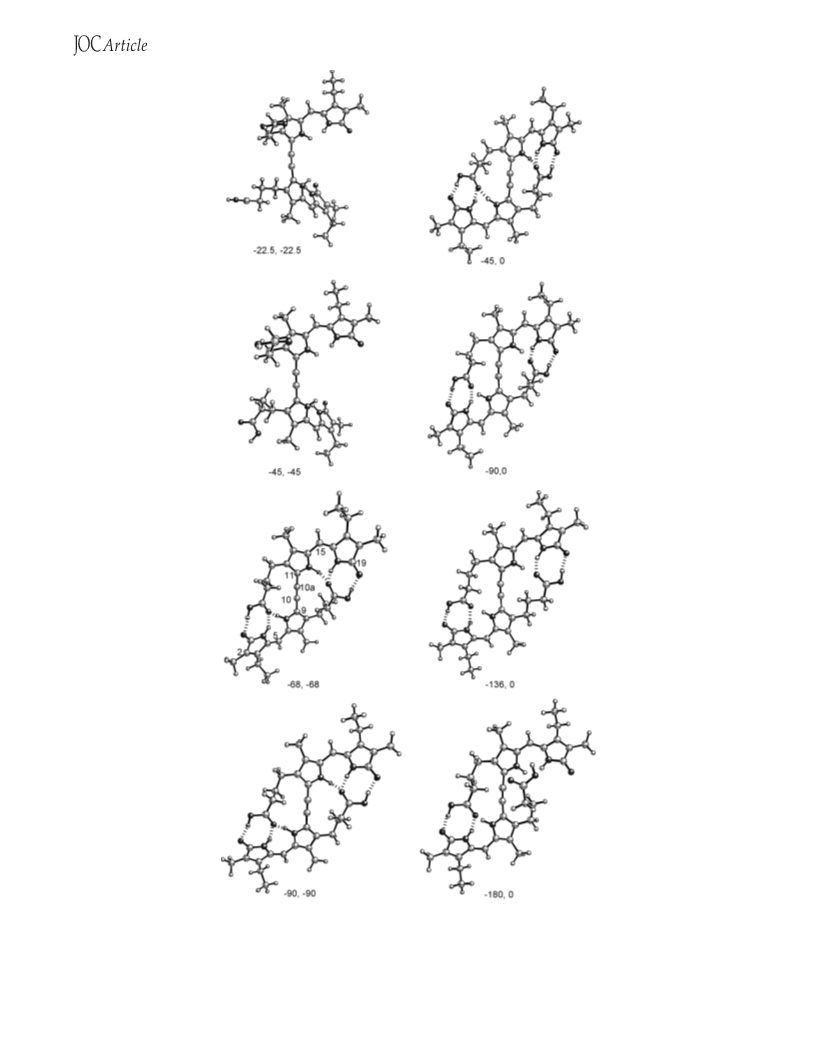
Tu et al.
(5-Iod o-2-for m yl-3-m et h yl-1H -4-p yr r olyl)-4-b u t a n oic
Acid (6b). Compound 6b was prepared in 60% yield as
described above for (5-iodo-2-formyl-3-methyl-1H-4-pyrrolyl)-
3-propanoic acid 6a : mp 152-3 °C; IR (KBr film) ν 3164, 2915,
1686, 1673, 1425, 1266, 833 cm-1; 1H NMR (DMSO-d6) δ 1.57
(m,, J ) 7.2 Hz, 2H), 2.15 (t, J ) 7.2 Hz, 2H), 2.28 (t, J ) 7.2
Hz, 2H), 2.20 (s, 3H), 9.35 (s, 1H), 12.0 (brs, 1H), 12.2 (brs,
1H) ppm; 13C NMR (DMSO-d6) δ 9.4, 25.3, 25.5, 33.5, 83.4,
Meth yl (2-F or m yl-3-m eth yl-5-(tr im eth ylsilyl)eth yn yl-
1H-p yr r olyl-4-bu ta n oa te (4b). Compound 4b was prepared
in 80% yield as described above for methyl (2-formyl-3-methyl-
5-(trimethylsilyl)ethynyl-1H-4-pyrrolyl-3-propanoate 4a : mp
64-5 °C; IR (NaCl, dichloromethane) v 3441, 1730, 1647, 1445,
1094, 906, 732, 650 cm-1; 1H NMR δ 0.25 (s, 9H), 1.85 (m, J )
7.5 Hz, 2H), 2.27 (s, 3H), 2.33 (t, J ) 7.5 Hz, 2H), 2.53 (t, J )
7.5 Hz, 2H), 3.66 (s, 3H), 8.88 (brs, 1H), 9.58 (s, 1H) ppm; 13
C
NMR δ -0.28, 8.5, 23.1, 24.9, 33.3, 51.4, 94.8, 102.6, 118.7,
100.6, 129.2, 134.0, 174.5, 177.2 ppm. Anal. Calcd for C10H12
-
129.2, 129.4, 130.1, 173.8, 177.0 ppm; GC-MS (m/z) 305 [M+•],
INO3 (321.2): C, 37.40; H, 3.77; N, 4.36. Found: C, 37.40; H,
4.02; N, 4.05.
274, 218, 202, 172, 144, 88, 73 amu. Anal. Calcd for C16H23
-
NO3Si (305.4): C, 62.92; H, 7.59; N, 4.59. Found: C, 62.70; H,
7.38; N, 4.86.
Met h yl (5-Iod o-2-for m yl-3-m et h yl-1H -4-p yr r olyl)-3-
p r op a n oa te (5a ). To a 125 mL Erlenmeyer flask were added
aq KOH (2.5 g in 2.5 mL water) and 20 mL of ether. The
mixture was cooled to 0 °C, and N-methyl-N-nitrosourea (0.55
g) was added and stirred slowly for 10 min. The yellow ether
layer was poured into a solution of (5-iodo-2-formyl-3-methyl-
1H-pyrrolyl)-3-propanoic acid 6a (200 mg, 0.652 mmol) in
methanol (15 mL) in two portions during 5 min. The reaction
mixture was stirred for another 10 min until no further N2
evolved. Glacial acetic acid (0.5 mL) was then added to destroy
excess diazomethane. The mixture was poured into water (50
mL) and extracted with dichloromethane (3 × 50 mL). The
combined organic layers were washed with water (2 × 50 mL),
saturated NaHCO3 solution (2 × 50 mL), and brine (2 × 50
mL) and then dried over anhydrous Na2SO4. After evaporation
of the solvent, the residue was recrystallized from chloroform-
hexane to give pure 5a : 170 mg, 78% yield; mp 142-144 °C;
IR (NaCl, dichloromethane) ν 3432, 2253, 1635, 1412, 1363,
1094, 909, 734, 650 cm-1; 1H NMR δ 2.33 (s, 3H), 2.47 (t, J )
7.5 Hz, 2H), 2.72(t, J ) 7.5 Hz, 2H), 3.69 (s, 3H), 9.11 (brs,
1H), 9.40 (s, 1H) ppm; 13C NMR δ 8.9, 21.6, 34.0, 51.7, 128.1,
128.8, 130.5, 133.9, 172.9, 176.1 ppm; GC-MS (m/z) 321 [M+],
Meth yl (2-F or m yl-3-m eth yl-5-eth yn yl-1H-4-p yr r olyl)-
3-p r op a n oa te (3a ). To a solution of pyrrole 4a (70 mg, 0.24
mmol) in THF (3 mL) was added n-Bu4NF (1.0 M THF
solution, 0.25 mL). The mixture was stirred at room temper-
ature for 1 h, and the color darkened. After removal of the
solvent under reduced pressure, the residue was passed
through a short column of silica gel using dichloromethane-
hexane (3:1 by volume) as eluent to give a yellow solution.
After evaporation of the solvent, 3a was obtained as a pale
yellow solid: 50 mg, 85% yield; mp 129-130 °C; IR (NaCl,
dichloromethane) ν 3441, 2252, 1647, 1378, 1094, 907, 737 650
cm-1; 1H NMR δ 2.30 (s, 3H), 2.57 (t, J ) 7.5 Hz, 2H), 2.84 (t,
J ) 7.5 Hz, 2H), 3.46 (s, 1H), 3.68 (s, 3H), 9.08 (brs, 1H), 9.60
(s, 1H) ppm; 13C NMR δ 8.5, 19.7, 34.0, 51.6, 74.0, 84.5, 117.6,
129.3, 129.4, 129.5, 173.1, 177.3 ppm; GC-MS (m/z) 219 [M+•],
160, 146, 132, 103, 77, 51 amu. Anal. Calcd for C12H13NO3
(219.2): C, 65.74; H, 5.98; N, 6.39. Found: C, 65.92; H, 6.18;
N, 6.42.
Met h yl (2-F or m yl-3-m et h yl-5-et h yn yl-1H -p yr r olyl-4-
bu ta n oa te (3b). Compound 3b was prepared in 85% yield as
described above for methyl (2-formyl-3-methyl-5-ethynyl-1H-
pyrrolyl-3-propanoate (3a ): mp 88-9 °C; IR (NaCl, dichlo-
262, 248, 194, 179, 152, 106, 77 amu. Anal. Calcd for C10H12
-
INO3 (321.1): C, 37.40; H, 3.77; N, 4.36. Found: C, 37.67; H,
3.62; N, 4.06.
romethane) ν 3443, 2250, 1648, 1370, 1094, 907, 737 649 cm-1
;
1H NMR δ 1.87 (m, J ) 7.5 Hz, 2H), 2.28 (s, 3H), 2.33 (t, J )
7.5 Hz, 2H), 2.55 (t, J ) 7.5 Hz, 2H), 3.43 (s, 1H), 3.66 (s, 3H),
9.16 (brs, 1H), 9.60 (s, 1H) ppm; 13C NMR δ 8.5, 23.5, 25.0,
33.2, 51.4, 74.3, 84.1, 117.9, 129.4, 129.7, 130.3, 173.8, 177.3
ppm; GC-MS (m/z) 233 [M+•], 202, 174, 146, 130, 117, 77, 65
amu. Anal. Calcd for C13H15NO3 (233.2): C, 66.94; H, 6.48; N,
6.00. Found: C, 66.65; H, 6.59; N, 5.71.
Meth yl (5-Iod o-2-for m yl-3-m eth yl-1H-4-p yr r olyl)-4-bu -
ta n oa te (5b). Compound 5b was prepared in 90% yield as
described above for methyl (5-iodo-2-formyl-3-methyl-1H-4-
pyrrolyl)-3-propanoate 5a : mp 124-5 °C; IR (NaCl, dichlo-
romethane) ν 3430, 1635, 1412, 1360, 1095, 909, 732, 649 cm-1
;
1H NMR δ 1.79 (m, J ) 7.5 Hz, 2H), 2.31 (s, 3H), 2.34 (t, J )
7.5 Hz, 2H), 2.42 (t, J ) 7.5 Hz, 2H), 3.67 (s, 3H), 9.40 (s, 1H),
9.40 (brs, 1H) ppm; 13C NMR δ 8.9, 24.9, 25.5, 33.2, 51.5, 80.9,
129.9, 130.2, 133.9, 173.7, 176.0 ppm; GC-MS (m/z) 335 [M+•],
1,2-Bis(3-m eth oxyca r bon yleth yl-4-m eth yl-5-for m yl-2-
p yr r olyl) (2a ). To a solution of pyrrole 3a (103 mg, 0.47 mmol)
and pyrrole 5a (154 mg, 0.48 mmol) in diethylamine (4.8 mL)
were added dichlorobis(triphenyphosphine)palladium(II) (5.9
mg, 0.008 mmol) and copper(I) iodide (3.0 mg, 0.016 mmol)
under an N2 atmosphere. The mixture was stirred at 50 °C
for 3 h. The final brown solution was evaporated under vacuum
to remove all of the solvent. The residue was passed through
a short column of silica gel using dichloromethane and ethyl
acetate (2:1, v/v) as eluent. All of the yellow eluent was
collected. After the solvent was evaporated (roto-vap), the
yellow solid was recrystallized from hot ethyl acetate to afford
acetylenic dipyrrole dialdehyde 2a as yellow crystals: 116 mg,
60% yield; mp > 216 °C dec; IR (KBr) ν 3216, 1741, 1731, 1625,
1614, 1452, 1396, 1278, 1198, 1175, 668 cm-1; 1H NMR δ 2.33
(s, 6H), 2.60 (t, J ) 7.5 Hz, 4H), 2.89 (t, J ) 7.5 Hz, 4H), 3.67
276, 248, 208, 180, 148, 120, 65 amu. Anal. Calcd for C11H14
-
INO3 (335.1): C, 39.42; H, 4.21; N, 4.18. Found: C, 39.58; H,
4.16; N, 4.18.
Meth yl (2-F or m yl-3-m eth yl-5-(tr im eth ylsilyl)eth yn yl-
1H-4-p yr r olyl-3-p r op a n oa te (4a ). To a solution of methyl
(5-iodo-2-formyl-3-methyl-1H-4-pyrrolyl-3-propanoate (5a ) (295
mg, 0.92 mmol) in diethylamine (9.5 mL) were added under
N2 (trimethylsilyl)acetylene (0.138 g, 1.38 mmol), dichlorobis-
(triphenyphosphine)palladium(II) (11.5 mg, 0.016 mmol), and
copper(I) iodide (5.98 mg, 0.032 mmol). The homogeneous
mixture was stirred at 50 °C for 1 h, during which time the
color became yellow and a brown oil separated. After evapora-
tion of the solvent under vacuum, the residue was subjected
to chromatography on a short column of silica gel using
dichloromethane-hexane (2:1) as eluent. After evaporation of
the solvent, the residue was recrystallized from hexane-
chloroform to give pure pyrrole 4a : 240 mg, 90% yield; mp
92-3 °C; IR (NaCl, dichloromethane) ν 3441, 2253, 1731, 1644,
1
(s, 6H), 9.64 (s, 2H) ppm; H NMR (DMSO-d6) δ 2.22 (s, 6H),
2.49 (t, J ) 7.5 Hz, 4H), 2.73 (t, J ) 7.5 Hz, 4H), 3.52 (s, 6H),
9.57 (s, 2H), 12.27 (brs, 2H) ppm; 13C NMR (DMSO-d6) δ 9.0,
19.8, 34.3, 71.7, 86.5, 117.7, 128.9, 129.1, 130.4, 172.9, 178.7
ppm. Anal. Calcd for C22H24N2O6 (412.1): C, 64.07; H, 5.87;
N, 6.79. Found: C, 63.91; H, 5.87; N, 6.79.
1
1447, 1379, 1251, 1094, 848, 732, 650 cm-1; H NMR δ 0.25
(s, 9H), 2.28 (s, 3H), 2.57 (t, J ) 7.5 Hz, 2H), 2.82 (t, J ) 7.5
Hz, 2H), 3.68 (s, 3H), 8.95 (brs, 1H), 9.58 (s, 1H) ppm; 13C NMR
δ -0.34, 8.5, 19.8, 33.9, 51.5, 94.6, 103.0, 118.7, 129.1, 129.4,
130.2, 173.1, 177.0 ppm; GC-MS (m/z) 291 [M+•], 260, 218,
204, 158, 130, 89, 73 amu. Anal. Calcd for C15H21NO3Si
(291.2): C, 61.82; H, 7.26; N, 4.81. Found: C, 62.12; H, 7.50;
N, 4.94.
1,2-Bis(3-m eth oxycar bon yl-n -pr opyl-4-m eth yl-5-for m yl-
2-p yr r oyl)eth yn e (2b). Compound 2b was prepared in 40%
yield as described for 2a , by the condensation of pyrrole 3b
and 5b: mp > 185 °C dec; IR (KBr) ν 3211, 1740, 1727, 1625,
1452, 1391, 1278, 1190, 1175, 666 cm-1; 1H NMR δ 1.89 (m, J
) 7.5 Hz, 4H), 2.29 (s, 6H), 2.42 (t, J ) 7.5 Hz, 4H), 2.56 (t, J
) 7.5 Hz, 4H), 3.78 (s, 6H), 9.63 (s, 2H), 9.95 (brs, 2H) ppm;
8962 J . Org. Chem., Vol. 68, No. 23, 2003

 Tu, Bin
Tu, Bin
 Ghosh, Brahmananda
Ghosh, Brahmananda
 Lightner, David A.
Lightner, David A.