
Angewandte Chemie - International Edition p. 1188 - 1192 (2019)
Update date:2022-08-29
Topics:
 Music, Arif
Music, Arif
 Hoarau, Clément
Hoarau, Clément
 Hilgert, Nicolas
Hilgert, Nicolas
 Zischka, Florian
Zischka, Florian
 Didier, Dorian
Didier, Dorian
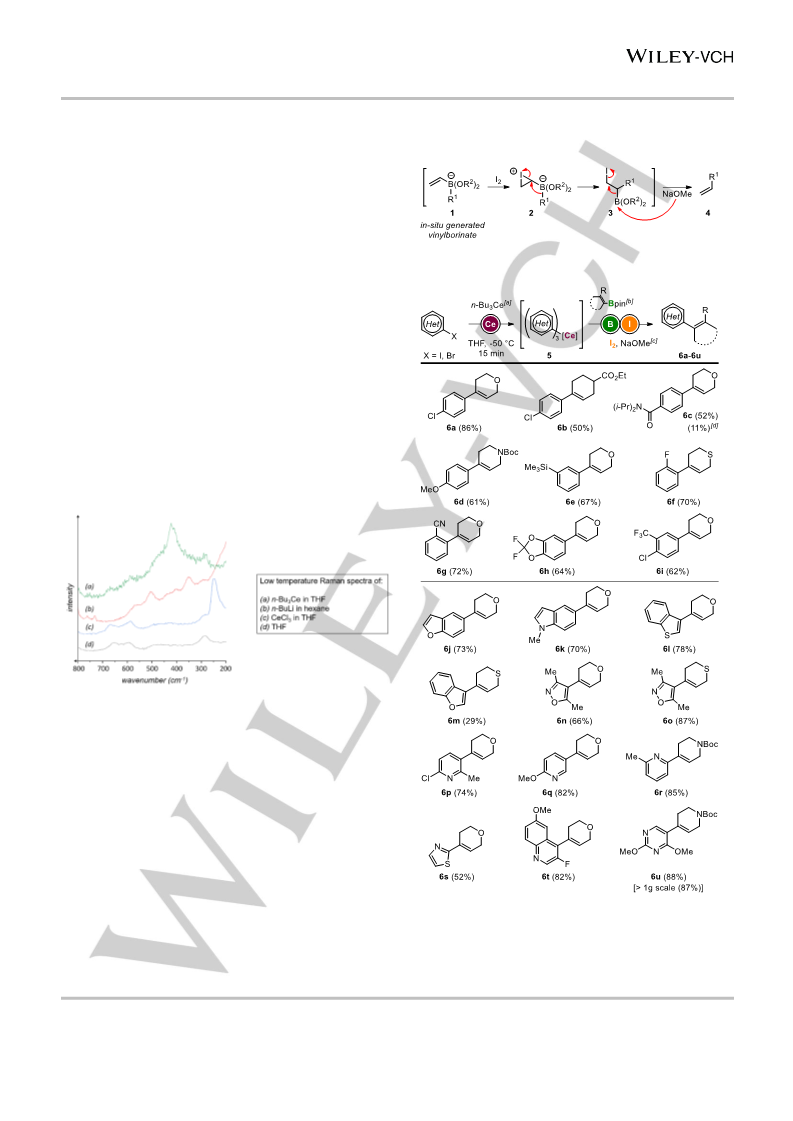
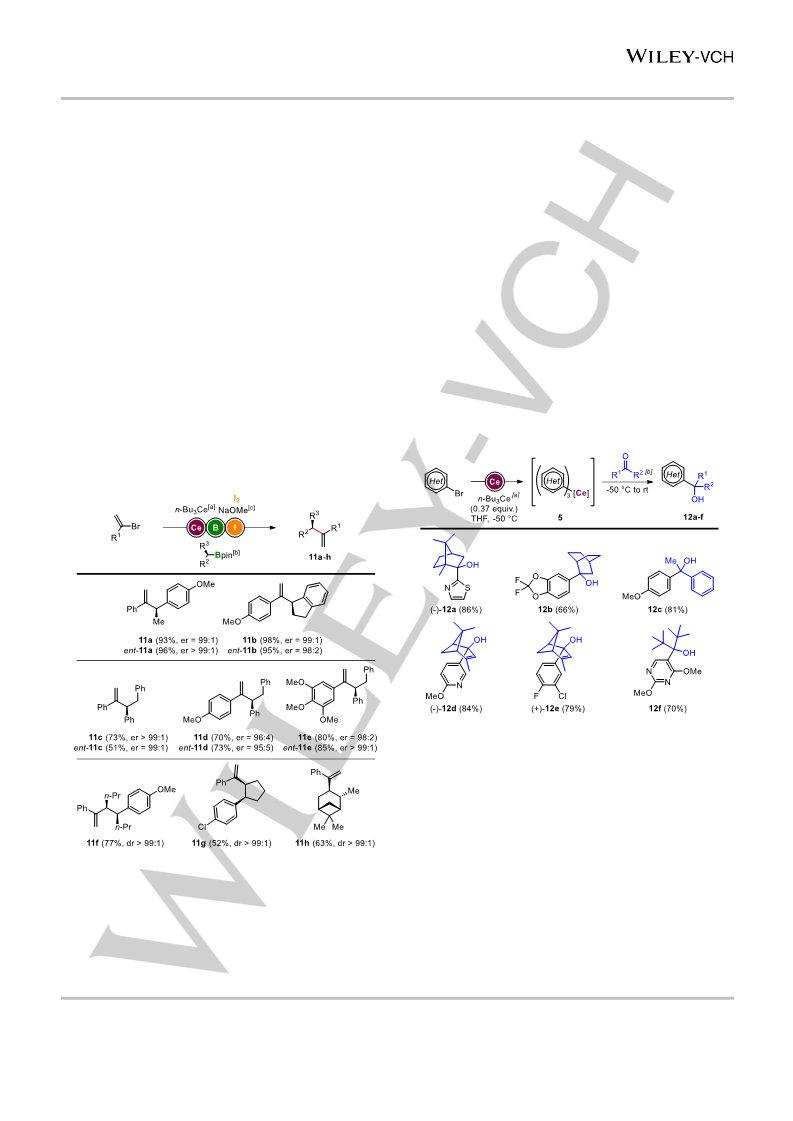
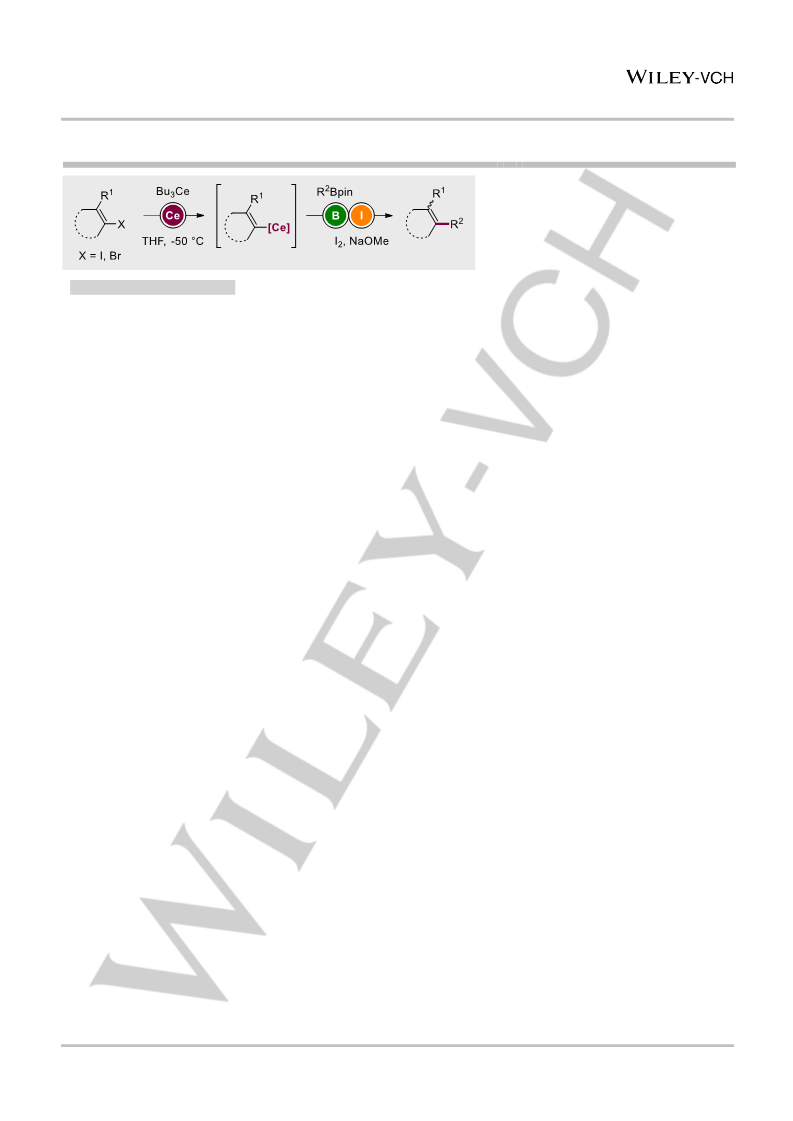
Described is the in situ formation of triorganocerium reagents and their application in catalyst-free Zweifel olefinations. These unique cerium species were generated through novel exchange reactions of organohalides with n-Bu3Ce reagents. The adequate electronegativity of cerium allowed for compensating the disadvantages of both usually functional-group-sensitive organolithium species and less reactive organomagnesium reagents. Exchange reactions were performed on aryl and alkenyl bromides, enabling enantiospecific transformations of chiral boron pinacol esters. Finally, these new organocerium species were engaged in selective 1,2-additions onto enolisable and sterically hindered ketones.
View More



Tianjin Te-An Chemtech Co., Ltd.
Contact:+86-22-65378638
Address:A5-8, No.80 Haiyun Street, TEDA
Contact:+86-571-86025531 / 86024803
Address:1218-24 Guangyin Mansion,42 Fengqi East Road
Hebei Kangtai Pharmaceutical Co.,Ltd
Contact:+86-0317-3512963
Address:Wugang Road,Mengcun of Cangzhou City,Hebei Province ,China
Suzhou Tianma Specialty Chemicals Co.,Ltd
Contact:+86-512-68090577
Address:#199, Huayuan Rd, Mudu, Suzhou, Jiangsu, CHINA
zhuzhou zhongle chemical co. ltd.
Contact:+86-0731 28228409
Address:Zhuzhou, Hunan, China
Doi:10.1007/s00706-015-1624-3
(2016)Doi:10.1039/c4ra10307f
(2014)Doi:10.1007/s10895-016-1823-2
(2016)Doi:10.1080/15421400490487353
(2004)Doi:10.1021/acs.organomet.5b00537
(2015)Doi:10.1016/j.apcata.2010.10.007
(2010)