L. Yang et al. / Journal of Catalysis 370 (2019) 310–320
311
reported. After modifying Ni with P or Si to form nickel silicides
[15,16,19,23] or nickel phosphides [24,25], the selectivity to styr-
ene at nearly 100% conversion of phenylacetylene was increased
to some extent, e.g., about 80% selectivity at 99% conversion for
N2Si/SBA-15 [23] and 88.2% selectivity at 98.6% conversion for
Ni2P/Al2O3 [25]. However, the modified catalysts in general had
yet to show an acceptable selectivity up to 90% at complete conver-
sion of phenylacetylene.
It is known in the literature that introduction of a second metal
such as Cu [11,12], Zn [11,12] and Ga [26] to Pd can effectively
improve the selectivity to styrene at high conversion of pheny-
lacetylene. This strategy has been tried recently for Ni-based cata-
lysts in spite of few studies available. Li et al. [27] prepared several
supported Ni-Ga intermetallic compounds via an in situ reduction
of layered double hydroxide (LDH) precursors, and the best cata-
lyst displayed around 72% selectivity at nearly 100% conversion.
Liu et al. [28] synthesized Ni3Ga/MgAl2O4 and Ni3Sn/MgAl2O4
nanocrystal catalysts that exhibited, respectively, 87% and 89%
selectivity at >99% conversion. Very recently, Liu et al. [29]
was also prepared using the same method except for without the
addition of aluminum precursor.
Ni-based monometallic and bimetallic supported catalysts were
prepared by a urea precipitation method [31,32]. A typical proce-
dure was as follows: first, predetermined amounts of nickel nitrate
(Ni(NO3)2ꢁ6H2O, ꢂ99%, Sinopharm) and M (M = Zn, Ga, Fe or Cu)-
bearing nitrate (Zn(NO3)2ꢁ6H2O, ꢂ99%, Sinopharm; Ga(NO3)3ꢁH2O,
99.99%, Sigma-Aldrich; Fe(NO3)3ꢁ9H2O, 99%, Sinopharm; Cu(NO3)2-
ꢁ3H2O, 99%, Sinopharm) and 0.5 g of SBA-15 or AlSBA-15(x) were
added to 15 g of deionized water and stirred at room temperature
for 1 h; next, 0.23 g of urea (ꢂ99%, Sinopharm) was added to the
above solution and stirred at 90 °C for 15 h; finally, the suspension
was filtered, washed with water, dried at 110 °C for 10 h, and cal-
cined at 500 °C for 5 h at a heating rate of 2 °Cꢁminꢀ1. Note that the
M-bearing nitrate was added only for NiMy/AlSBA-15 catalysts,
where y represented the nominal M/Ni atomic ratio; moreover,
the nominal total metal loading (Ni + M) was kept at 10 wt%.
2.2. Catalyst characterization
reported 90.3% selectivity at 95.8% conversion over
a LDH-
derived NiACu nanoalloy catalyst, but the selectivity at almost
100% conversion was not presented. It is clear that, as mentioned
above, the selectivity to styrene at nearly complete conversion of
phenylacetylene needs to be improved. In addition, worthy of
mention is that, to date, most of the catalysts reported in the liter-
ature have been tested using a model feedstock composed of
phenylacetylene and solvent such as methanol and n-hexane,
which is different from the practical situation where pheny-
lacetylene is present in excess of styrene [2,22]. In such a case it
is unclear whether the catalysts were still effective in removing
phenylacetylene and preserving styrene owing to the competitive
adsorption of phenylacetylene and styrene over the active sites
of catalyst [4].
The Brunauer-Emmett-Teller (BET) surface area (SBET), pore vol-
ume (Vpore) and pore diameter (dpore) of samples were determined
by N2 adsorption-desorption data collected at ꢀ196 °C on Micro-
metrics ASAP 2010. Before analysis the samples were degassed at
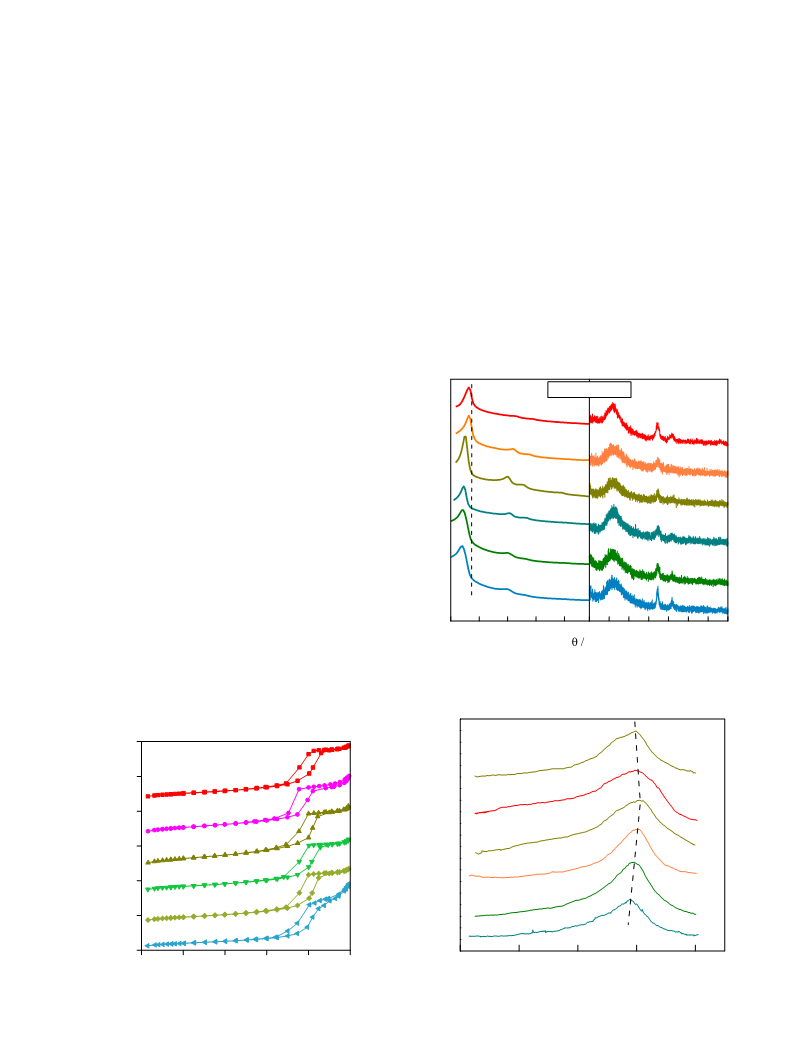
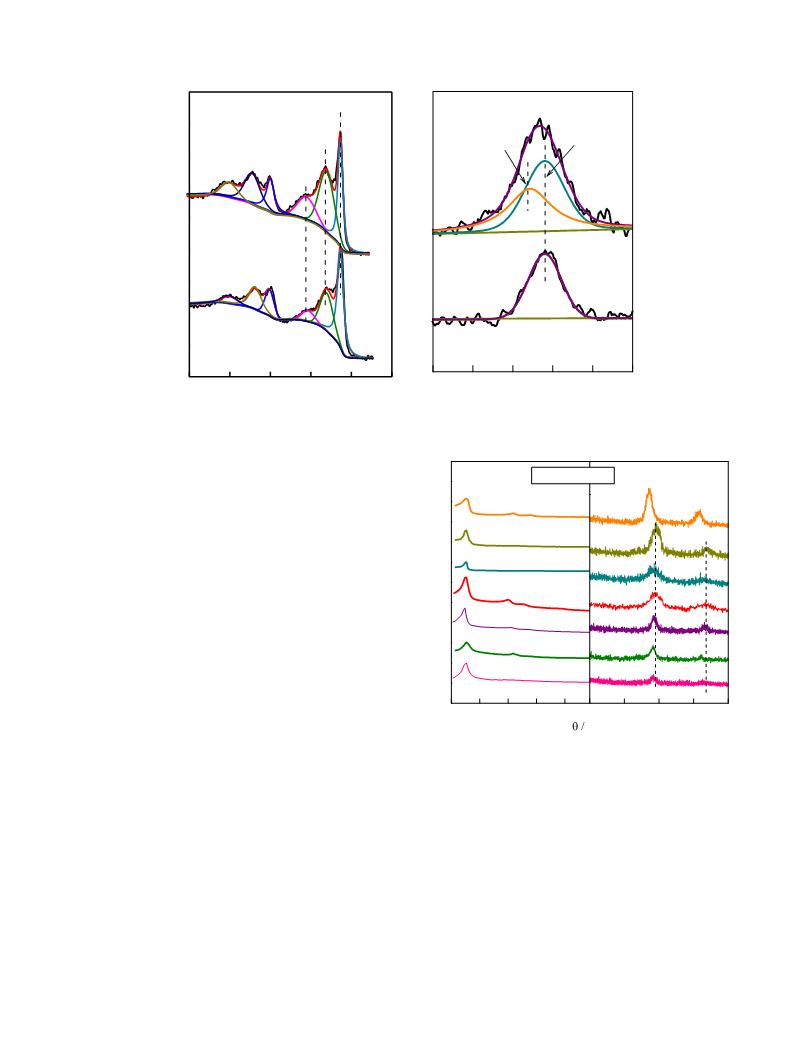
133 Pa and 200 °C for 6 h. Powder X-ray diffraction (XRD) on
Rigaku D/Max 2550 was conducted to confirm the crystalline
structure of sample by using a Cu K
a radiation (k = 0.15406 nm)
in the range of 2h = 0.5–5° and 10–80° at a scan rate of 0.02°ꢁsꢀ1
.
Inductively coupled plasma-optical emission spectroscopy (ICP-
OES) was performed on Varian 710-ES to detect the actual compo-
sition and metal loading of catalyst. High-resolution transmission
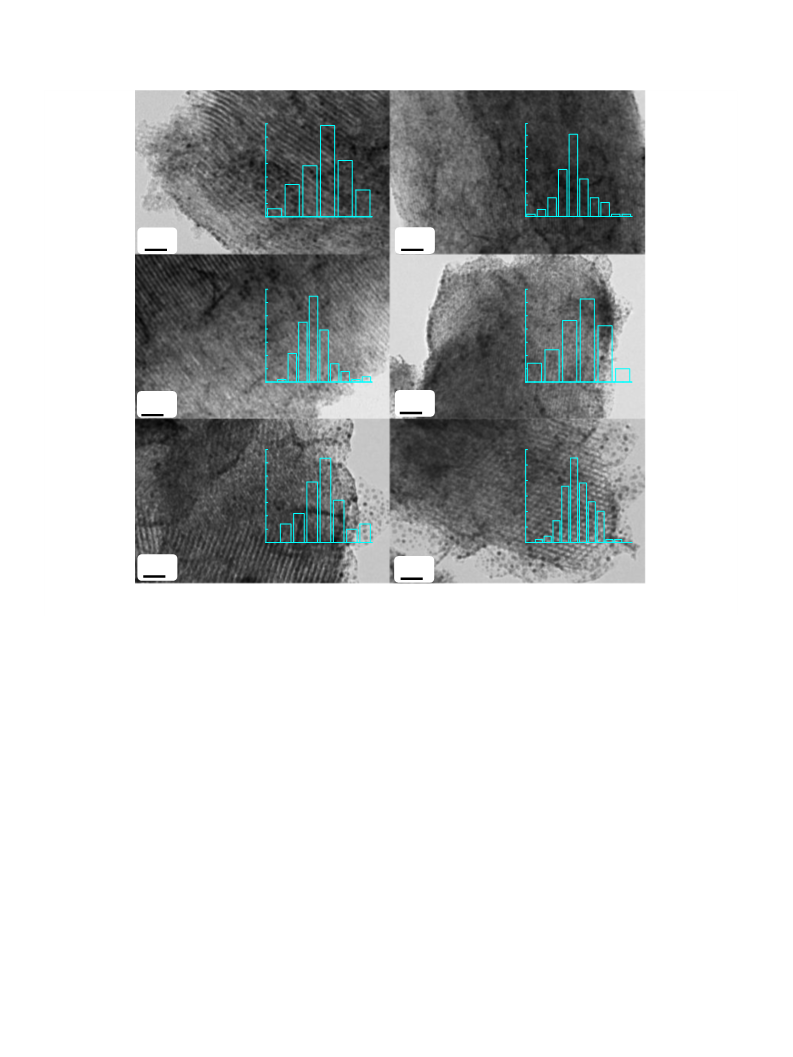
electron microscopy (HRTEM) was recorded on JEOL JEM-2100 to
observe the morphology and the metal particle size of catalyst.
Temperature- programmed reduction (TPR) of catalyst was carried
out on Micromeritics AutoChem 2920 to investigate the metal-
support interaction. The sample was reduced from room tempera-
ture to 800 °C in 10% H2/Ar at a heating rate of 10 °Cꢁminꢀ1, and the
H2 consumption was monitored by a thermal conductivity detector
(TCD). The amount of active sites of Ni-based catalysts were also
acquired on this instrument by CO pulse chemisorption, assuming
the chemisorption stoichiometry of CO:Ni = 1. The catalyst was
first in situ reduced at 800 °C for 2 h in 10% H2/Ar at a heating rate
of 10 °C minꢀ1, followed by switching to He at 830 °C for 30 min to
remove adsorbed H2. After that, the catalyst was cooled to room
temperature in He, and pulsed by CO until the CO peak area
remained constant. The CO uptakes were monitored by TCD and
calculated based on the accumulated adsorbed CO. X-ray photo-
electron spectroscopy (XPS) was investigated on Thermo Scientific
ESCALAB 250 Xi to analyze the surface electronic states of catalyst
In this study,
a series of monometallic Ni/AlSBA-15 and
bimetallic NiAM (M = Zn, Ga, Cu, or Fe)/AlSBA-15 nonprecious cat-
alysts with varying Si/Al atomic ratio were prepared by a urea pre-
cipitation method and applied to the semihydrogenation of
phenylacetylene. The structure- activity/selectivity relationships
of these catalysts were explored and discussed. The best catalyst
identified was assessed in the hydrogenation of model C8 aromatic
fraction of pygas (a mixture of phenylacetylene, styrene, ethylben-
zene and xylene) for 100 h in a fixed-bed reactor to simulate the
practical application. Moreover, the semihydrogenation of other
phenylacetylene derivatives was attempted.
2. Materials and methods
2.1. Preparation of Ni-based AlSBA-15 catalysts
by using an Al Ka radiation (hv = 1486.6 eV) and a pass energy of
The ordered mesoporous support AlSBA-15 was prepared by a
modified technique of Vinu et al. [30]. First, 30 g of deionized water
and 70 g of 0.28 molꢁLꢀ1 hydrochloric acid (36–38%, Sinopharm)
were mixed at 40 °C, and then 4.0 g of Pluronic P123 (Mav = 5800,
Sigma-Aldrich) was added with stirring until the P123 was com-
pletely dissolved. Next, 9.0 g of tetraethyl orthosilicate (ꢂ98%,
Sinopharm) and a predetermined amount of aluminium isopropox-
ide (ꢂ99%, Sinopharm) were added with the trace injection pump
under slowly stirring for 24 h at 40 °C, after which the solution was
transferred to a Teflon-lined stainless-steel autoclave and heated at
110 °C for 24 h. Finally, the precipitate was filtered, washed succes-
sively with deionized water and ethanol (ꢂ99.7%, Sinopharm),
dried at 80 °C for 10 h, and calcined at 550 °C for 6 h at a heating
rate of 1 °Cꢁminꢀ1. For simplicity, the as-prepared support was
denoted as AlSBA-15(x), where x (x = 5, 10, 20, 40 or 80) repre-
sented the nominal Si/Al atomic ratio. For comparison, SBA-15
40 eV, and the binding energies of spectra were referenced by C
1 s (284.8 eV).
2.3. Catalyst test
The semihydrogenation of phenylacetylene over various cata-
lysts was carried out in a semibatch stirred tank reactor (300 mL,
Parr 5100). First, 5 g of phenylacetylene (>98%, Alfa Aesar), 5 g of
n-octane (ꢂ98%, Sinopharm) acting as internal standard, 90 g of
methanol as solvent, and 0.15 g of pre-reduced catalyst (reduced
at 800 °C for 2 h in 10% H2/N2 at a heating rate of 10 °Cꢁminꢀ1) were
added into the reactor, and the mixture was heated to 40 °C in N2.
Next, H2 was charged into the reactor to purge N2 for several times.
Finally, the reaction occurred at 40 °C and 0.1 MPa in H2 with vig-
orous stirring at 1000 rpm. A small amount of liquid product
(around 1 mL) was withdrawn from the reactor in a certain interval

 Yang, Lei
Yang, Lei
 Yu, Shiyi
Yu, Shiyi
 Peng, Chong
Peng, Chong
 Fang, Xiangchen
Fang, Xiangchen
 Cheng, Zhenmin
Cheng, Zhenmin
 Zhou, Zhiming
Zhou, Zhiming