Organic Letters
Letter
Scheme 2. Reaction under a 13CO Atmosphere
equilibrium between C and D. Thus, the reaction of 1o gave a
nearly 1:1 mixture of 2-fluoroflorenone (2o−a) and 3-
fluoroflorenone (2o−b). The formation of the unusual product
2
o−b suggested that rhodafluorene D′ would be formed, which
would undergo C−Rh bond cleavage by protonolysis to give
16
C′. The free rotation and recyclization at C−H would form
rhodafluorene D″, which is a key species leading to 20-b.
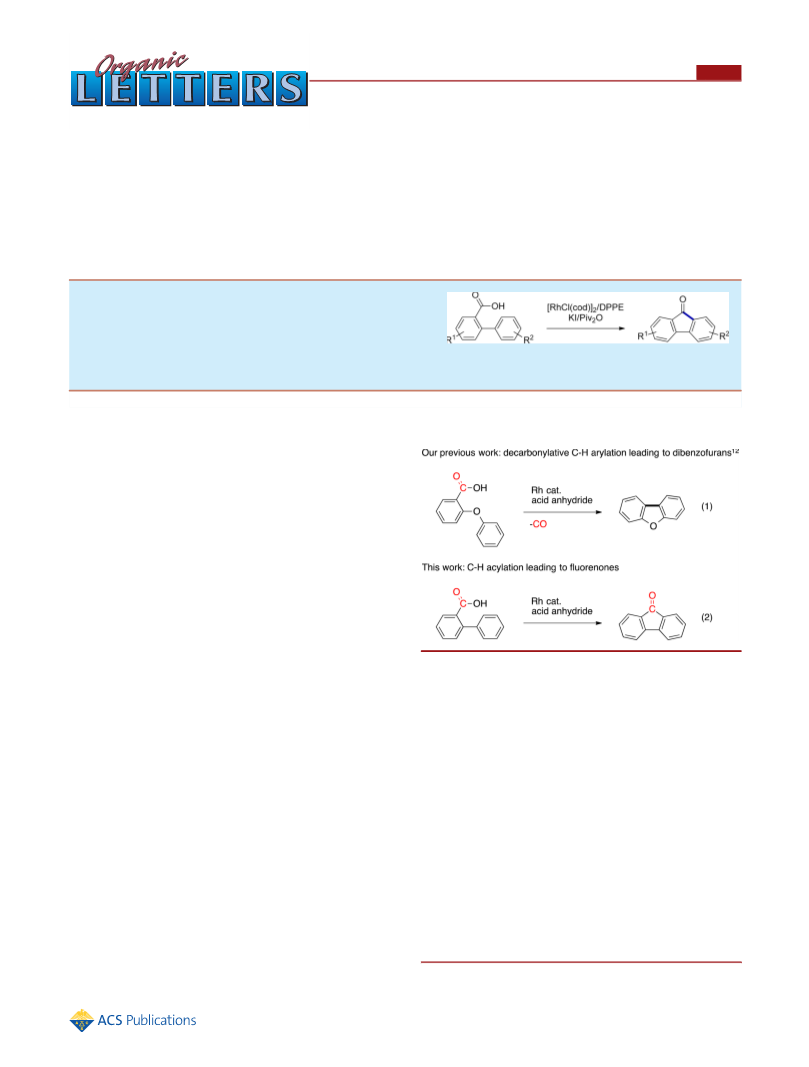
In summary, we developed the first transition-metal-catalyzed
approach to the synthesis of fluorenones by intramolecular
acylation of biarylcarboxylic acids. [RhCl(cod)] /DPPE gave
2
the most effective transformation. Microwave irradiation
shortened the reaction time significantly. Mechanistic studies
suggested an equilibrium for rhodaphenanthrenone and
rhodafluorenes. Further applications of these catalytic acylation
methods are currently underway in our laboratories.
Scheme 3. Proposed Reaction Mechanism
ASSOCIATED CONTENT
Supporting Information
■
*
S
The experimental procedure and compound characterization.
AUTHOR INFORMATION
■
*
*
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
This work was supported by a Grant-in-Aid for Scientific
Research from the MEXT and the JSPS.
■
REFERENCES
■
With two positional isomers, 2o−a and 2o−b, from 2-fluoro
substituted substrate 1o (Scheme 4), we believe there was an
(
1) (a) Greenlee, M. L.; Laub, J. B.; Rouen, G. P.; DiNinno, F.;
Hammond, M. L.; Huber, J. L.; Sundelof, J. G.; Hammond, G. G.
Bioorg. Med. Chem. Lett. 1999, 9, 3225. (b) Perry, P. J.; Read, M. A.;
Davies, R. T.; Gowan, S. M.; Reszka, A. P.; Wood, A. A.; Kelland, L. R.;
Neidle, S. J. Med. Chem. 1999, 42, 2679. (c) Tierney, M. T.; Grinstaff,
M. W. J. Org. Chem. 2000, 65, 5355.
Scheme 4. Reaction of 2-Fluoro-6-Phenyl Benzoic Acid (1o)
(
2) (a) Yang, G.; Zhang, Q.; Miao, H.; Tong, X.; Xu, J. Org. Lett.
005, 7, 263. (b) Catino, A. J.; Nichols, J. M.; Choi, H.; Gottipamula,
S.; Doyle, M. P. Org. Lett. 2005, 7, 5167.
3) (a) Shi, Z.; Glorius, F. Chem. Sci. 2013, 4, 829. (b) Wertz, S.;
2
(
Leifert, D.; Studer, A. Org. Lett. 2013, 15, 928. (c) Seo, S.; Slater, M.;
Greaney, M. F. Org. Lett. 2012, 14, 2650. (d) Lockner, J. W.; Dixon, D.
D.; Risgaard, R.; Baran, P. S. Org. Lett. 2011, 13, 5628.
(4) (a) Ames, D. E.; Opalko, A. Tetrahedron 1984, 40, 1919.
(b) Haggam, R. A. Tetrahedron 2013, 69, 6488. (c) Gandeepan, P.;
Hung, C.-H.; Cheng, C.-H. Chem. Commun. 2012, 48, 9379. (d) Li,
H.; Zhu, R.-Y.; Shi, W.-J.; He, K.-H.; Shi, Z.-J. Org. Lett. 2012, 14,
4
850. (e) Zhang, Z.; Kodumuru, V.; Sviridov, S.; Liu, S.; Chafeev, M.;
Chowdhury, S.; Chakka, N.; Sun, J.; Gauthier, S. J.; Mattice, M.;
Ratkay, L. G.; Kwan, R.; Thompson, J.; Cutts, A. B.; Fu, J.; Kamboj, R.;
Goldberg, Y. P.; Cadieux, J. A. Bioorg. Med. Chem. Lett. 2012, 22, 5108.
(
5) Wan, J.-C.; Huang, J.-M.; Jhan, Y.-H.; Hsieh, J.-C. Org. Lett. 2013,
5, 2742.
6) (a) Blum, J.; Lipshes, Z. J. Org. Chem. 1969, 34, 3076. (b) Blum,
J.; Milstein, D.; Sasson, Y. J. Org. Chem. 1970, 35, 3233.
7) (a) Campo, M. A.; Larock, R. C. Org. Lett. 2000, 2, 3675.
b) Pletnev, A. A.; Larock, R. C. J. Org. Chem. 2002, 67, 9428.
8) (a) Wade, L. G., Jr.; Acker, K. J.; Earl, R. A.; Osteryoung, R. A. J.
1
(
(
(
(
Org. Chem. 1979, 44, 3724. (b) Ladd, D. L.; Weinstock, J.; Wise, M.;
Gessner, G. W.; Sawyer, J. L.; Flaim, K. E. J. Med. Chem. 1986, 29,
C
dx.doi.org/10.1021/ol5012407 | Org. Lett. XXXX, XXX, XXX−XXX

 Fukuyama, Takahide
Fukuyama, Takahide
 Maetani, Shinji
Maetani, Shinji
 Miyagawa, Kazusa
Miyagawa, Kazusa
 Ryu, Ilhyong
Ryu, Ilhyong