R.J. Kalbasi, N. Mosaddegh / Catalysis Communications 12 (2011) 1231–1237
1235
Table 4 (continued)
Entry
Substrate
Product
Time
(h)
Yield
(%)
Mp (°C)
b
Found
Reported ref
N(CH3)2
172–174
168–170 [16]
7
3
98
(H3C)2N
CHO
O
CN
NH2
O
a
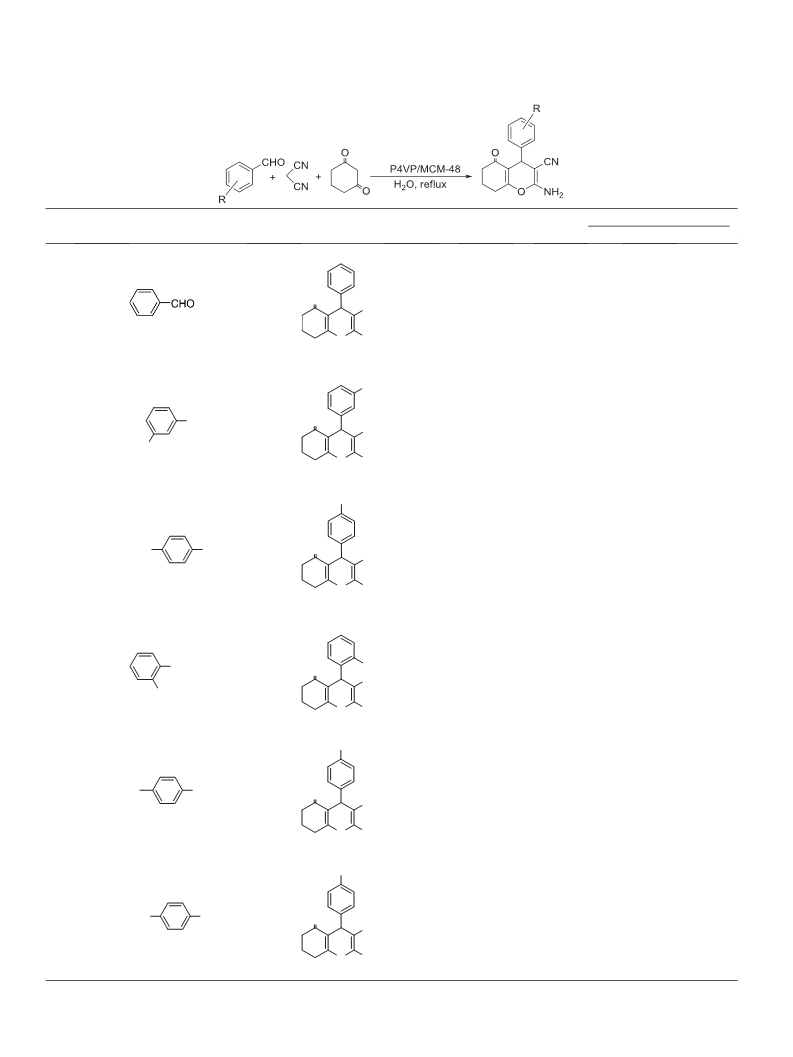
Reaction conditions: P4VP/MCM-48 (0.14 g), aromatic aldehydes (1 mmol), malononitrile (1 mmol), 1,3-cyclohexadione (1 mmol), H2O (5 mL), 95 °C.
Isolated yield.
b
of a well-ordered cubic structure are present in both cases indicating
that polymerization has no adverse effects on the long-range
structure.
TGA curves of bulk P4VP and P4VP/MCM-48 composite are
presented in Fig. 5 under N2 atmosphere. The weight loss (around
62%, w/w) of P4VP begins at 210 °C because of thermo degradation of
P4VP polymer chains, and the degradation ends at 500 °C (Fig. 5a).
Whereas for P4VP/MCM-48, the weight loss (around 14%, w/w)
begins at 360 °C that is related to the degradation of the polymer
chains and the degradation ends at 460 °C (Fig. 5b). Obviously, the
hybrid P4VP/MCM-48 shows higher thermal stability and a slower
degradation rate than P4VP. Therefore, after hybridization, the
thermal stability is enhanced greatly, which is very important for
the catalyst application.
Fig. 2 presents the FTIR spectra of MCM-48 (a), similarly prepared
bulk P4VP (b), and P4VP/MCM-48 composite (c). Similar to mesoporous
silica MCM-48 (Fig. 2a), P4VP/MCM-48 composite shows the typical
vibrations of asymmetric and symmetric stretching, as well as the
rocking of Si–O–Si at around 1083, 821, and 461 cm−1 (Fig. 2c). The
existence of P4VP in the composite is evidenced by the appearance of
typical P4VP vibrations on the FTIR spectrum. In the FT-IR spectrum of
P4VP/MCM-48 (Fig. 2c), the new bands at 1606, 1559 and 1422 cm−1
are the characteristic absorptions of pyridine ring. Among them, the
band which appeared at around 1606 cm−1 (this band in the spectrum
of P4VP is around 1595) is the stretching vibration absorption of C–N
bond, and the bands at 1559 and 1422 cm−1 are attributed to the
stretching vibration absorption of aromatic C–C bonds. Moreover, the
presence of peaks at around 2800–3100 cm−1 corresponds to the
aromatic and aliphatic C–H stretching in P4VP/MCM-48. These are in
accordance with the spectrum of P4VP (Fig. 2b). The appearance of the
above bands shows that P4VP has been attached to the surface of MCM-
48 and the P4VP/MCM-48 has been obtained.
The BET specific surface areas and the pore size, the host MCM-48
and P4VP/MCM-48 had already been calculated using the Brunauer–
Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods,
respectively (Table 1). Both samples show isotherms of type IV,
according to the IUPAC nomenclature, which are the typical
characteristics of mesoporous materials. The corresponding BJH
pore size distribution curves for the MCM-48 and P4VP/MCM-48
materials are shown in Fig. 3. Compared to these two pore size
distribution curves, it can be seen clearly that the pore size
distribution is narrowed after the insertion of polymer, which
indicates that the polymer is distributed uniformly on the surface of
MCM-48. It is known that calcined MCM-48 has a high BET surface
area (1673 m2 g−1), a large pore volume (0.89 cm3 g−1) and pore size
(2.1 nm), indicative of its potential application as a host in organic
materials. After hybridization with P4VP through in situ polymeriza-
tion, P4VP/MCM-48 exhibits a smaller specific surface area, pore size
and pore volume in comparison to those of pure MCM-48, which
might be due to the presence of polymer on the surface of MCM-48.
Although these textural properties are smaller than those found for
mesoporous silica MCM-48, P4VP/MCM-48 still has a mesoporous
form. So, it is suitable to act as a basic catalyst.
3.2. Catalytic activity
The effect of different parameters on the three-component
reaction of benzaldehyde and malononitrile with 1,3-cyclohexadione
over P4VP/MCM-48 as catalyst was investigated, and the results are as
follows:
The effect of reaction temperature and catalyst amount on the
reaction yield was studied and the values are shown in Table 2, for the
reaction of benzaldehyde (1 mmol), malononitrile (1 mmol), 1,3-
cyclohexadione (1 mmol) and catalyst (P4VP/MCM-48) in H2O
(5 mL). When the catalyst amount increases from 0.10 to 0.14 g the
yield increases from 70% to 98% in 95 °C. With excess amount of
catalyst, the yield obviously increases because of the availability of
more basic sites. As the amount of catalyst exceeds 0.14 g, the yield
still increases but not so obviously as that below 0.14 g. So, the 0.14 g
of the catalyst was chosen. In addition, it should be mentioned that in
the absence of the catalyst, the product was not observed after 6 h.
The yield increases from 60% to 98% by increasing the temperature
from 25 °C to 95 °C. Also, by increasing the temperature, the duration of
reaction decreases from 6 h to 2 h. Thus, 95 °C (reflux) was considered
as an optimum reaction temperature (Table 2).
The effect of MCM-48 on the catalytic activity of P4VP/MCM-48
composite was investigated for three-component reactions of benz-
aldehyde and malononitrile with 1,3-cyclohexadione, and the results
are shown in Table 3. One of the greatest advantages of this catalyst is
that it is used easily due to its powdery structure. P4VP is adhesive,
and this characteristic makes it hard to separate it from the vessel; but
after mixing with MCM-48 and making P4VP/MCM-48 composite, it
becomes powdery which is easy to use and recycle. Also, comparative
reaction by using P4VP and P4VP/MCM-48 shows that P4VP/MCM-48
is more efficient, by completing the reaction in a short time. On the
other hand, the yield is decreased and the reaction is carried out in a
long period of time using P4VP. On the basis of the turnover frequency
(TOF) defined in Table 3, it is obvious that composite P4VP/MCM-48
system is advantageous. In fact, the activity of P4VP is much lower
The morphologies of the MCM-48 host and P4VP/MCM-48 are
shown in Fig. 4. MCM-48 host (Fig. 4a) and P4VP/MCM-48 (Fig. 4b)
are agglomerations of small irregular particles. Virtually no difference
in particle surface morphology between the MCM-48 host and the
P4VP/MCM-48 composite is observed.

 Kalbasi, Roozbeh Javad
Kalbasi, Roozbeh Javad
 Mosaddegh, Neda
Mosaddegh, Neda