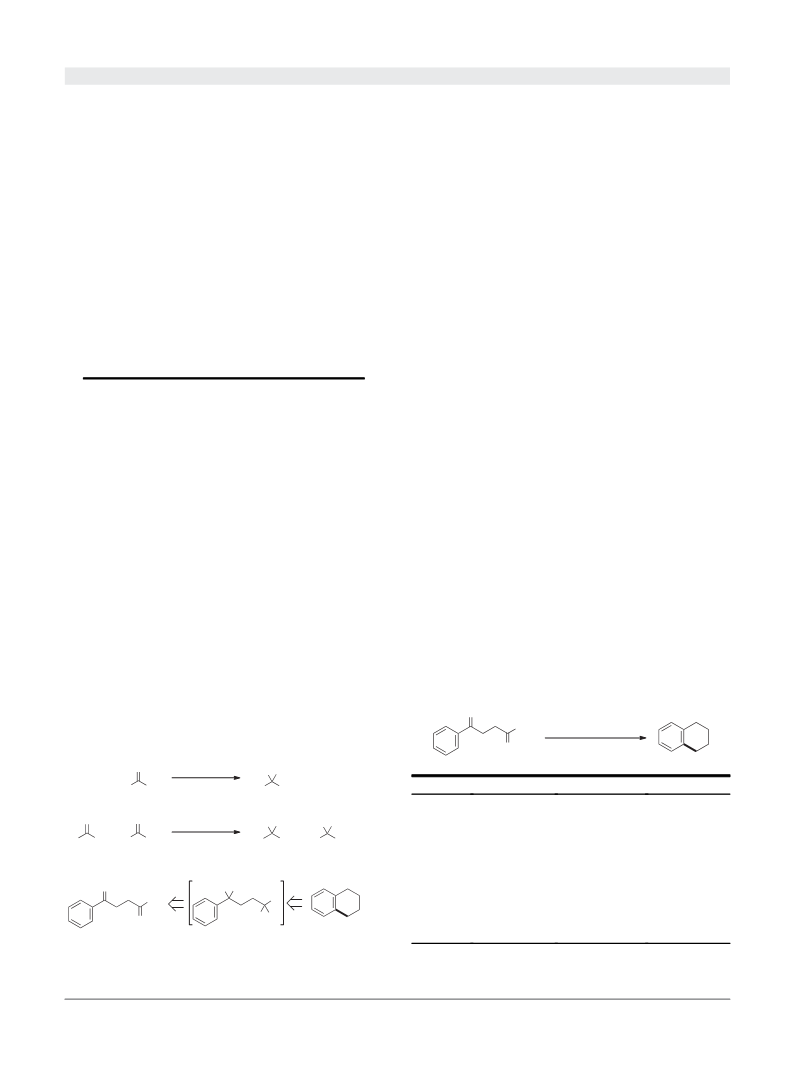Table 2. Preparation of tetralins with a variety of functional groupsa
Table 3. Control experiments for the intramolecular cyclization of
presumed intermediate 14
In(OAc)3 (1 mol %)
O
Me PhSiH (6.5 equiv)
In(OAc)3
2
OH
I2 (1 equiv)
Me PhSiH
I2
2
R
I
R
O
CHCl , 60 ˚C, 5 h
3
CHCl , 60 ˚C, 5 h
3
1
4
1
Me
Me
Me
Me
In(OAc)3
mol %
Me2PhSiH
/equiv
I2
Yield
Entry
+
a
/
/equiv
/%
Me
1
2
3
4
5
1
1
1
1
0
0
1
1
1
1
0
0
0.01
1
0
0
trace
93
3
: 75%
3
4
2
: 82%
2% (3 : 4 = 1 : 2)b
7
1
29
Me
F
Cl
aGC yield.
4
: 70%
5: 65% (7%)c,d
6: 60% (25%)c,d
caused the catalytic hydrosilylation and subsequent intramolec-
ular cyclization to occur smoothly, producing the corresponding
tetralin derivative 10 in an 80% yield. As a facile approach to a
tricyclic carbon skeleton, when the reaction was performed with
the substrates having either a naphthalene or a tetralin ring, the
expected tricyclic products, 1,2,3,4-tetrahydrophenanthrene (11)
and a mixture of 1,2,3,4,5,6,7,8-octahydroanthracene (12) and
Br
MeO
7
: 80% (7%)c,d
8: 85%
9: 0%
O
0: 80%
1
,2,3,4,5,6,7,8-octahydrophenanthrene (13) were obtained in 87
1
1
1: 87%
and 86% yields, respectively.
To clarify the reaction path series, several control experi-
ments with the presumed intermediate were performed, and the
results are summarized in Table 3. Initially, when the intra-
molecular cyclization of alkyl iodide 14 was conducted with
either In(OAc)3 or a combination of In(OAc)3 and the hydro-
silane, both cases did not undertake the expected cyclization to
recover the starting material (Entries 1 and 2). Then, the addition
of a small amount of I2 gave a trace amount of tetralin (Entry 3).
Thus, when 1 equiv of I2 was added to the reaction system
+
1
2
13
8
6% (12 : 13 = 3 : 1)b
a
b
c
Isolated yield. The ratio was determined by NMR. The yield of
the corresponding iodinated product is in parenthesis. Reaction
time: 24 h.
d
consisting of In(OAc) and the hydrosilane, the intramolecular
3
cyclization proceeded smoothly to form tetralin in a 93% yield
(Entry 4). Although the preparation of a tetralin skeleton via
intramolecular FriedelCrafts alkylation of 4-phenyliodobutane
To generalize this cyclization, several 3-benzoylpropionic
acid derivatives were treated with our optimal conditions
including 1 mol % of In(OAc) , Me PhSiH (Si-H: 6.5 equiv),
1
2
has been difficult, this reducing system enabled us to produce
the similar product in a high yield. On the other hand, when a
similar reaction was conducted without In(OAc)3, the GC yield
of tetralin was drastically decreased to 29% (Entry 5). There-
fore, these results strongly supported that the indium catalyst
was essential to promote the intramolecular cyclization series
effectively, and implied that some sort of complex composed of
3
2
11
and I2 (1 equiv), and the results are summarized in Table 2.
With no relationship to the location of a substituent, substrates
with an electron-donating group, such as a methyl group, gave
the expected tetralin derivatives 24 in good yields. Among
them, the meta-substituted substrate, with two regioisomers 3
and 4, were obtained, wherein the product 4 was prefentially
substituted at the para position rather than at the ortho position.
For those substrates with an electron-withdrawing group, such
as halogens and a phenyl group, both reduction and cyclization
proceeded smoothly, producing tetralin derivatives 58 in good
yields. However, in the cases of the substrate with a halogen,
a small amount of the linear alkyl iodides 5¤7¤, which would
be anticipated as a reaction intermediate, were isolated as
side-products. With these results, it seemed that the decrease in
the nucleophilicity of the benzene ring with an electron-
withdrawing group hindered the final intramolecular cyclization.
Unfortunately, to the substrate with a methoxy group, this
reducing system did not undertake the desired reaction to
recover the starting material, though we have no clear reasons at
this step. On the other hand, the substrate with a phenoxy group
1
3
indium acetate and the silyl iodide, which would be generated
1
4
from the hydrosilane and I2, might function as a catalyst to
activate the alkyl iodide intermediate.
On the basis of the results of control experiments, a
plausible reaction pathway for the reduction and the intra-
molecular cyclization of 3-benzoylpropionic acids is shown in
Scheme 2. This reaction pathway is proposed based on our
5
previous results. Initially, a carboxylic acid moiety was reacted
with a hydrosilane to produce silyl ester A with a liberation of
H . Subsequently, the ketone group, which was activated by
2
an indium compound, of silyl ester A was reduced with two
equivalents of the hydrosilane to produce silyl ester B. Activated
silyl ester B was further reduced by two more equivalents of
the hydrosilane to form silyl ether C. Even without an indium
© 2015 The Chemical Society of Japan

 Sakai, Norio
Sakai, Norio
 Kobayashi, Taichi
Kobayashi, Taichi
 Ogiwara, Yohei
Ogiwara, Yohei