122
V.I. Sobolev et al. / Journal of Fluorine Chemistry 192 (2016) 120–123
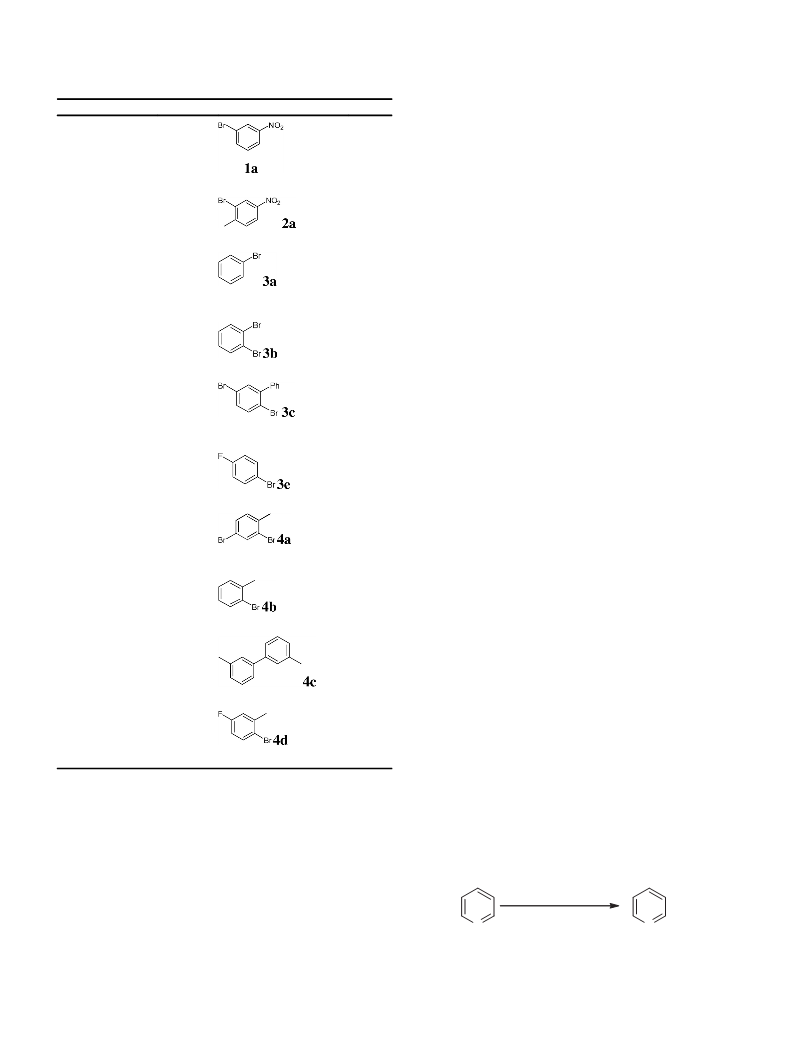
Table 2
Reactions of Py
d
: 122.1 (C-3),122.8 (C-6),126.7 (C-2),130.6 (C-5),137.6 (C-4),148.7
ꢀ
BrF3(BaF2) with arenes 1–4.a
Product
(C-1).
2-Bromo-4-nitrotoluene (2a). Following general protocol using
Substrate
Yield%
1
2
3
4
1a
2a
3a
4b
23b
19b
89b
87c
2 and Ba(BrF4)2 the 2a was obtained as yellow crystals (yield 87%),
mp 76 ꢂC. RT: 15.04 min. GC–MS, 70 eV, m/z (rel. int): 30 (46), 39
(28), 63 (63), 78 (26), 90 (100), 169 (27), 185 (18), 199 (1), 215 (58).
1H NMR (300 MHz, CDCl3)
d
: 2.5 (3H, s), 7.4 (1H, d, J = 8.4 Hz), 8.05
(1H, dd, J = 8.4, 2.0 Hz), 8.37 (1H, d, J = 2.0 Hz). 13
NMR (75 MHz,
CDCl3) : 23.2 (CH3),122.2 (C-5),124.9 (C-2),127.4 (C-3),131.1 (C-6),
145.8 (C-1), 146.8 (C-4).
a
E
Representative procedure: arenes (4 mL), 6-BaF2 (1.92 mmol), rt.
b
c
Isolated yields.
GC–MS.
d
Bromobenzene (3a). Yield 16%, GC data. RT: 5,18 min. GC–MS,
70 eV, m/z (rel. int): 12 (1), 26 (4), 38 (16), 51 (49), 77 (100), 156
(75).
reaction products and fluoroorganic products (except from minor
products 3e and 4d).
1,2-Dibromobenzene (3b). Yield 34%, GC data. RT: 10.71 min GC–
MS, 70 eV, m/z (rel. int): 26 (1), 50 (19), 75(28), 118 (12), 155 (38),
236 (100).
In summary we have shown for the first time that Ba(BrF4)2 and
its complex with pyridine diluted by BaF2 are convenient reagents
and they can be considered as safe forms of BrF3. These compounds
have strong and selective electrophilic bromination abilities
towards not only deactivated nitrobenzenes but also benzene
and toluene.
2,5-dibromo-1,10-biphenyl (3c). Yield 17%, GC data. RT: 16.63 min.
GC–MS, 70 eV, m/z (rel. int): 50 (19), 76 (37), 98 (8), 126 (11), 152
(100), 312 (87).
Biphenyl (3d). Yield 12%, GC data. RT: 13.72 min. GC–MS, 70 eV,
m/z (rel. int): 27 (1), 39 (5), 51 (16), 63 (11), 76 (22), 89 (4), 102 (7),
115 (7), 128 (8), 139 (4), 154 (100).
1-Bromo-4-fluorobenzene (3e). Yield 8%, GC data. RT: 4.95 min.
GC–MS, 70 eV, m/z (rel. int): 25 (2), 37 (4), 50 (12), 62 (5), 75 (36),
95 (100), 174 (96).
2,4-Dibromo-1-methylbenzene (4a). Yield 24%, GC data. RT:
12.65 min. GC–MS, 70 eV, m/z (rel. int): 44 (11), 63 (20), 90 (41),169
(59), 250 (100).
1-Bromo-2-methylbenzene (4b). Yield 14%, GC data. RT: 7.77 min.
GC–MS, 70 eV, m/z (rel. int): 27 (2), 39 (17), 51 (11), 65 (21), 77 (1),
91 (100), 170 (40).
3. Experimental section
All chemicals were analytical grade and used as purchased
without further purification. TFBs were synthesized using previ-
ously described methods [7–9] directly before use. Pyridine was of
analytical grade and was additionally dried using metal sodium.
Freon R 113 was of analytical grade and was additionally dried
using metal sodium. All experiments with pyridine and its’ derive
compounds were performed in dry box installation with less than
5 ppm H2O concentration. 1H, 13C NMR spectra were recorded on a
Bruker AC 300 spectrometer with tetramethylsilane (TMS) as the
internal standard, solvents CDCl3. Multiplicities of signals are
described as follows: s = singlet, d = doublet, dd = doublet of
doublet, t = triplet, m = multiplet. Melting points were determined
on melting point system MP50. The identification of obtained
compounds was performed by comparing the analytical and
physical-chemical characteristics as the authentic sample synthe-
sized by known methods. GC–MS analysis was performed on
Agilent 7890A (Agilent Technologies, USA) combined with a mass
3,30-Dimethyl-1,10-biphenyl (4c). Yield 17%, GC data. RT:
16.63 min. GC–MS, 70 eV, m/z (rel. int): 25 (2), 39 (5), 51 (6), 63
(8), 76 (8), 89 (6), 115 (8), 128(7), 152 (10), 167 (37), 182 (100).
1-Bromo-4-fluoro-2-methylbenzene (4d). Yield 9%, GC data. RT:
7.53 min. GC–MS, 70 eV, m/z (rel. int): 15 (2), 39 (14), 57 (26), 83
(41), 109 (100), 190 (51).
3.2. Reactions of TFBs with pyridine 5
Pyridine (4 mmol) was dissolved in Freon R113 (4.1 mL) and
cooled to ꢁ25 ꢂC. Ba(BrF4)2 (2 mmol) was added to the pyridine
solution with vigorous stirring and stirred for 5 h to prevent solid
product agglomeration. The obtained white solid product 6 in a
mixture with BaF2 was decanted from Freon 113 and used for
further experiments without additional purification.
detector Agilent 5975C,
a
carrier gas
ꢁ
helium. Products
identification was performed by mass spectra and retention times
(RT) in comparison with authentic samples. Reaction progress was
monitored by TLC with UV detection using Silufol UV-254.
3.1. Reactions of TFBs with arenes 1–4
3.3. Reactions of 6-BaF2 with arenes 1–4
TFBs were synthesized using previously described methods [7–
9] directly before use. Corresponding arene (4 mmol) was
dissolved in Freon R 113 (4.1 mL), and cooled to ꢁ25 ꢂC. The
corresponding TFB (2 mmol) was slowly added to the arene
solution with vigorous stirring and the cooling bath was removed.
The reaction mass was stirred at 45 ꢂC for 5 h. After reaction
completion the reaction mass was treated by H2O and filtered to
remove the metal fluoride precipitate. The liquid phase was treated
by 10% aqueous NaNO2 in order to remove traces of bromine and
with 30% aqueous CaCl2 to remove the Fꢁ anion. Freon R 113 was
evaporated from the organic phase and the obtained product
purified by silica gel flash chromatography, eluent hexane:EtOAc.
3-Bromonitrobenzene (1a). Following general protocol using 1
and Ba(BrF4)2 the 1a was obtained as yellow crystals (yield 84%),
mp 52 ꢂC. RT: 12.81 min. GC–MS, 70 eV, m/z (rel. int): 30 (26), 50
(55), 75 (86), 143 (21), 155 (100), 201 (92). 1H NMR (300 MHz,
6-BaF2 (1.92 mmol) was added to corresponding arene 1–4
(5 mL) at room temperature and stirred for 5 h. The reaction
mixture was filtered from solid BaF2, liquid products were analysed
by GC–MS and bromoarenes 1a, 2a and 3a were isolated by silica
gel flash chromatography, eluent hexane:EtOAc ꢁ 3:1 (v/v).
3-Bromonitrobenzene (1a). Following general protocol using 1
and 6-BaF2 the 1a was obtained as yellow crystals (yield 23%). RT:
12.81 min. GC–MS, 70 eV, m/z (rel. int): 30 (26), 50 (55), 75 (86),143
(21), 155 (100), 201 (92). 1H NMR (300 MHz, CDCl3)
J = 8.7, 1.5 Hz), 7.83 (1H, d, J = 8,7 Hz), 8.15 (1H, dd, J = 8.7, 1.5 Hz),
8.36 (1H, d, J = 1.5 Hz). 13E NMR (75 MHz, CDCl3)
: 122.1 (C-3),
122.8 (C-6), 126.7 (C-2), 130.6 (C-5), 137.6 (C-4), 148.7 (C-1).
2-Bromo-4-nitrotoluene (2a). Following general protocol using 2
and 6-BaF2 the 2a was obtained as yellow crystals (yield 19%). RT:
15.04 min. GC–MS, 70 eV, m/z (rel. int): 30 (46), 39 (28), 63 (63), 78
(26), 90 (100), 169 (27), 185 (18), 199 (1), 215 (58). 1H NMR
d: 7.44 (1H, dd,
d
CDCl3)
d
: 7.44 (1H, dd, J = 8.7,1.5 Hz), 7.83 (1H, d, J = 8,7 Hz), 8.15 (1H,
NMR (75 MHz, CDCl3)
dd, J = 8.7, 1.5 Hz), 8.36 (1H, d, J = 1.5 Hz). 13
E
(300 MHz, CDCl3) d: 2.5 (3H, s), 7.4 (1H, d, J = 8.4 Hz), 8.05 (1H, dd,

 Sobolev, Vasily I.
Sobolev, Vasily I.
 Filimonov, Victor D.
Filimonov, Victor D.
 Ostvald, Roman V.
Ostvald, Roman V.
 Radchenko, Vyacheslav B.
Radchenko, Vyacheslav B.
 Zherin, Ivan I.
Zherin, Ivan I.