Page 5 of 6
ACS Catalysis
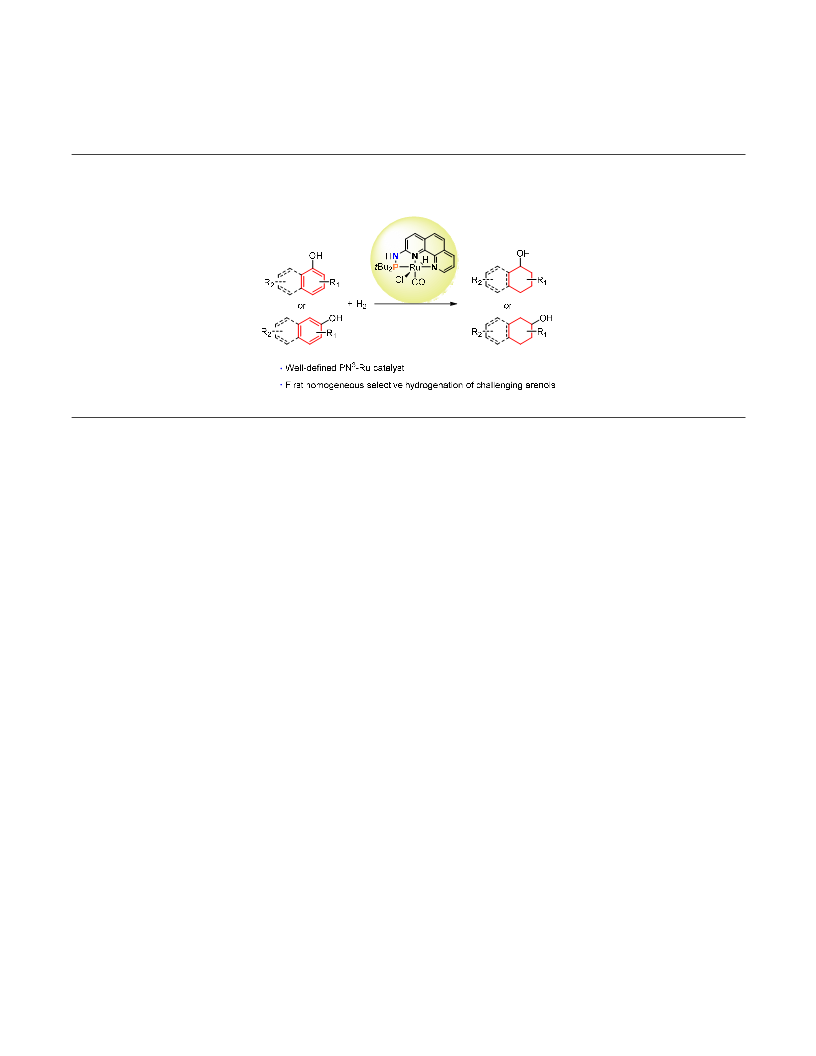
the first partial hydrogenation of naphthols to tetrahy-
Soc. 1948, 70, 4179-4181; (g) Ungnade, H. E.; McLaren, A. D. J.
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
Am. Chem. Soc. 1944, 66, 118-122.
7. Kusumoto, S.; Nozaki, K. Nat. Commun. 2015, 6, 6296.
dronaphthols with a high selectivity for the hydrogena-
tion of the hydroxylated ring to afford the corresponding
products in good to excellent yields. Compared with those
heterogeneous systems, this new, atom economical cata-
lytic protocol exhibits s higher selectivity with a broader
substrate scope. The fact that the methoxyl- and amino-
analogs showed no or little reactivity suggests that a hy-
droxyl group is necessary and the reaction may proceed
via the keto tautomer. Further investigations on the syn-
thetic implications of these efficient homogeneous cata-
lytic systems and the detailed mechanisms of the reac-
8
. Zhou et al. reported a method for hydrogenation of phenols to
alcohols by [Rh(COD)Cl] , however, mechanistic studies revealed
2
that the actual catalyst in this process may be rhodium
nanoparticles generated from the complex [Rh(COD)Cl] in situ
under aerobic condition. See ref 6b.
9. (a) Oldenhof, S.; van der Vlugt, J. I.; Reek, J. N. H. Catal. Sci.
Technol. 2016, 6, 404-408; (b) Gunanathan, C.; Milstein, D.
Chem. Rev. 2014, 114, 12024-12087; (c) Ikariya, T.; Kayaki, Y.
Pure Appl. Chem. 2014, 86, 933-943; (d) Otsuka, T.; Ishii, A.;
Dub, P. A.; Ikariya, T. J. Am. Chem. Soc. 2013, 135, 9600-9603;
(e) Balaraman, E.; Fogler, E.; Milstein, D. Chem. Commun. 2012,
2
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
5
tions are ongoing.
4
1
8, 1111-1113; (f) Dub, P. A.; Ikariya, T. ACS Catal. 2012, 2,
718-1741; (g) Balaraman, E.; Gunanathan, C.; Zhang, J.;
ASSOCIATED CONTENT
Supporting Information.
The Supporting Information is available free of charge on the
ACS Publications website at DOI:
Experimental details and characterization data Crystallo-
graphic data
Shimon, L. J. W.; Milstein, D. Nat. Chem. 2011, 3, 609-614; (h)
Ito, M.; Ootsuka, T.; Watari, R.; Shiibashi, A.; Himizu, A.;
Ikariya, T. J. Am. Chem. Soc. 2011, 133, 4240-4242.
10. (a) de Boer, S. Y.; Korstanje, T. J.; La Rooij, S. R.; Kox, R.;
Reek, J. N. H.; van der Vlugt, J. I. Organometallics 2017, 36,
1
541-1549. (b) Li, H.; Zheng, B.; Huang, K.-W. Coord. Chem.
Rev. 2015, 293–294, 116-138; (c) Qu, S.; Dang, Y.; Song, C.;
Wen, M.; Huang, K.-W.; Wang, Z.-X. J. Am. Chem. Soc. 2014,
AUTHOR INFORMATION
Corresponding Author
1
36, 4974-4991; (d) Zhao, B.; Han, Z.; Ding, K. Angew. Chem.
Int. Ed. 2013, 52, 4744-4788; (e) Nielsen, M.; Alberico, E.;
Baumann, W.; Drexler, H.-J.; Junge, H.; Gladiali, S.; Beller, M.
Nature 2013, 495, 85-89; (f) Bullock, R. M. In The Handbook of
Homogeneous Hydrogenation; De Vries, J. G.; Elsevier, C. J.;
WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, 2008; p
*
Notes
The authors declare no competing financial interest.
1
53; (g) Sandoval, C. A.; Ohkuma, T.; Muñiz, K.; Noyori, R. J.
Am. Chem. Soc. 2003, 125, 13490-13503; (h) Blum, Y.; Czarkie,
D.; Rahamim, Y.; Shvo, Y. Organometallics 1985, 4, 1459-1461.
11. (a) Bruneau-Voisine, A.; Wang, D.; Roisnel, T.; Darcel, C.;
Sortais, J.-B. Catal. Commun. 2017, 92, 1-4; (b) Pan, Y.; Pan, C.-
L.; Zhang, Y.; Li, H.; Min, S.; Guo, X.; Zheng, B.; Chen, H.;
Anders, A.; Lai, Z.; Zheng, J.; Huang, K.-W. Chem. - Asian J.
2016, 11, 1357-1360; (c) Roesler, S.; Obenauf, J.; Kempe, R. J.
Am. Chem. Soc. 2015, 137, 7998-8001; (d) Chen, T.; Li, H.; Qu,
S.; Zheng, B.; He, L.; Lai, Z.; Wang, Z.-X.; Huang, K.-W.
Organometallics 2014, 33, 4152-4155; (e) He, L.-P.; Chen, T.;
Gong, D.; Lai, Z.; Huang, K.-W. Organometallics 2012, 31,
ACKNOWLEDGMENT
Financial support was provided by King Abdullah University
of Science and Technology (KAUST).
REFERENCES
1
. (a) Zhu, S.; Liang, R.; Jiang, H.; Wu, W. Angew. Chem. Int.
Ed. 2012, 51, 10861-10865 and references therein; (b) Shirai, M.;
Rode, C. V.; Mine, E.; Sasaki, A.; Sato, O.; Hiyoshi, N. Catal.
Today 2006, 115, 248-253; (c) Capriati, V.; Florio, S.; Luisi, R.;
Perna, F. M.; Salomone, A.; Gasparrini, F. Org. Lett. 2005, 7,
5
1
1
208-5211.
2. See SI for full details.
3. A trace amount of 3,4-dihydro-1(2H)-naphthalenone was
4
2
2
895-4898.
. (a) Xu, T.; Zhang, Q.; Cen, J.; Xiang, Y.; Li, X. Appl. Surf. Sci.
015, 324, 634-639; (b) Tan, S.-L.; Liu, G.-B.; Gao, X.;
observed: (<1%).
14. Rappoport, Z. The chemistry of phenols; Wiley: Hoboken, NJ,
Thiemann, T. J. Chem. Res. 2009, 5-7 and references therein.
. (a) De Vries, J. G.; Elsevier, C. J. The Handbook of
3
2
1
003.
Homogeneous Hydrogenation; WILEY-VCH Verlag GmbH &
Co. KGaA: Weinheim, 2008; (b) Burke, S. D.; Danheiser, R. L.
Handbook of Reagents for Organic Synthesis: Oxidizing and
Reducing Agents; John Wiley & Son Ltd: New Jersey, 1999; (c)
Rylander, P. N. Hydrogenation methods; Academic Press Inc:
London, 1985.
5. The presence of Hg affected neither the conversions nor
yields of the reactions for complex 2 and 3 (Table S2). These
results suggest that the 2- or 3-catalyzed hydrogenation is mainly
a homogeneous process. See SI for details.
4
2
. (a) Qi, S.-C.; Wei, X.-Y.; Zong, Z.-M.; Wang, Y.-K. RSC Adv.
013, 3, 14219-14232; (b) Gual, A.; Godard, C.; Castillon, S.;
Claver, C. Dalton Trans. 2010, 39, 11499-11512.
5. Li, C. L.; Xu, Z. R.; Cao, Z. A.; Gates, B. C.; Petrakis, L.
AIChE J. 1985, 31, 170-174.
6
. (a) Cui, X.; Surkus, A.-E.; Junge, K.; Topf, C.; Radnik, J.;
Kreyenschulte, C.; Beller, M. Nat. Commun. 2016, 7, 11326; (b)
Wang, D.-W.; Lu, S.-M.; Zhou, Y.-G. Tetrahedron Lett. 2009, 50,
1
282-1285; (c) Mine, E.; Haryu, E.; Arai, K.; Sato, T.; Sato, O.;
Sasaki, A.; Rode, C. V.; Shirai, M. Chem. Lett. 2005, 34, 782-
83; (d) Shao, J.; Song, C. Catal. Today 2001, 65, 59-67; (e)
7
Curtis, C. W.; Pellegrino, J. L. Energy Fuels 1989, 3, 160-168; (f)
Dauben, H. J., Jr.; McKusick, B. C.; Mueller, G. P. J. Am. Chem.
ACS Paragon Plus Environment

 Li, Huaifeng
Li, Huaifeng
 Wang, Yuan
Wang, Yuan
 Lai, Zhiping
Lai, Zhiping
 Huang, Kuo-Wei
Huang, Kuo-Wei