1790
S. Alcaro et al. / Bioorg. Med. Chem. 12 (2004) 1781–1791
5
.9. Viruses
combinatorial libraries and has proven to be an effective
pre-screen.
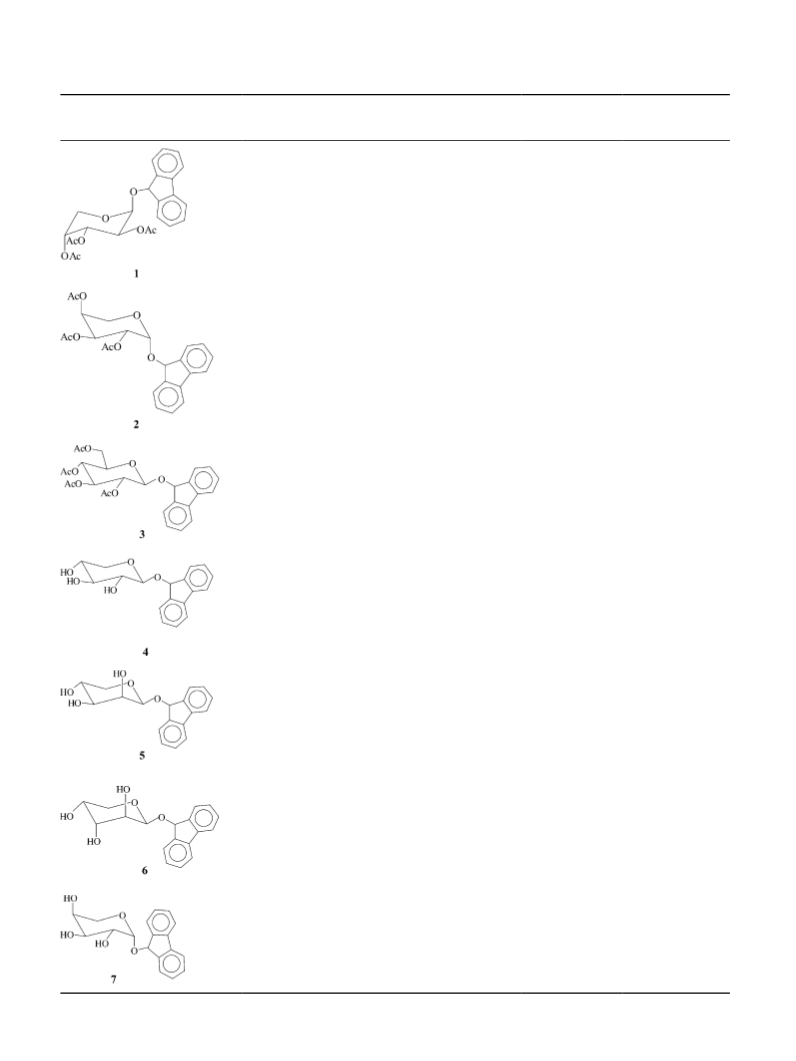
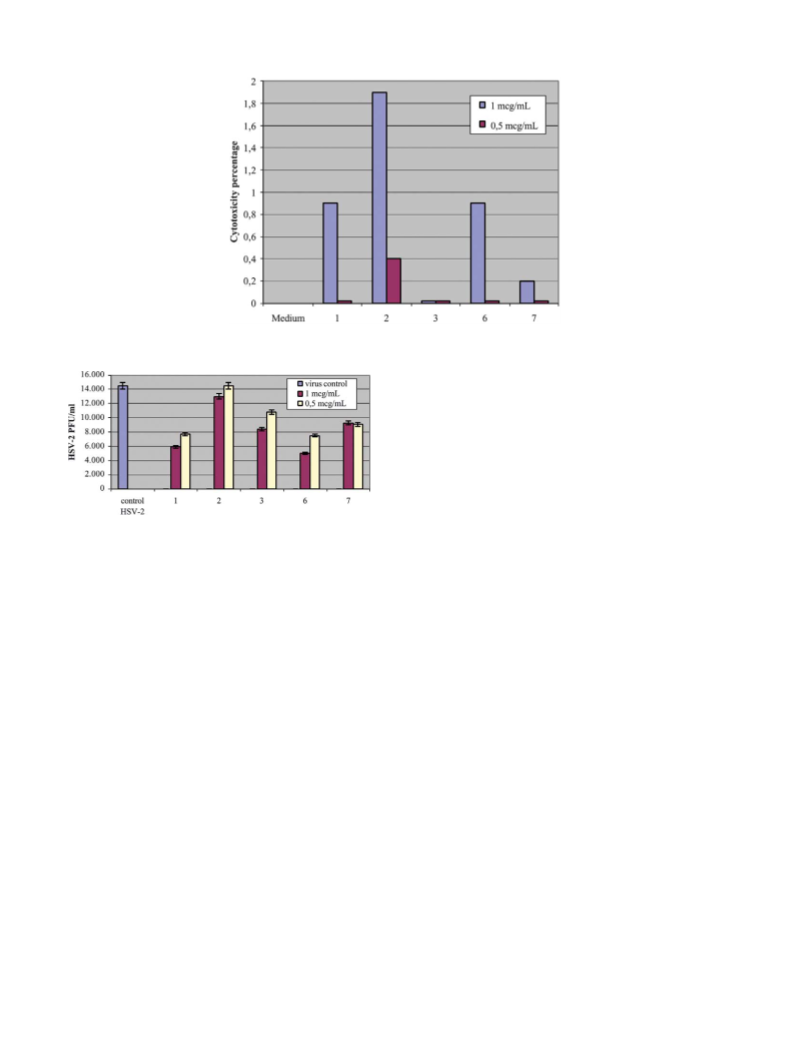
Herpes simplex virus type-2 (HSV-2) and Herpes sim-
plex virus type-1 (HSV-1) were prepared on WISH cells.
Virus titer was determined by assaying for PFU/mL on
WISH cells.
In the current protocol, each cell line is inoculated and
preincubated on a microliter plate. Test agents are then
ꢁ
4
added at a single concentration (10 M) and the cul-
ture incubated for 48 h. End point determinations are
made with sulforhodamine B, a protein-binding dye.
Results for each test agent are reported as the percent of
growth of the treated cells when compared to the
untreated control cells. Compounds which reduce the
growth of any one of the cell lines to 32% or less
5
.10. U937 cells culture and differentiation
The human monocytic cell line U937 was a kind gift
from Prof. J.P. Liautard, INSERM, University of
Montpellier II, France.
(
negative numbers indicate cell-kill) are passed for eval-
ꢀ
atmosphere in RPMI 1640 medium supplemented with
The cells were maintained in culture at 37 C in 5% CO
uation in the full panel at 60 cell lines over a 5-log dose
range.
2
5
0 mM 2-mercaptoethanol, 1 mM pyruvate, 1 mM non-
essential aminoacids, 1 mM HEPES, gentamycin (50
mcg/mL) and 10% heat inactivated FCS (all reagents
Acknowledgements
5
from Seromed, Milan, Italy). The cells (3ꢂ10 per well)
were differentiated for 72 h with 100 nM 1a,25-dihy-
droxyvitamin D3 (VD) (Calbiochem, Milan, Italy) plus
This work was financially supported by the Ministero
dell’Universita e della Ricerca Scientifica e Tecnologica
`
1
00 nM all-transretinoic acid (RA) (Sigma, Milan,
(MURST-Rome). We thank Prof. Cristina Nativi for
her helpful suggestions during the work preparation.
Italy) in tissue culture multi-well plates (Sigma, Milan,
Italy). VD and RA were dissolved in absolute ethanol
ꢀ
ꢁ3
and stored at ꢁ80 C at an initial concentration of 10
M. The final concentration of ethanol had no effects on
cell growth and differentiation. After 72 h of incubation
with VD-RA, differentiation was complete, and most
cells were adherent. Differentiation was confirmed by
growth inhibition, morphological changes, increased
phagocytosis of C. albicans. Culture media and reagents
tested for the presence of endotoxin by E-Toxate kit
References and notes
1
2
. Osorio, Y.; Ghiasi, H. J. Virol. 2003, 77, 5774.
. Goodman & Gilman, ‘Le basi farmacologiche della ter-
apia’, Zanichelli Ed.: Bologna, 1996, IX ed., cap. 51, pp
1203–1254.
(Sigma) were found to contain 10 pg of endotoxin per
mL. All media were free of Mycoplasma.
3. Arcamone, F. Doxorubicin — Anticancer Antibiotics;
Academic Press: New York, 1981.
4
. Wang, H. K.; Morris-Natschke, S. L.; Lee, K. H. Med.
Res. Rev. 1997, 17, 367.
5
.11. IFN-ꢁ/ꢀ titer
5. Pullman, B. Molecular mechanisms of specificity in DNA-
antitumor drug interactions. In Advances in Drug
Research, vol. 18; Academic Press: New York, 1989; p 74–
WISH cell lines and vesicular stomatitis virus (VSV)
were used to measure the levels of IFN-a/b in all the
U937 cell supernatants. All IFN titers were corrected
against standard (recombinant human IFN-a/b, specific
activity: 1 10 U/mg, Genzyme, Milan, Italy). One unit
of IFN-a/b was defined as the reciprocal value of
the highest dilution reducing the cytopathic effect by
7
. Sainz, B.; Halford, W. P. J. Virol. 2002, 76, 11541.
. Samuel, C. E. Curr. Top. Microbiol. Immunol. 1998, 233, 125.
6.
6
7
7
8. Vilcek, J.; Sen, J. Interferons and other cytokines. In Vir-
ology; Fields, B. N., Knipe, D. M., Howley, P. M., Eds.;
Raven Publishers: Philadelphia, Pa, 1996; p 375–400.
2
8
9
. Fischer, J. Biochem. J. 1995, 312, 215.
5
0%.
1
0. (a) Fisher, J. Gen. Pharmacol. 1996, 27, 1317. (b) Lull-
mann-Rauch, R. Exp. Toxicol. Pathol. 1994, 46, 315. (c)
Prokopek, M. Biochem. Pharmacol. 1991, 42, 2187.
1. (a) Fisher, J. Biochem. J. 1996, 315, 369. (b) Lullmann-
Rauch, R. Biochem. Pharmacol. 1995, 49, 1223.
5
.12. Statistical evaluation
1
Results are expressed as the mean of four experi-
mentsꢃstandard deviation (SD). Data were analysed by
one-way analysis of variance (ANOVA) and Student–
Newman–Keuls test.
1
1
2. Fenick, D. J. J. Med. Chem. 1997, 40, 2452.
3. Fischer, J. F.; Harrison, A. W.; Bundy, G. L.; Wilkinson,
K. F.; Rush, B. D.; Ruwart, M. J. J. Med. Chem. 1991,
3
4. (a) Kennedy, J. F.; White, C. A. Bioactive Carbohydrates.
4, 4140.
1
5
.13. Anticancer in vitro pre-screen
In Chemistry, Biochemistry and Biology; John Wiley &
¨
Sons: New York, 1983 (b) Musser, J. H.; Fugedi, P.;
Anderson, M. B. Carbohydrate-based therapeutics.
In Burger’s Medicinal Chemistry and Drug Discovery,
vol. I, 4th ed.; John Wiley & Sons: New York, 1995;
p 902–947.
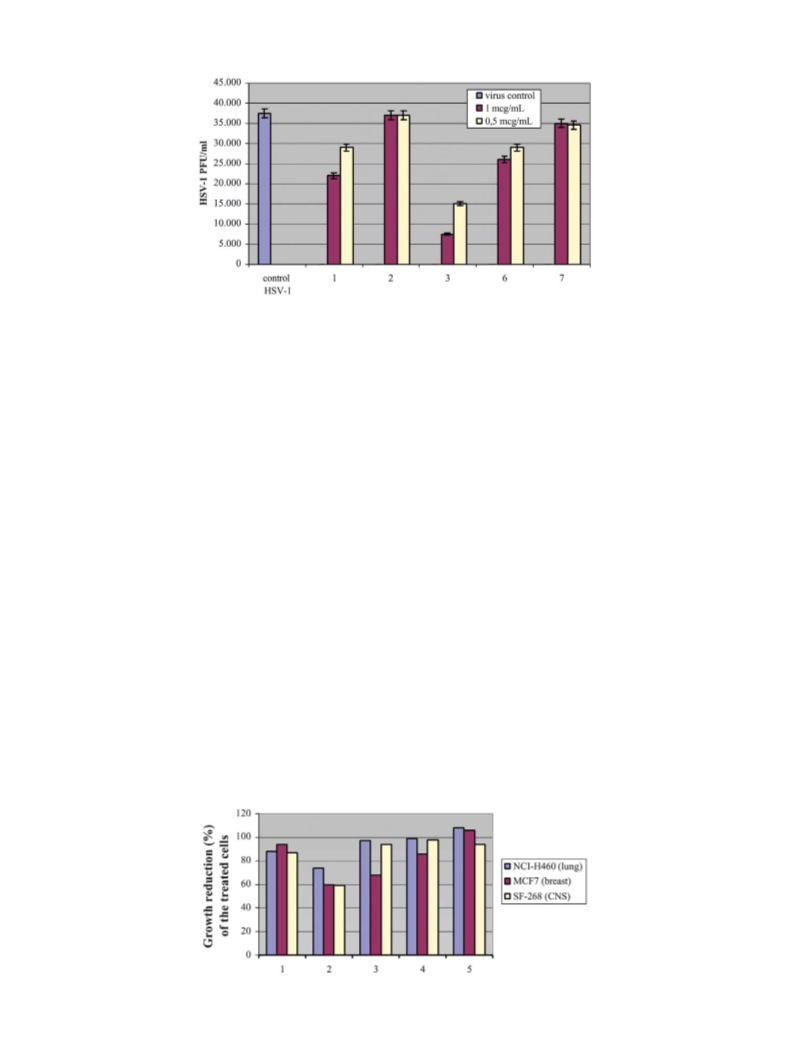
The in vitro disease-oriented primary antitumor screen-
ing performed in the US-National Cancer Institute
NCI) Laboratories of Bethesda—Maryland,24 consists
(
of the evaluation of the effects of each compound
against 3 tumoral cell lines: MCF7 (breast), NCI-H460
1
1
5. Simmons, A. J. Infect. Dis. 2002, 186, 71.
(
lung) and SF-268 (CNS). These 3-cell lines, one-dose
6. Hanessian, S.; Haskell, T. H. Antibiotics containing
sugars. In The carbohydrates — Chemistry and Biochem-
istry, 2nd ed.; Academic Press: London, 1970; p 139–200.
assay have been in use by Developmental Therapeutics
Program (DTP) for several years for the evaluation of

 Alcaro
Alcaro
 Arena
Arena
 Neri
Neri
 Ottana
Ottana
 Ortuso
Ortuso
 Pavone
Pavone
 Vigorita
Vigorita