
Journal of Physical Chemistry p. 2684 - 2688 (1990)
Update date:2022-08-17
Topics:
 Franzyshen, S. K.
Franzyshen, S. K.
 Schiavelli, M. D.
Schiavelli, M. D.
 Stocker, K. D.
Stocker, K. D.
 Ingram, M. D.
Ingram, M. D.
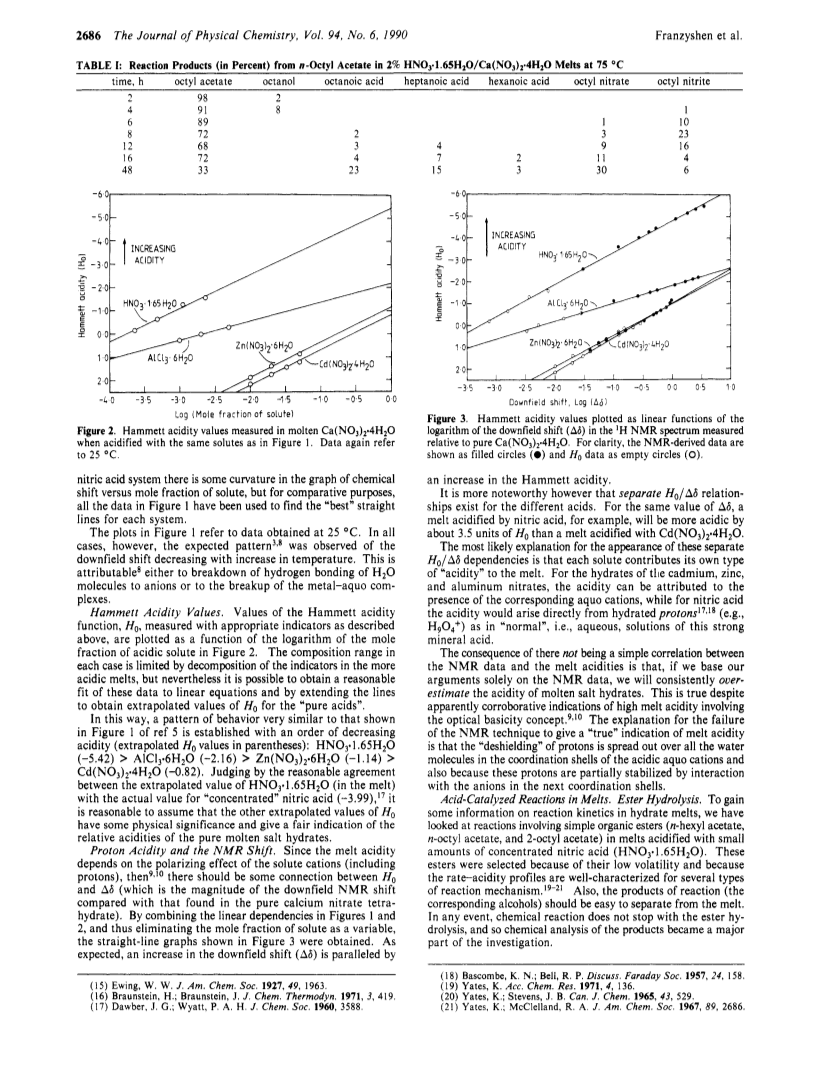
Molten Ca(NO3)2*4H2O has been used as a model system for studies of proton acidity and chemical reactions (including ester hydrolyses and aromatic nitrations) in molten salt hydrates.Increases in proton acidity caused by addition of acidic hydrates such as AlCl3*6H2O and Cd(NO3)2*4H2O or of aqueous HNO3 can be quantified in terms of changes in Hammett acidity H0, but usually not by the downfield shifts observed in the NMR spectrum.The rates of hydrolysis of selected primary esters (e.g., n-octyl acetate) were found to be linearly dependent on nitric acid concentration, indicating that the acidity of these melts can be changed with little change in water activity.The measurement of kinetic parameters is complicated, however, by side reactions between the organic substrates and HNO2, NO2 (or NO2+) and other species originating from the nitrate ions in the melt.
View More



Contact:+852-8198 2399
Address:9E, Leapont Industrial Building, 18-28 Wo Liu Hang Road, Shatin, New Territories, Hong Kong
Jiaxing Trustworthy Import And Export Co.,Ltd
Contact:+86-573-82030555
Address:Room 1202, Unit B, Charming plaza,No.1558 East Zhongshan Road , Jiaxing City, Zhejiang Province, China.
website:http://www.debyesci.com
Contact:+85221376140
Address:Rm. 19C, Lockhart Ctr., 301-307 Lockhart Rd., Wan Chai
Jiangsu Zenji Pharmaceuticals LTD
Contact:+86-025-83172562; +1-224-888-1133(USA)
Address:No.5 Xinmofan Road
Contact:+86-13666670345
Address:Agricultural Development Zone, Haining, Jiaxing, Zhejiang
Doi:10.1002/ejoc.201701663
(2018)Doi:10.1021/ja00162a047
(1990)Doi:10.3390/12071341
(2007)Doi:10.1021/acs.oprd.8b00076
(2018)Doi:10.1166/jnn.2016.11770
(2016)Doi:10.1134/S0022476617020184
(2017)