9380
C. Lehtinen et al. / Tetrahedron 56 (2000) 9375±9382
correlation between the stability and the length of O±O
bond. For example, in the case of 1c0 lengths of the O±O
bonds of the 0most stable and least stable one of the confor-
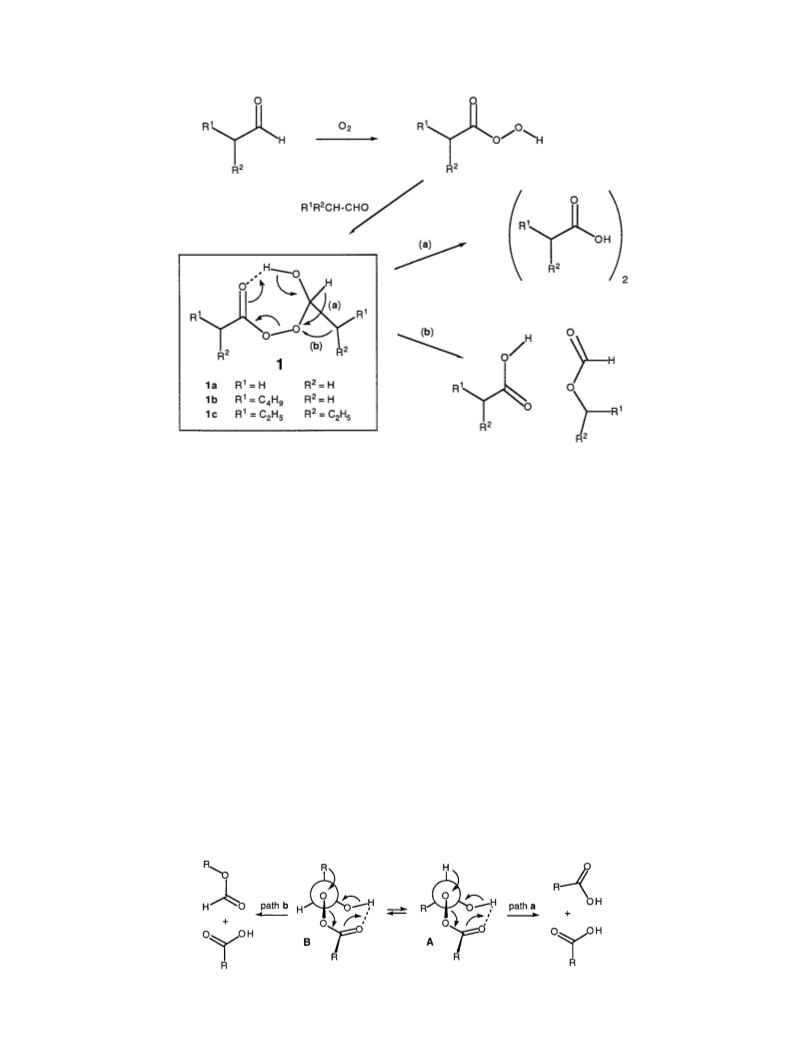
pathway a and b, Fig. 1) could operate giving rise to the
formation of formate along with the acid.
0
Ê
mers [i.e. 1c (C) and 1c (E)] differ only by 0.001 A. The
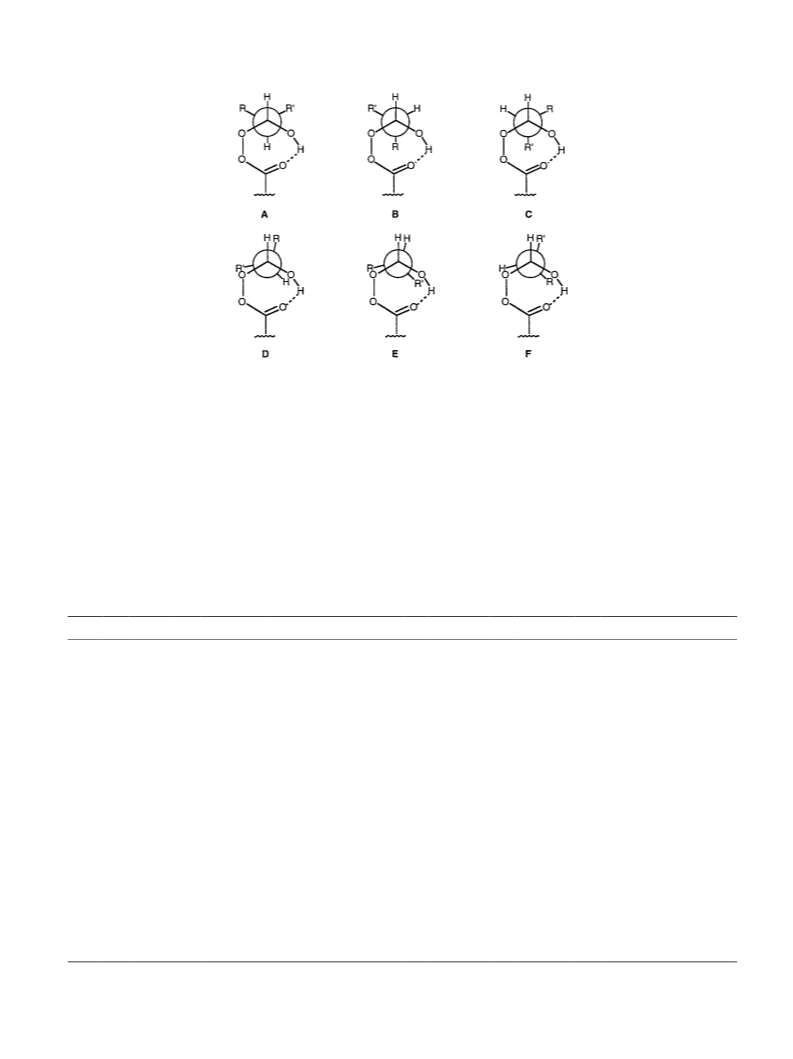
One way to inspect the results of the computational study
(Tables 2 and 3) is to select structures of which the geo-
metrical parameters show maximum similarity with those of
the transition state of the reaction. This type of comparison
can be carried out although the exact transition state had not
been determined because the direction of the change of the
structural parameters is known, as illustrated in Table 4. In a
maximum similarity structure the shortening bonds belong
to the group of the shortest bonds and the lengthening bonds
belong to the group of the longest bonds. The adducts 1b
(B), 1b0 (C), 1c (C) and 1c0 (F) (Table 4) turn out to
represent the structures of maximum similarity with the
transition state structure.
lengths of the OOH±Cald., Cald.±OOO, OOO±CCvO bonds vary
Ê
Ê
in the following ranges: 1.376±1.386 A, 1.407±1.417 A and
Ê
1.345±1.354 (i.e. all #0.010 A, Table 2).
As the conversion of adducts 1 to the products (the Scheme)
will involve migration of either R or H of the R-CHO
moiety of 1 (Fig. 1), the bond angles R±C±O and H±C±
O along with the torsion angle X±C±O±O [in which X is
the migrating group (i.e. R or H)] could be worth
comparing. The values of these angles are presented in
Table 3.
When the migration of hydrogen takes place along pathway
a (Fig. 1) the bond angle H±Cald.±O will ®rst approach 908
and then decrease signi®cantly below the level of 908.
Therefore, the smaller the value of this angle in adduct 1,
the more closely its structure resembles the transition state
structure of the pathway (a, Fig. 1). Among the conformers
of 1b and 1c (H migrating) the least stable ones are the
adducts 1b (F) and 1c (D). However, the H±Cald.±O bond
angle of 1b (F) is closer to 908 than that of any other of the
conformers of 1b. The same observation can be made in the
case of 1c (D). When the values of the R±Cald.±O angle of
conformers of 1b0 and 1c0 (R migrating) are compared it can
be seen that the values closest to 908 belong to the con-
formers 1b0 (F) and 1c0 (F) both of which in turn belong
to the group of the most unstable conformers. This is
consistent with the well known Hammond postulate.
When maximum similarity adducts 1b (B), 1b0 (C), 1c (C)
and 1c0 (F) (Table 4) are compared the following observa-
tions can be made: (1) the angle X±Cald.±OOO of 1c0 is
smaller than that of 1b0 (indicating that in the case of
branched 10c the formation of formate is more probable
than in the case of straight chain 10b); (2) the difference
of the values of the X±Cald.±OOO (X is the migrating
group) angles is smaller in the case of 1c and 1c0 than in
that of 1b and 1b0 [indicating that in the case of 1c/1c0 the
probability of the reactions taking place along both path-
ways a and b (Fig. 1) could be higher than in the case of 1b/
1b0]; (3) the OCvO±HOH bond of 1c (which should shorten in
the reaction) is clearly longer than that of 1b, 1b0 or 1c0
[indicating that the pseudo cyclic ring system of 1c is less
activated for the formation of acid (along pathway a, Fig. 1)
than that of 1b, 1b0 or 1c0]; (4) the of OOH±Cald. bond of 1b0
(which should shorten in the reaction) is clearly longer than
that of 1b, 1c or 1c0 [indicating that the pseudo cyclic ring
system of 1b0 is less activated for the formation of formate
(along pathway b, Fig. 1) than that of 1b, 1c or 1c0]; (5) the
value of the X±Cald.±OOO±OOO torsion angle of 1b0 deviates
clearly more from 1808 than that of 1b, 1c or 1c0 [indicating
that the conformation of the migrating R group of 1b0 is less
favorable for the reaction (formation of formate along
pathway b, Fig. 1) than that of 1b, 1c or 1c0]. These ®ve
observations indicate, that the formation of acid could be
more favorable in the mixture containing 1b and 1b0 than in
that containing 1c and 1c0 mainly because the competing
reaction along pathway b (Fig. 1) is inhibited by unfavor-
able structural factors in the case of adducts of straight chain
aldehydes such as 1b0, respectively.
When the H±Cald.±O bond angles of adducts 1b and 1c
(hydrogen should migrate) are compared with those of 1b0
and 1c0 (hydrogen should not migrate) the values of the
former group are all clearly closer to 908. In the same
way, when the R±Cald.±O bond angles of adducts 1b and
1c (R should not migrate) are compared with those of 1b0
and 1c0 (R should migrate), the values of the latter group are
all clearly closer to 908. On the other hand, the H±Cald.±O
bond angles of adducts 1b and 1c (hydrogen should
migrate) are about 5±68 smaller than the values of the
related R±Cald.±O angles of their epimers 1b0 and 1c0
(R should migrate). Therefore, migration of hydrogen (a,
Fig. 1) should be clearly more favorable than that of R (b,
Fig. 1). This is consistent with the experimental results
(Table 1).
The X±C±O±O [X is the migrating group (i.e. R or H)]
torsion angles listed in Table 3 are also revealing when
one is trying to understand why formates are formed when
a-substituted aldehydes are oxidized but not when the corre-
sponding straight chain aldehydes are treated accordingly.
Namely, the X±C±O±O torsion angles of all conformers of
epimer 1b0 [except that of 1b0 (B), Table 3] clearly deviate
from 1808 more (four of the values in the range of 172.5±
173.78) than those of 1b, 1c or 1c0 (only two values out of
the range 176.4±178.78). Therefore, the less favorable
spatial arrangement of the migrating group in the case of
straight chain aldehydes (analogous to 1b) could inhibit the
formation of formates (via pathway b, Fig. 1) whereas in the
case of a-branched aldehydes both mechanisms (along with
Conclusions
Results of this study indicate, that the 2-substitution of the
reacting aldehyde appears to be crucial for formate produc-
tion. A phenyl group at the a-position directed the reaction
more ef®ciently to the corresponding formate than a straight
chain group (2-phenylpropionaldehyde was converted
almost completely to formate when m-CPBA was used as
an oxidant). Under the typical conditions of O2 in situ oxida-
tion in chlorinated solvents 50% of the adduct formed from
a-branched aldehydes, such as 2-ethylhexanal and 2-ethyl-
butanal, reacted via carbon chain migration. An even higher
preference (e.g. 79/21 in the case of 2-ethylhexanal) for

 Lehtinen, Christel
Lehtinen, Christel
 Nevalainen, Vesa
Nevalainen, Vesa
 Brunow, G?sta
Brunow, G?sta