1
4
structure-reactivity correlations spanning the diverse cata-
Supporting Information). Their reactivity was screened for
lytic routines.2 In this light, an appealing alternative is
,5
cis-cyclooctene epoxidation under MW irradiation (Table
1
3
represented by H
playing persistent structures under oxidation turnovers.
Worthy of notice is the catalytic performance of [γ-SiW10
2
O
2
-stable polyoxotungstates (POM), dis-
1).
2
,7,8
34
O
-
4
-
(H
2
O)
2
]
with selectivity up to >99% in the epoxidation
2 2
Table 1. MW-Assisted cis-Cyclooctene Epoxidation with H O
Catalyzed by Hybrid [G XW12-nO ]
z 40-m
8
of internal and terminal double bonds (2-10 h at 32 °C).
k- a
This latter catalyst is characterized by a divacant structure,
catalyst
POMb
G (z)
PhSi (2)
epoxide yield (%)c
featuring a tetra-oxygenated, nucleophilic site on the POM
R-PW11O397
-
54
92
9
1
2
2 2
surface and four W(VI) atoms capable of H O coordination.
PhPO (2)
PhSi (2)
PhPO (2)
MeSi (2)
PhSi (2)
PhSi (2)
PhPO (2)
NH2(CH2)2Si (2)
Ph(CH2)2Si (2)
PhSi (4)
In this system, two major drawbacks are represented by
protonation equilibria, likely engaging the lacunary site,
d
R-SiW11O398-
e
e
e
3
4
5
6
7
8
9
1
48
85
28
73
91
97
97
75
92
87
1
0
d
which alter the POM solubility and reactivity and by a
thermally induced catalyst deactivation. A convenient remedy
can be provided by the covalent functionalization of the POM
lacunary oxygens through the attachment of organic moi-
γ-PW10O367
γ-SiW10O36
-
8
-
e
e
1
1,12
eties.
Such a hybrid way to the POM synthesis imparts
0
9
-
a strong stabilization of the vacant structure while generating
catalyst diversity that might also include the most desirable
chiral upgrade. The key strategy is to provide the W(VI)
sites with proximal stereoelectronic effectors, which are
11
12
9 34
R-PW O
PhPO (2)
PhSi (4)
R-SiW9O341
0-
>99e
13
a
POM, 0.8 µmol; cis-cycloctene, 0.5 mmol; H2O2 (35%), 0.1 mmol;
CH3CN (0.6 mL); MW irradiation for 50 min at 240 W; Tbulk ) 120 °C.
1
1,12
expected to tune the reactivity of the competent oxidant.
b
c
Lacunary precursor used as TBA salts. Calculated with respect to initial
1
3
H O . POM used as Ph4P salt. e After 100 min of irradiation.
d
+
Herein we report on (i) the MW-assisted fast screening of
hybrid Keggin 9-11 polyoxotungstates as epoxidation cata-
lysts, (ii) a remarkably wide reaction scope accessed through
the combined used of the most performing catalyst and MW
irradiation, and (iii) LFER studies under conventional and
MW-induced dielectric heating. The first evidence of enan-
tioselective oxygen transfer mediated by a chiral hybrid POM
is also included. As corollary, the stability of the different
catalytic systems has been addressed by FT-IR, heteronuclear
NMR and ESI-MS techniques (see Supporting Information).
2
2
MW-induced dielectric heating is efficiently used by these
poly-charged catalysts, behaving as MW-activated molecular
1
5
heat carriers. Furthermore, functionalization of the lacunary
site prevents the thermal rearrangement of the POM struc-
1
6
ture. The catalyst performance is strongly dependent on
the structure/composition of the inorganic framework as well
as on the nature of the organic moiety decorating the POM
surface. A superior performance, with yields in the range
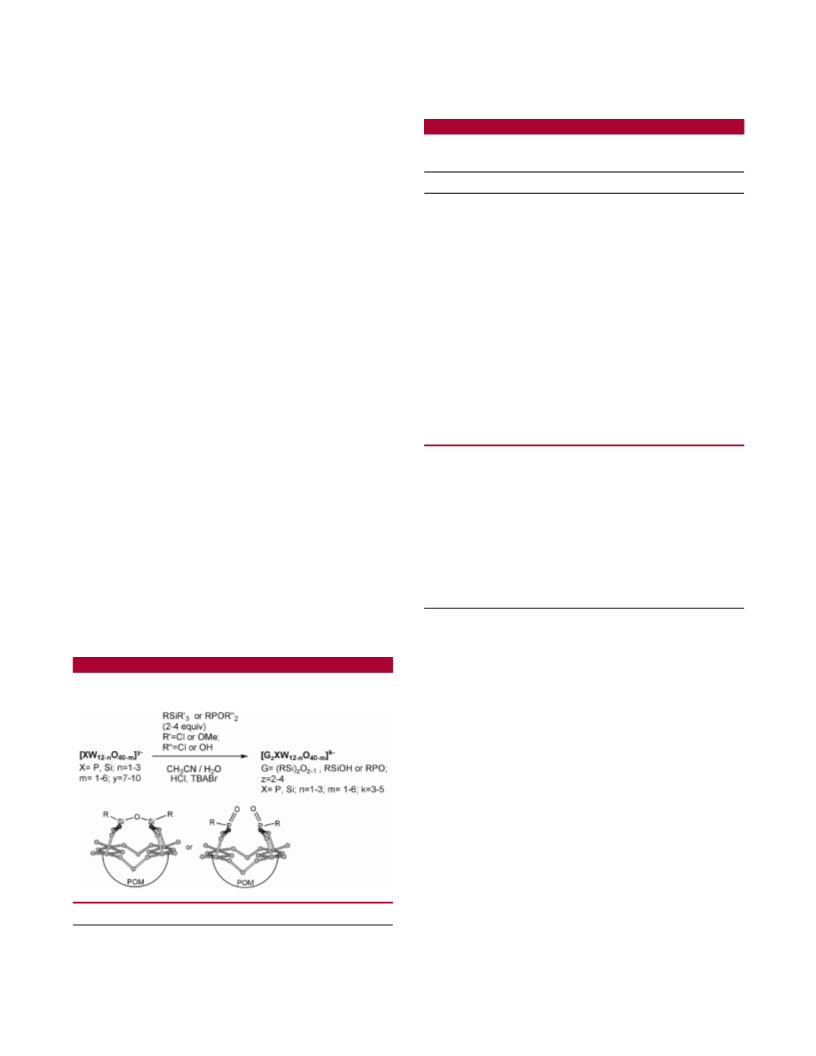
Hybrid complexes were prepared from lacunary polyoxo-
tungstates and organosilanes or phosphonic reagents accord-
ing to straightforward literature protocols (Scheme 1 and
(
8) (a) Mizuno, N.; Kamata, K.; Yonehara, K.; Sumida, Y. Science 2003,
300, 964. (b) Mizuno, N.; Yamaguchi, K.; Kamata, K. Coord. Chem. ReV.
005, 249, 1944-1956. (c) Kamata, K.; Nakagawa Y.; Yamaguchi, K.;
Mizuno, N. J. Catal. 2004, 224, 224-228.
9) The tetraperoxo complex derived from addition of H2O2 to the four
2
(
9-
lacunary W(VI) of R-Keggin [CoW11O39] has been characterized by X-ray
analysis and found to be reactive in the epoxidation of 2-cyclohexenol.
See: Server-Carri o´ , J.; Bas-Serra, J.; Gonz a` lez-Nu n˜ ez, M. E.; Garc `ı a-
Gastaldi, A.; Jameson, G. B.; Baker, L. C. W.; Acerete, R. J. Am. Chem.
Soc. 1999, 121, 977-984.
Scheme 1. Surface Derivatization of Lacunary
Polyoxotungstates
(10) The catalytic activity of the lacunary complex is highly dependent
on the pH at which it is isolated. For a discussion on the speculated structure
of the active catalyst, see: Musaev, D. G.; Morokuma, K.; Geletii, Y. V.;
Hill, C. L. Inorg. Chem. 2004, 43, 7702-7708.
(
11) Zeng, H.; Newkome, G. R.; Hill, C. L. Angew. Chem., Int. Ed. 2000,
3
9, 1772-1774.
(12) (a) Bonchio, M.; Carraro, M.; Scorrano, G.; Fontananova, E.; Drioli,
E. AdV. Synth. Catal. 2003, 345, 1119-1126. (b) Bonchio, M.; Carraro,
M.; Scorrano, G.; Bagno A. AdV. Synth. Catal. 2004, 346, 648-654.
(
13) Bonchio, M.; Carraro, M.; Kortz, U.; Scorrano, G. AdV. Synth. Catal.
2
005, 347, 1909-1912.
14) (a) Weeks, M. S.; Hill, C. L.; Schinazi, R. F. J. Med. Chem. 1992,
5, 1216-1221. (b) Mayer, C. R.; Fournier, I.; Thouvenot, R. Chem. Eur.
(
3
J. 2000, 6, 105-110. (c) Niu, J.; Li, M.; Wang, J. J. Organomet. Chem.
2
003, 675, 84-90. (d) Mayer, C. R.; Thouvenot, R. J. Chem. Soc., Dalton
Trans. 1998, 7-13. (e) Mayer, C. R.; Herson, P.; Thouvenot, R. Inorg.
Chem. 1999, 38, 6152-6158. (f) Sun, Z.-G.; Liu, Q.; Liu, J.-F., Polyhedron
2
000, 19, 125-128. (g) Kim, G.-S.; Hagen, K. S.; Hill C. L. Inorg. Chem.
(7) (a) Adam, W.; Alsters, P. L.; Neumann R.; Saha-M o¨ ller, C. R.;
1992, 31, 5316-5324. (h) Ammari, N.; Herv e´ , G.; Thouvenot, R. New. J.
Sloboda-Rozner, D.; Zhang, R. J. Org. Chem. 2003, 68, 1721-1728. (b)
Sloboda-Rozner, D. Alsters, P. L.; Neumann, R. J. Am. Chem. Soc. 2003,
Chem. 1991, 15, 607-608.
(15) Roberts, B. A.; Strauss, C. R. Acc. Chem. Res. 2005, 38, 653-661.
(16) Nonfunctionalized lacunary POMs evolve to saturated W12-deriva-
tives under MW-assisted catalysis.
1
25, 5280-5281. (c) Sloboda-Rozner, D.; Witte, P.; Alsters P. L.; Neumann,
R. AdV. Synth. Catal. 2004, 346, 339-345.
3672
Org. Lett., Vol. 8, No. 17, 2006

 Carraro, Mauro
Carraro, Mauro
 Sandei, Luca
Sandei, Luca
 Sartorel, Andrea
Sartorel, Andrea
 Scorrano, Gianfranco
Scorrano, Gianfranco
 Bonchio, Marcella
Bonchio, Marcella