A. Ghosh et al. / Journal of Molecular Liquids 211 (2015) 48–62
49
electron oxidant (E° = 1.4 V in 1.0 mol dm−
3
H SO
2
) [13]. In sulfuric
by using the weighing balance (Sartorius BSA224S-CW) and dissolving
calculated amount of surfactants in double distilled water through
Digital Ultrasonic Cleaner CD 4820 instrument. Surfactant substrate
mixture solution and other mixture solution containing surfactant
were centrifuged to get a complete mixing through Centrifuge-Z206A
(Hermle Labortechnik GmbH).
4
acid and sulfate media cerium(IV) forms several sulfate complexes.
Generally transition metals in a higher oxidation state can be stabilized
by chelation with suitable complex agent. However, our preliminary ob-
servations indicate that oxidation of some organic compounds by Ce(IV)
in aqueous sulfuric acid is kinetically sluggish, the process can be effi-
ciently catalyzed by various metal ions at trace concentration. Different
metal ion catalysts like chromium(III), ruthenium(III), iridium(III) [14]
etc. are generally used in the oxidation by cerium(IV). Among the differ-
ent metal ions, ruthenium(III) [15] and iridium(III) [16] are highly effi-
cient [17]. Reaction mechanism of various elementary reactions must be
investigated to analyze the effect on selectivity. Therefore, the basic
study of catalytic reaction will prove the scientific basis for improving
catalyst selectivity and making high-efficiency catalyst. Various metal
ions such as Ag(I), Mn(II), Cu(II), Os(VIII), Hg(II), Cr(III), Ru(III), Ir(III),
etc. have been employed as catalysts in cerium(IV) oxidation of differ-
ent types of substrates. However, ruthenium(III) and iridium(III) are
highly efficient catalysts for cerium(IV) oxidations even at trace concen-
2.2. Kinetic measurements
Kinetic experiments were carried out spectrophotometrically
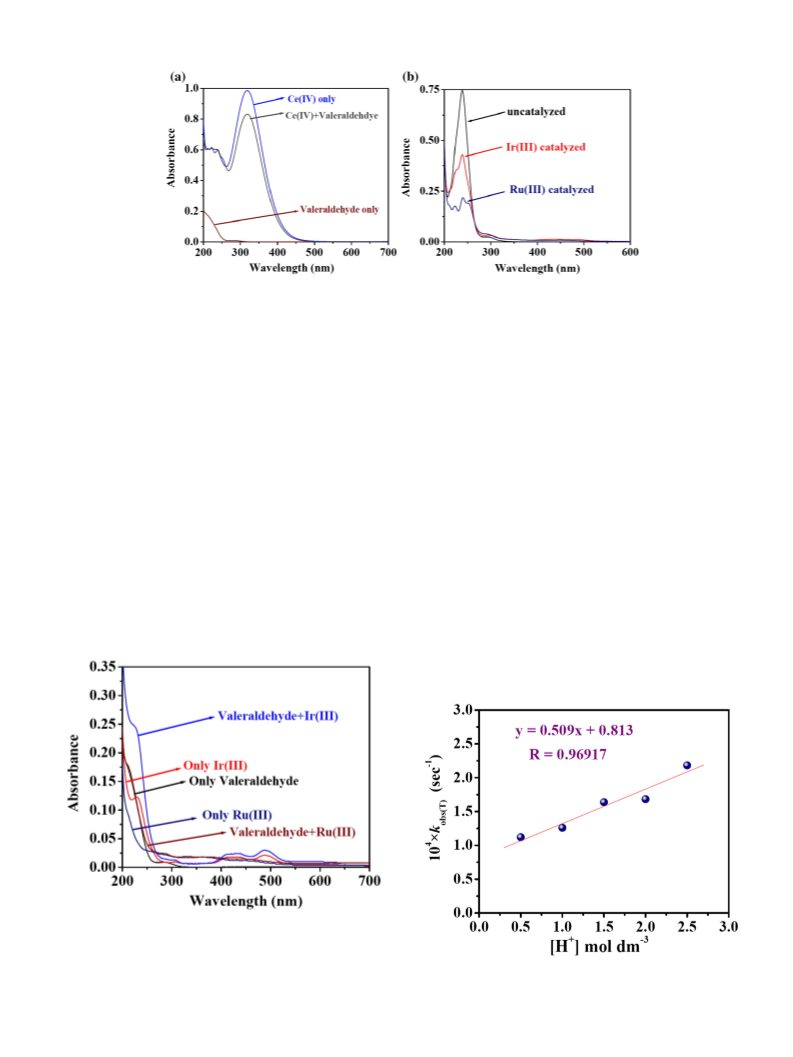
by recording the changes in the Ce(IV) absorbance at 320 nm with
a computer-controlled UV–VIS Shimadzu-1800 spectrophotometer,
equipped with thermostated compartments for 1 cm cuvettes. The
temperature of the experiments was regulated to 30.0 ± 0.1 °C with a
thermostat TCC Shimadzu. The kinetics of the oxidation of the organic
substrates by cerium(IV) were studied in the presence of a large
excess of organic substrate over the Ce(IV) species: [valeraldehydel]
T
≥
1
0[Ce(IV)] . The observed pseudo-first-order rate constants, kobs, were
T
−
6
−7
−3
tration levels (ca. 10 –10 mol dm ) [17,18].
obtained from the slopes of the linear least-squares fit of the plot
of −ln(A320) vs time (t) (Fig. 1), and were reproducible to within 3%
with good correlation coefficient values [16,20,25,26]. The kinetics
was followed up to 80% completion of the reaction and good first-
order kinetics were observed. The t1/2 values are directly calculated in
The present work continues our kinetic studies of micellar effects
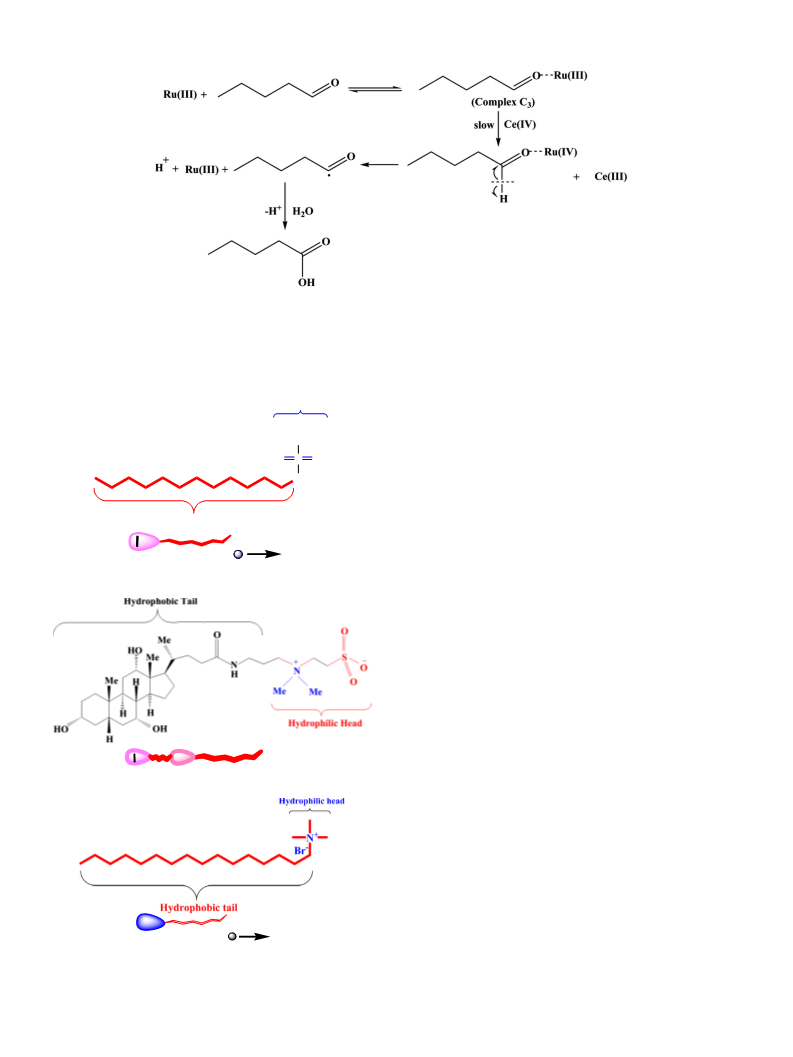
on the metal-catalyzed oxidation of valeraldehyde. In this study,
we have experimented with a nontoxic (benign) amphiphile (3-[(3-
cholamidopropyl)dimethylammonio]-1-propanesulfonate (CHAPS)) in
both water and metal-water media to look into their interactions to pro-
duce results that would lead to their uses in catalysis and industries.
CHAPS is a zwitterionic derivative of cholic acid having combined prop-
erties of both sulfobetaine type detergents and bile salts [19]. Once
again we have taken a more precise and detail experimental effort on
the catalyzing properties of non-toxic surfactant CHAPS in oxidation
kinetics [20].
Generally organic substrates are very often poorly soluble in water.
Micellar systems can be used to overcome the solubility problems of
the organic substrates. Self-organized assemblies such as micelles can
change the rates of chemical and enzymatic reactions. Effects of micelles
of surfactants on these reactions can be attributed to their electrostatic
and hydrophobic interactions with reactants [21–23]. Surfactants are
amphiphilic organic compounds, containing both hydrophobic groups
and hydrophilic groups. Thus, a surfactant molecule contains both
a water insoluble and a water soluble component [24]. We have
employed an approach to the determination of kinetic constants of
metal mediated-micelle catalyzed oxidation of valeraldehyde that al-
lows the identification of important mechanism and function of differ-
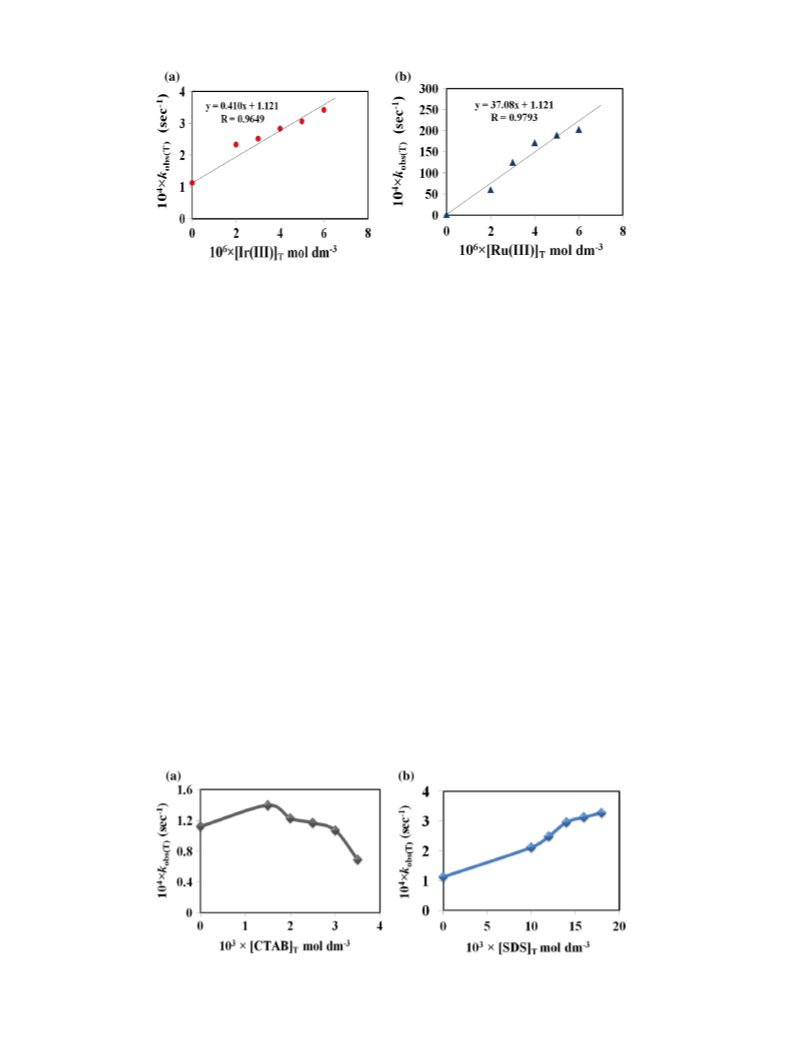
ent surfactants on the reaction rates. The different rate constants in
the presence and absence of surfactants have been determined with
the corresponding plots. SDS, CHAPS and CTAB have been found to ac-
celerate the rate process for the valeraldehyde oxidation process.
Table 1 by using the relation t1/2 = (ln2/kobs), where ln2 = 0.693,
−1
k
obs = pseudo-first-order rate constant in s . The kobs values were
found to be independent of the initial Ce(IV) concentration, and conse-
quently, the oxidation rates are first order with respect to the oxidizing
species. The surfactant concentrations used in all experiments were no-
tably above the critical micelle concentrations [27,28] to make sure of
the existence of micellar aggregates in the solution. The possibility of
decomposition of the surfactants SDS, CTAB and CHAPS by Ce(IV) has
been investigated and the rate of decomposition has been found negli-
gible [29].
2.3. Product analysis and stoichiometry
Ce(IV) (2.0 × 10−4 mol dm−3) was taken in distilled water (15 ml)
−3
−3
2 4
and H SO (4.2 ml). A solution of substrate (2.0 × 10 mol dm ) was
added to the reaction mixture and stirred for 24 h at 30–35 °C. Ether was
added to the reaction mixture. The organic layer was extracted, washed
with water, and dried over anhydrous MgSO . Ether was removed by
4
warming, and the products [4] were obtained (valeric acid from
valeraldehyde), whose boiling points were in agreement with literature
values (yields ~85–90%). The reaction product was subjected to FTIR
(
Fig. S1, Supplementary material) (Shimadzu Prestige-21) and proton
NMR analysis, and characterized as follows: Valeric acid: υ = 2958
−
1
(
br, s, −OH), 2673, 1705 (s, C_O), 1411, 1211, 937, 748 cm
.
2
. Experimental
1
The oxidation products were analyzed by the H NMR with a Bruker
ASCEND spectrometer at a frequency of 400 MHz using CDCl as the sol-
vent and SiMe as internal standard (Fig. S2, Supplementary material)
30].
The stoichiometry of the reaction in the case of valeraldehyde may
be given by Eq. (1):
3
2
.1. Materials and reagents
4
[
Valeraldehyde (SRL, AR, Mumbai, India), cerium(IV) ammonium
sulfate (E. Merck, AR), cetyltrimethylammonium bromide (CTAB)
(
(
(
SRL, AR, Mumbai, India), CHAPS (SRL, AR, Mumbai, India), H
E. Merck, AR), SDS (Qualizen) Na SO (E. Merck, AR), HCl
E. Merck, AR), pyrene (SRL, AR, Mumbai, India), iridium(III)
2 4
SO
CeðIVÞ þ C4H9CHO þ H2SO4→2CeðIIIÞ þ C4H9COOH þ 2Hþ:
2
4
2
ð1Þ
chloride (SRL, AR, Mumbai, India), ruthenium(III) chloride (SRL, AR,
Mumbai, India), and all other chemicals used were of the highest purity
available commercially. The stock solution of Ce(IV) was obtained by
2.4. Test for free radicals
−
3
dissolving cerium(IV) ammonium sulfate in 1 mol dm sulfuric acid
and was standardized with iron(II) ammonium sulfate solution using
ferroin as an external indicator [25,26]. Cerium(IV) solution was always
made up and stored in a black coated flask to prevent photochemical re-
action. A solution of iridium(III) chloride and ruthenium(III) chloride
were prepared by dissolving the sample in a minimum amount of con-
centrated hydrochloric acid. The solution of surfactants were prepared
Acrylonitrile (monomer) was used for the identification of free rad-
icals. In a typical experiment, reaction mixture containing [Ce(IV)] =
−
4
−3
−3
−3
2.0 × 10
mol dm , [valeraldehyde] = 2.0 × 10
mol dm
,
−
3
[H SO ] = 0.5 mol dm , and acrylonitrile (=30% v/v) at 30 °C led to
2
4
the formation of white polymeric product indicating in situ generation
of free radicals [13,25].

 Ghosh, Aniruddha
Ghosh, Aniruddha
 Sar, Pintu
Sar, Pintu
 Malik, Susanta
Malik, Susanta
 Saha, Bidyut
Saha, Bidyut