DOI: 10.1002/cssc.200900154
Valeronitrile Hydrolysis in Supercritical Water
Michael Sarlea, Sabine Kohl, Nina Blickhan, and Herbert Vogel*[a]
Nitriles act as important intermediates for the chemical indus-
try and are accessible on a large scale through hydrocyanation
or ammonoxidation. Nitrile hydrolysis yields amides and acids
used in various applications. The conventional nitrile hydrolysis
process relies on stoichiometric amounts of mineral acid or
base which inherently deliver great amounts of waste brine.
Improving this process towards green chemistry would require
reaction conditions which can provide technically significant
results without the use of catalysts. Under these conditions,
the hydrolysis of valeronitrile in pure supercritical water was in-
vestigated. The experiments were performed in a continuous
high pressure laboratory-scale apparatus at a temperature be-
tween 400 and 5008C, 30 MPa pressure and maximal residence
time of 100 s. Nitrile conversion and valeric acid selectivity
greater than 90% were achieved.
Introduction
Results and Discussion
In the past few years sustainability has gained in importance in
the chemical industry. Supercritical (sc) fluids have awakened
interest as new, environmental friendly solvents and processes
and methods of synthesis have been evaluated.[1–3] Sc fluids
and especially water offer many advantages in comparison to
ordinary organic solvents.[4,5] Supercritical water is cheap, non-
flammable, and environmentally friendly. Furthermore, it poses
no health threat and is easily available worldwide .[6] The out-
standing characteristic of water as a reaction medium is the
possibility of tuning properties, such as polarity or ionic prod-
uct, by changing temperature and pressure.[7–10]
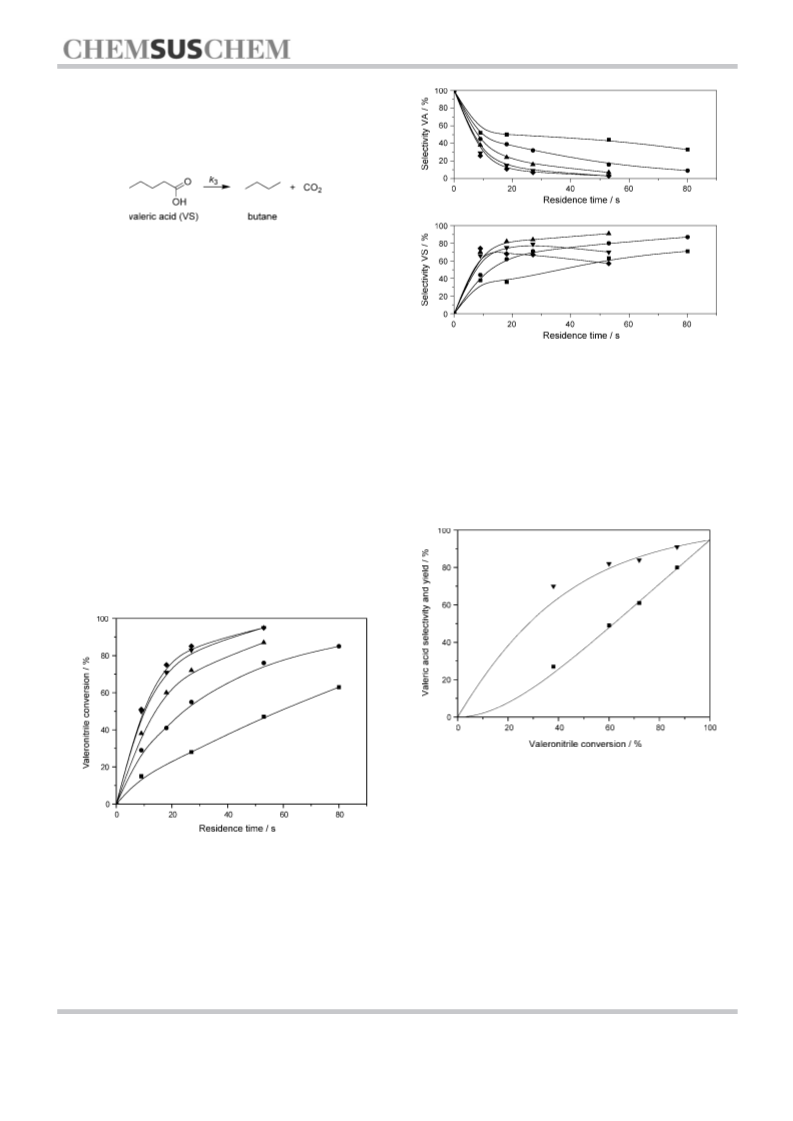
Kinetic tests on valeronitrile, valeramide and valeric acid were
performed at temperatures between 400–5008C, 30 MPa pres-
sure, and maximal residence time of 100 s. The collected data
and findings are presented below.
Usually hydrolysis reactions under hydrothermal conditions
are performed at near-critical temperature to take full account
of the high dissociation rate and thus high catalytic potential
exhibited by water. However, valeronitrile demonstrates a fair
level of temperature stability and therefore higher tempera-
tures had to be applied for the kinetic investigations.
The reaction mechanism is well-known and described in lit-
erature.[20–22] Nitriles react with water yielding amide intermedi-
ates which react in a second step to form the corresponding
carboxylic acid (Scheme 1).
Nitriles are of great interest with regard to sustainability
both because of their appearance in industrial waste-streams
and their familiar usage as starting materials for commodity
chemicals production.[11,12] The default hydrolysis process is
based on the addition of strong acids, bases, or enzymes. A
good example is the biotechnological hydrolysis of acryloni-
trile, which shows high selectivity under mild reaction condi-
tions (20–408C, pH 7, ambient pressure).[13,14] The deficiency of
the acid or base-dependant process is the high amount of
waste brine which results during the extraction of the desired
products.[15,16] This disadvantage can be circumvented by the
use of supercritical water which serves as a catalyst, solvent,
and reactant.[17–19]
Under the aforementioned experimental conditions, valeric
acid can undergo a decarboxylation reaction to butane.[23,24]
The goal of this work was the confirmation of the reaction
network suggested in literature by hydrolyzing both valeroni-
trile and the intermediate valeramide.[20] Furthermore, optimal
reaction conditions for maximizing the valeric acid yield were
determined, which are significant for potential industrial appli-
cation. This has proven difficult due to product consumption
by decarboxylation reactions. Hence the stability of the main
product, valeric acid, was investigated. Kinetic results were
gathered and compared with modeled findings provided by
the program Presto-Kinetics.
Scheme 1. Hydrolysis of valeronitrile to valeric acid via the intermediate va-
leramide.
[a] M. Sarlea, S. Kohl, N. Blickhan, Prof. H. Vogel
Ernst-Berl-Institut fꢀr Technische und Makromolekulare Chemie
Technische Universitꢁt Darmstadt
Petersenstrasse 20, 64287 Darmstadt (Germany)
Fax: (+49)6151-163465
ChemSusChem 2010, 3, 85 – 90
ꢀ 2010 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
85

 Sarlea, Michael
Sarlea, Michael
 Kohl, Sabine
Kohl, Sabine
 Blickhan, Nina
Blickhan, Nina
 Vogel, Herbert
Vogel, Herbert