2
1
1
of 1b (65%, crude) containing no detectable bromoalkanol 1c
the 1921 procedure , involving H SO , remains to be improved
2 4
upon.
1
(
HNMR).
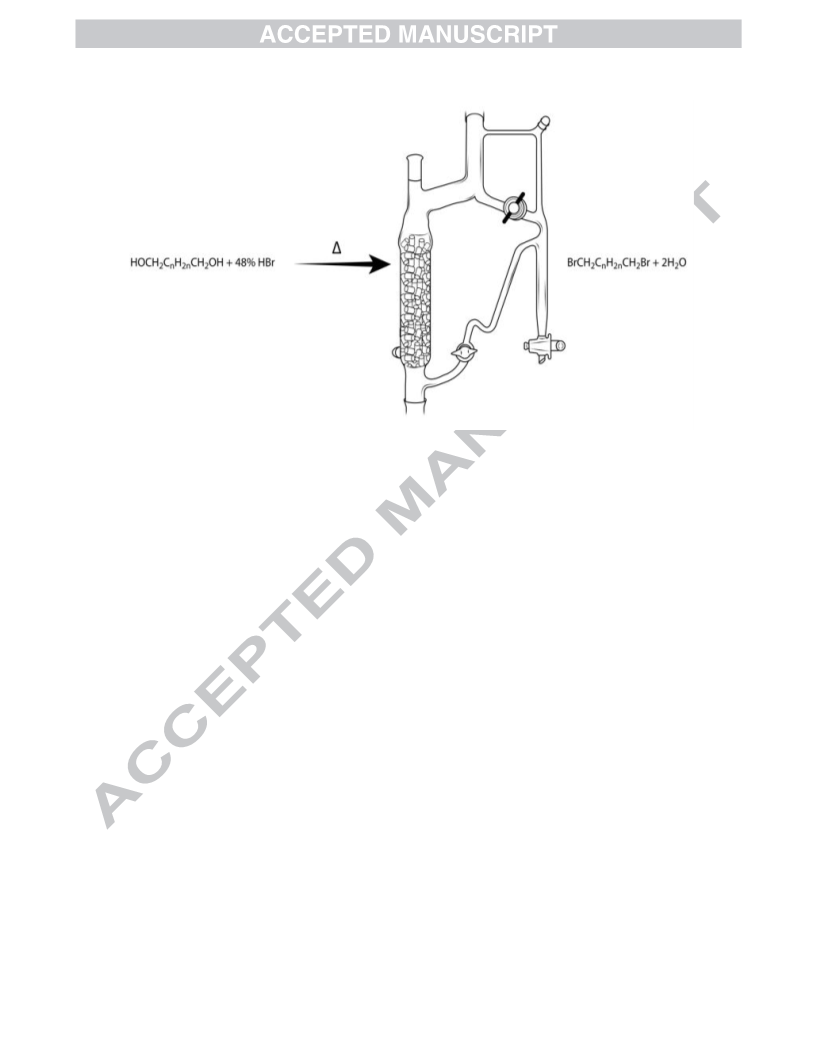
Revised apparatus for the second synthesis of 1b (entry 2)
In summary, introduction of a fractionating column, use of
octane as the optimal non-aqueous phase, intensified distillation, and
optimization of other parameters have enabled the reaction of a range
of ,-diols with 48% aqueous HBr to become a simple, rapid,
economical, and scalable route to very useful ,-dibromides. It
appears likely that further scaleup, modification of the fractional
distillation apparatus, and development of excess HBr recovery
methodology could lead to a viable commercial process.
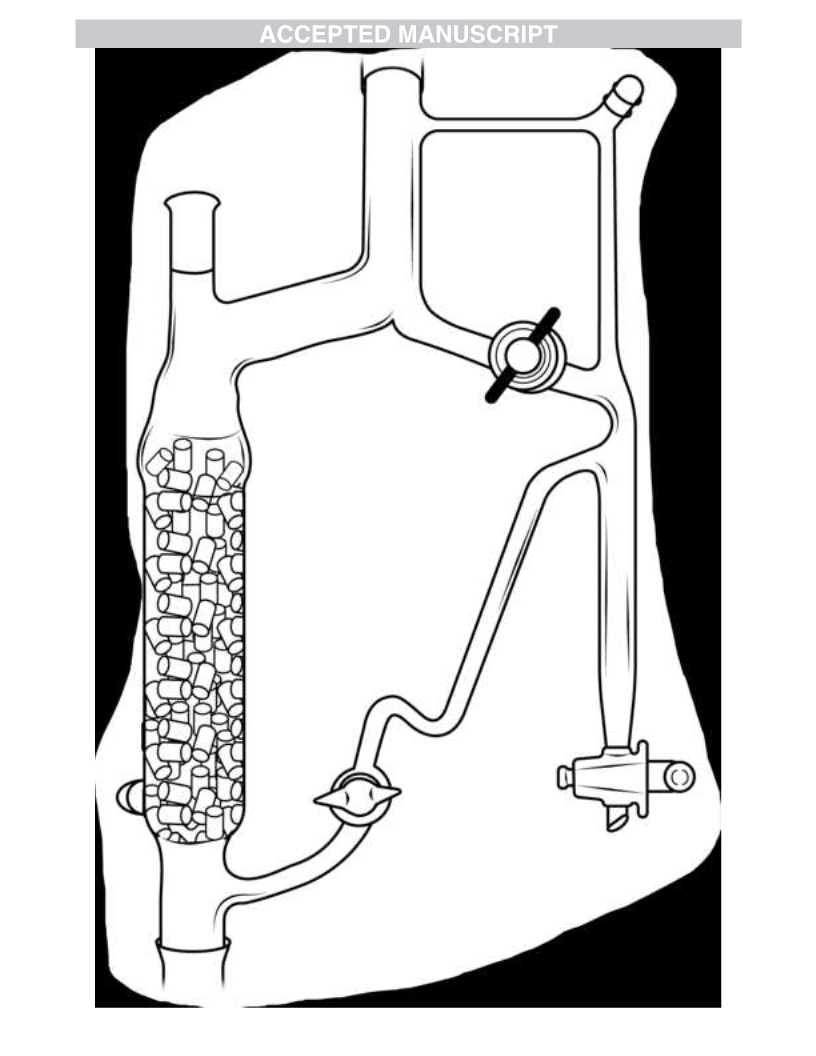
incorporated a vacuum-insulated, Raschig ring-packed, 19-cm
fractionating column, cold finger splitter head, solvent return to the
reaction flask, and a standard Dean-Stark graduated collector to
remove condensed aqueous phase (Figure 1). Under conditions
described in entry 2, Table 1, a 72% crude yield of 1b was obtained
(no 1c). We will return to 1b later.
Figure 1. Modified Dean-Stark apparatus
1
The widely used straight-chain dibromides 2b, 3b, 5b, and 6b were
chosen to explore the effects of chain length on the HBr-diol
Supplementary Data
1
1
reaction. The 1921 preparation of 2b (5-6 h refluxing) yielded 88-
1
2
9
5% of distilled product. A 2008 report, using 2a, anhydrous HBr,
1
13
General and selected experimental procedures; H and C NMR
data for all dibromides.
and toluene, claimed 100% yield of 2b, but gave no experimental
details. Our preparation of 2b appears in entry 3, Table 1. The fair
yield (66%) was attributed to a too-low pot temperature and loss of
HBr in the high-boiling azeotrope.
Acknowledgments
Attention turned to 3b. A viable preparation of this very useful
1
13,14
We thank the Syracuse University Department of Chemistry and
the Hahn Gift Fund for financial support.
dibromide was first reported by von Braun (Eq. 1),
and has
1
5
been used as recently as 1986. The first run with 3a and 48% HBr
entry 4) used the same conditions as in entry 2 (1a to 1b), with only
References
(
slightly better results (77% crude 3b). Looking for a higher-boiling
organic phase (to raise the pot temperature and the reaction rate), we
chose p-xylene (bp 138°). However, -bromo-p-xylene
1
. While few uses are listed in SciFinder for 1b and 4b, thousands of
uses and reactions of 2b,3b,5b, and 6b are given, including (e.g.)
construction of functional polymeric membranes, organic
photovoltaic solar cells, liquid crystals, and bioactive materials. See
also Ref. 2.
(
lachrymator) was found in p-xylene-mediated preparations of 3b,
b, 5b, and 6b. Octane (easily recovered) was the only organic
4
phase used after this discovery. Although costs of piperidine (von
Braun starting material) and 3a are similar, the lower expense and
greater convenience of reaction of 3a with 48% HBr should greatly
2
. For assessment of some methods, see Li, H.; Liu, J.; Zhu, J.;
1
6
Wang, H. J. Korean Chem. Soc. 2011, 55, 685-690, and references
therein.
favor this method. This work appears to be its first use in 3b
1
7
synthesis.
In the preparations of 5b and 6b using p-xylene (entries 5 and 6,
Table 1), before the benzylic bromide problem was recognized,
bromoalkanols (5c < 6c) appeared in the organic phase. This
behavior, not seen in reactions of shorter chain diols, was attributed
to the increased lipophilicity of the longer chain compounds, to the
extent that a high concentration of HBraq no longer could hold them
in the aqueous phase. Greater solubility of bromoalkanols in the
organic phase could account also for the decreased yields of 5b and
3. M. Braun, Science of Synthesis, 2007, 35, 323-353.
4
5
. Chong, J. M.; Heuft, M. A.; Rabbat. P. J. Org. Chem. 2000, 65,
837-5838.
5. Segall, J.; Shorr, L. M. Process for the Preparation of Alkyl
Halides, U.S. Patent 5,138,110, August 11, 1992.
6
1
.
Wrewsky, M.; Sawaritzky, N.; Scharloff, L. Z. physik. Chem.
924, 112, 97-108.
6
b, through decreased exposure of 5c and 6c to HBraq.
th
At this point, faced also with the undesirably long reaction
times and low yields in entries 2 and 4, we took a closer look at a
7
. CRC Handbook of Chemistry and Physics, 68 Edition, 1987-88,
p. D-12.
remarkable rate acceleration achieved by “intensified azeotropic
8
8. Grosjean, C.; Parker, J.; Thirsk, C.; Wright, A. R. Org. Process
Res. Dev. 2012, 16, 781-787.
distillation”, and at the more general concept of process
1
8,19
intensification.
Consequently, the starting HBr:diol mole ratio
was raised to 6.0:1, the starting octane/diol ratio (v:w) was lowered
to ~7:1, to minimize accumulation of bromoalkanol in the organic
phase, the bath temperature was held in the range 145-150 °C, and as
9
6
. Smith, M. B.; March, J. March’s Advanced Organic Chemistry,
th Edn, p. 479, John Wiley & Sons: New Jersey, 2007.
1
0. Kloxin, C. J.; Scott. T. F.; Bowman, C. N. Macromolecules
soon as the head temperature rose 2-3° above the pure H O-octane
2
7
2009, 42, 2551-2556.
azeotrope value (89.6 °C), the distillation fractionation head was set
to total reflux. Only aqueous condensate (lower of two phases) was
tapped off, forcing the condensed octane to return to the pot via the
fractionating column. The 3b, 4b, and 6b preparations then were
repeated using these conditions; the gratifying results are shown in
Table 1 (entries 7-9). The cost of 5a dissuaded us from repeating the
1
1. Kamm, O.; Marvel, C. S. Organic Syntheses 1921, 1, 3. One use
of this procedure was found, without attribution: Ashnagar, A.;
Naseri, N. G.; Alboghobesh, S. Biosci. Biotech. Res. Asia, 2007, 4,
65-70.
5
1
2. Kurata, T.; Kurita, N. Fain Kemikaru 2008, 37, 43-49
5
b preparation.
(translation obtained).
Dibromide 4b was included in the study as another substrate of
interest in desymmetrization reactions, and as a “medium” size
1
3. Braun, J. v. Chem. Ber. 1904, 37, 3210-3213.
member of the chosen range of compounds. The first reported
2
0
14. Braun, J. v. Organic Syntheses 1929, 9, 70.
preparation of 4b used the von Braun method (65%), which was
1
5
1
used again in 1986 (61%).
Few other methods have appeared,
1
2
5. Nguyen, B. T.; Cartledge, F. K. J. Org. Chem. 1986, 51, 2206-
210.
none from diol 4a, despite its low cost availability. The optimized
HBr-diol procedure (entry 8) again favors this method over the von
Braun route.
1
6. For conversion of 3a to 3b by other means, see (a) Nguyen, H.-
P.; Kirilov, P.; Matondo, H.; Baboulène, M. J. Mol. Cat. A: Chem.
004, 218, 41-45 [IL catalysis; 60% gc yield], and Ref. 12 (HBrg;
100%; no experimental details).
The optimized procedure was applied also to preparation of 1b,
but gave no improvement in time or yield. No 1c or diol was
2
1
recovered, and H NMR analysis showed the presence of unsaturated
material, indicating that extensive elimination had occurred. Thus,

 Mekala, Shekar
Mekala, Shekar
 Hahn, Roger C.
Hahn, Roger C.