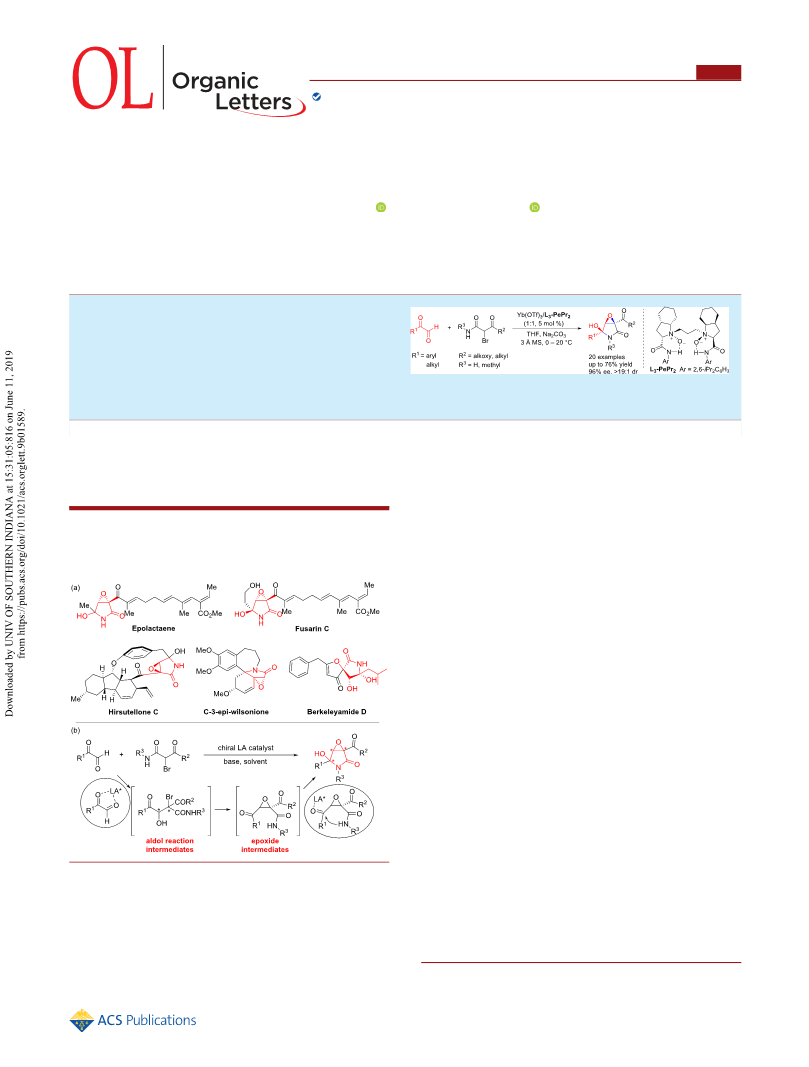
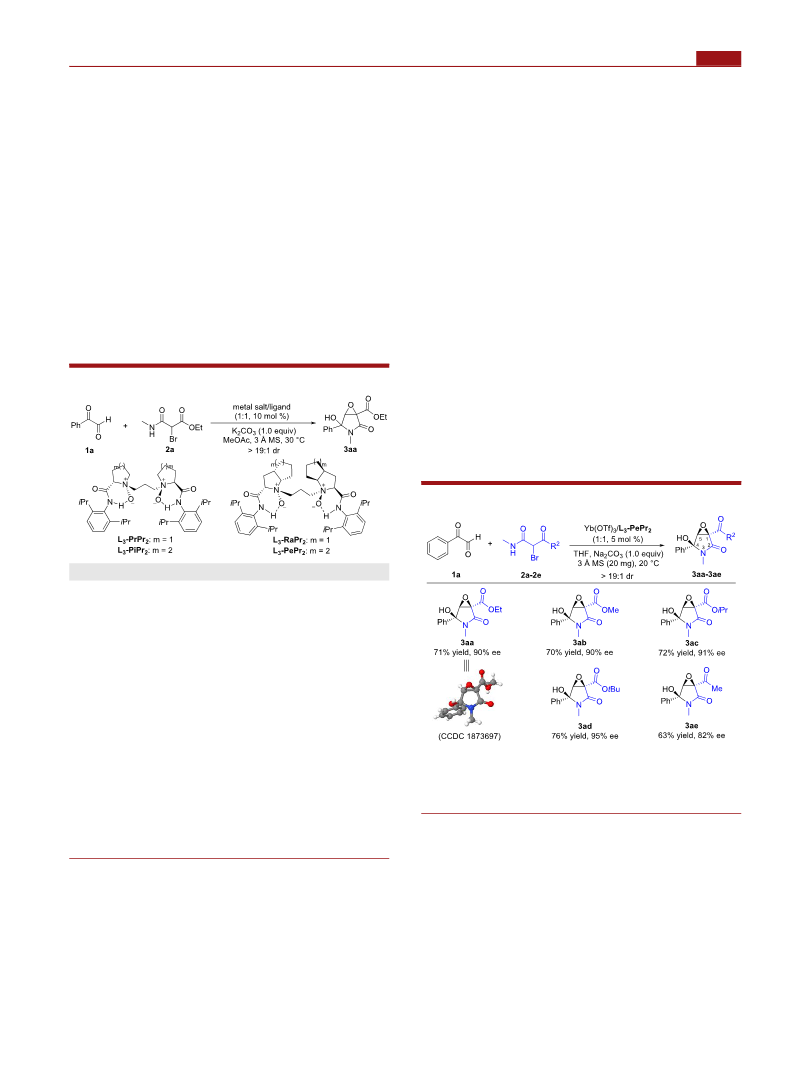
Organic Letters
Letter
3
9, 2145. (c) Arai, S.; Ishida, T.; Shioiri, T. Asymmetric Synthesis of
ACKNOWLEDGMENTS
■
α,β-Epoxysulfones under Phase-Transfer Catalyzed Darzens Reaction.
Tetrahedron Lett. 1998, 39, 8299. (d) Achard, T.; Belokon, Y.; Ilyin,
M.; Moskalenko, M.; North, M.; Pizzato, F. Enantio- and
Diastereoselective Darzens Condensation. Tetrahedron Lett. 2007,
We appreciate the National Natural Science Foundation of
China (Nos. 21890723 and 21801174) for financial support.
4
8, 2965. (e) Liu, W. J.; Lv, B. D.; Gong, L. Z. An Asymmetric
REFERENCES
■
Catalytic Darzens Reaction between Diazoacetamides and Aldehydes
Generates cis-Glycidic Amides with High Enantiomeric Purity. Angew.
Chem., Int. Ed. 2009, 48, 6503. (f) Watanabe, S.; Hasebe, R.; Ouchi,
J.; Nagasawa, H.; Kataoka, T. Enantioselective Darzens Reaction
Using Organoselenide-lithium Hydroxide Complexes. Tetrahedron
Lett. 2010, 51, 5778. (g) Rapi, Z.; Bako,
A.; Drahos, L.; Botyan
(
1) (a) Kuramochi, K.; Nagata, S.; Itaya, H.; Matsubara, Y.; Sunoki,
T.; Uchiro, H.; Takao, K.; Kobayashi, S. A Convergent Total
Synthesis of Epolactaene: An Application of the Bridgehead Oxiranyl
Anion Strategy. Tetrahedron 2003, 59, 9743. (b) Nagumo, Y.; Kakeya,
H.; Shoji, M.; Hayashi, Y.; Dohmae, N.; Oasda, H. Epolactaene Binds
Human Hsp60 Cys4 Resulting in the Inhibition of Chaperone
Activity. Biochem. J. 2005, 387, 835.
2) Song, Z.; Cox, R. J.; Lazarus, C. M.; Simpson, T. J. Fusarin C
Biosynthesis in Fusarium moniliforme and Fusarium venenatum.
ChemBioChem 2004, 5, 1196.
3) Nicolaou, K. C.; Sun, Y.; Sarlah, D.; Zhan, W.; Wu, T. R.
Bioinspired Synthesis of Hirsutellones A, B, and C. Org. Lett. 2011,
3, 5708.
4) Wang, L.; Su, H.; Yang, S.; Won, S.; Lin, C. New Alkaloids and a
Tetraflavonoid from Cephalotaxus wilsoniana. J. Nat. Prod. 2004, 67,
182.
5) Ling, T. T.; Potts, B. C.; Macherla, V. R. Concise Formal
Synthesis of (−)-Salinosporamide A (Marizomib) Using a Regio- and
Stereoselective Epoxidation and Reductive Oxirane Ring-Opening
Strategy. J. Org. Chem. 2010, 75, 3882.
́
P.; Keglevich, G.; Szo
szki, A.; Holczbauer, T. Asymmetric Phase
llosy,
̈ ̈
42
́
́
Transfer Darzens Reactions Catalyzed by D-glucose and D-mannose-
based Chiral Crown Ethers. Tetrahedron: Asymmetry 2012, 23, 489.
(
(
h) Kuang, Y. L.; Lu, Y.; Tang, Y.; Liu, X. H.; Lin, L. L.; Feng, X. M.
Asymmetric Synthesis of Spiro-epoxyoxindoles by the Catalytic
(
Darzens Reaction of Isatins with Phenacyl Bromides. Org. Lett.
2
Wang, Z.; Dai, Z. Synthesis of New Chiral Phase Transfer Catalysts
and their Application in the Asymmetric Darzens Reaction. Austin J.
Anal. Pharm. Chem. 2015, 2, 1055.
014, 16, 4244. (i) Zhao, Y.; Xu, P.; Zhang, X.; Chen, S.; Yu, Q.;
1
(
1
(
(
11) Rosen, T. Comprehensive Organic Synthesis; Trost, B. M.,
Fleming, I., Eds.; Pergamon: Oxford, 1991; Vol. 2, p 409.
12) Giubellina, N.; Mangelinckx, S.; Tornroos, K. W.; De Kimpe,
(
̈
N. Synthesis of 2-Chloro-2-imidoylaziridines via Aza-Darzens-type
Reaction of 3,3-Dichloro-1-azaallylic Anions and N-(Arylsulfonyl)-
imines. J. Org. Chem. 2006, 71, 5881.
(
6) Yamaguchi, J.; Kakeya, H.; Uno, T.; Shoji, M.; Osada, H.;
Hayashi, Y. Determination by Asymmetric Total Synthesis of the
Absolute Configuration of Lucilactaene, a Cell-Cycle Inhibitor in p53-
Transfected Cancer Cells. Angew. Chem., Int. Ed. 2005, 44, 3110.
7) Kang, T.; Jo, D.; Han, S. Six-Step Total Synthesis of Azaspirene.
J. Org. Chem. 2017, 82, 9335.
8) (a) Komori, K.; Taniguchi, T.; Mizutani, S.; Monde, K.;
Kuramochi, K.; Tsubaki, K. Short Synthesis of Berkeleyamide D and
Determination of the Absolute Configuration by the Vibrational
Circular Dichroism Exciton Chirality Method. Org. Lett. 2014, 16,
(
13) Kuang, Y. L.; Shen, B.; Dai, L.; Yao, Q.; Liu, X. H.; Lin, L. L.;
Feng, X. M. Diastereodivergent Asymmetric Michael-alkylation
Reaction Using Chiral N,N’-Dioxide/metal Complexes. Chem. Sci.
2
(
Carbonyl-Ene Reaction Catalyzed by Chiral N,N’-Dioxide-Nickel(II)
Complex: Remarkably Broad Substrate Scope. J. Am. Chem. Soc. 2008,
(
018, 9, 688.
14) (a) Zheng, K.; Shi, J.; Liu, X. H.; Feng, X. M. Asymmetric
(
1
30, 15770. (b) Wu, W. B.; Liu, X. H.; Zhang, Y. H.; Ji, J.; Huang, T.
Y.; Lin, L. L.; Feng, X. M. Chiral N,N’-Dioxide-FeCl3 Complex-
catalyzed Asymmetric Intramolecular Cannizzaro Reaction. Chem.
Commun. 2015, 51, 11646. (c) Luo, W. W.; Lin, L. L.; Zhang, Y.; Liu,
X. H.; Feng, X. M. Construction of Distant Stereocenters by
Enantioselective Desymmetrizing Carbonyl-Ene Reaction. Org. Lett.
1
386. (b) Mizutani, S.; Komori, K.; Kai, C.; Kuramochi, K.; Tsubaki,
K. The Second Generation Synthesis of (±)-berkeleyamide D.
Tetrahedron 2016, 72, 6640. (c) Kuramochi, K.; Komori, K.;
Mizutani, S.; Tsubaki, K. Syntheses of Naturally Occuring Lactams
by the Use of Darzens Reaction. Yuki Gosei Kagaku Kyokaishi 2018,
6, 218.
9) (a) Hayashi, Y.; Kanayama, J.; Yamaguchi, J.; Shoji, M.
Diastereoselective Total Synthesis of Both Enantiomers of Epolac-
taene. J. Org. Chem. 2002, 67, 9443. (b) Snider, B. B.; Neubert, B. J. A
Novel Biomimetic Route to the 3-Acyl-5-hydroxy-3-pyrrolin-2-one
and 3-Acyl-3,4-epoxy-5-hydroxypyrrolidin-2-one Ring Systems. J. Org.
Chem. 2004, 69, 8952. (c) Kuramochi, K.; Mizushina, Y.; Nagata, S.;
Sugawara, F.; Sakaguchi, K.; Kobayashi, S. Structure-activity Relation-
ships of Epolactaene Analogs as DNA Polymerases Inhibitors. Bioorg.
Med. Chem. 2004, 12, 1983. (d) Tan, Z.; Negishi, E. Selective
Synthesis of Epolactaene Featuring Efficient Construction of Methyl
2
017, 19, 3374.
15) For recent reviews on N,N′-dioxide/metal complexes, see:
a) Liu, X. H.; Lin, L. L.; Feng, X. M. Chiral N,N′-Dioxides: New
7
(
(
(
Ligands and Organocatalysts for Catalytic Asymmetric Reactions. Acc.
Chem. Res. 2011, 44, 574. (b) Liu, X. H.; Lin, L. L.; Feng, X. M. Chiral
N,N′-Dioxide Ligands: Synthesis, Coordination Chemistry and
Asymmetric Catalysis. Org. Chem. Front. 2014, 1, 298. (c) Liu, X.
H.; Zheng, H. F.; Xia, Y.; Lin, L. L.; Feng, X. M. Asymmetric
Cycloaddition and Cyclization Reaction Catalyzed by Chiral N,N′-
Dioxide-Metal Complexes. Acc. Chem. Res. 2017, 50, 2621. (d) Liu, X.
H.; Dong, S. X.; Lin, L. L.; Feng, X. M. Chiral Amino Acids-Derived
Catalysts and Ligands. Chin. J. Chem. 2018, 36, 791. For selected
examples catalyzed by N,N’-dioxide/Yb(III), see: (e) Zhang, Y.; Liao,
Y. T.; Liu, X. H.; Zhou, Y. H.; Lin, L. L.; Feng, X. M. Catalytic
Michael/Ring-Closure Reaction of α,β-Unsaturated Pyrazoleamides
with Amidomalonates: Asymmetric Synthesis of (−)-Paroxetine.
Chem. - Eur. J. 2016, 22, 15119. (f) Wang, G. J.; Tang, Y.; Liu, X.
H.; Lin, L. L.; Feng, X. M. Enantioselective Synthesis of N-H-Free
(
Z)-2-Iodo-2-butenoate and (2R,3S,4S)-2-Trimethylsilyl-2,3-epoxy-4-
methyl-γ-butyrolactone. Org. Lett. 2006, 8, 2783. (e) Rees, D. O.;
Bushby, N.; Cox, R. J.; Harding, J. R.; Simpson, T. J.; Willis, C. L.
13
15
Synthesis of [1,2- C , N]-L-Homoserine and Its Incorporation by
2
the PKS-NRPS System of Fusarium moniliforme into the Mycotoxin
Fusarin C. ChemBioChem 2007, 8, 46. (f) Han, M.; Nam, K.; Hahn,
H.; Shin, D. Unexpected Formation of New Bicyclic γ-Lactams by
Dimerization of α-Chloroacetoacetanilides. Tetrahedron Lett. 2008,
9, 5217. (g) Tanaka, K.; Kobayashi, K.; Kogen, H. Total Synthesis of
−)-L-755,807: Establishment of Relative and Absolute Config-
urations. Org. Lett. 2016, 18, 1920.
10) For representative reviews and examples, see: (a) Santos, J.;
1
(
,5-Benzothiazepines. Chem. - Eur. J. 2017, 23, 554.
16) Stierle, A.; Stierle, D.; Patacini, B. The Berkeleyamides, Amides
from the Acid Lake Fungus Peniciluum rubrum. J. Nat. Prod. 2008, 71,
56.
17) The absolute configuration of berkeleyamide D was determined
4
(
8
(
(
27
D
by comparing the specific rotation with that in ref 8a([α] = + 76.9
Retana, A.; Marigorta, E.; Vicario, J.; Palacios, F. Catalytic
Asymmetric Darzens and Aza-Darzens Reactions for the Synthesis
of Chiral Epoxides and Aziridines. ChemCatChem 2018, 10, 5092.
17
(
c 0.05, MeOH) vs [α] = +84.6 (c 0.25, MeOH)). The absolute
D
configuration of 3pf was determined by the absolute configuration of
berkeleyamide D and NOESY correlations.
(
b) Arai, S.; Shioiri, T. Catalytiv Asymmetric Darzens Condensation
under Phase-Transfer-Catalyzed Conditions. Tetrahedron Lett. 1998,
D
Org. Lett. XXXX, XXX, XXX−XXX

 Shen, Bin
Shen, Bin
 Liu, Wen
Liu, Wen
 Cao, Weidi
Cao, Weidi
 Liu, Xiaohua
Liu, Xiaohua
 Feng, Xiaoming
Feng, Xiaoming