Journal of the American Chemical Society
Article
bonding L ligands to X. Perkins, C. W.; Martin, J. C.; Arduengo, A. J.;
Lau, W.; Alegria, A.; Kochi, J. K. J. Am. Chem. Soc. 1980, 102, 7753−
7759.
resonances.
We are grateful to the personnel in the support services (mass
spectrometry, NMR spectroscopy, microanalysis) at the
University of Illinois at Urbana−Champaign. Dr. Lingyang
Zhu is especially thanked for helpful suggestions on NMR
spectroscopy. Mr. Alexander Shved is thanked for preliminary
experiments.
(18) Noth, H.; Wrackmeyer, B. In Nuclear Magnetic Resonance
̈
Spectroscopy of Boron Compounds; Diehl, P., Fluck, E., Kosfeld, R., Eds.;
NMR Basic Principles and Progress; Springer-Verlag, 1978; Vol. 14,
Chapter 7, pp 74−100.
REFERENCES
■
(19) Furthermore, the 19F NMR signals remained as sharp singlets
indicating that no dynamic process was evident. A similar result was
observed for neopentyl 4-fluorophenylboronate ester 22. Where a
discrete intermediate was not observed; however, cross-coupling was
observed.
(1) (a) Roughley, S. D.; Jordan, A. M. J. Med. Chem. 2011, 54, 3451−
3479. (b) Brown, D. G.; Bostrom, J. J. Med. Chem. 2016, 59, 4443−4458.
̈
(2) Hall, D. G. Structure, Properties, and Preparation of Boronic Acid
Derivatives. Boronic Acids; Wiley-VCH Verlag GmbH & Co. KGaA:
2011; pp 1−133.
(20) This conclusion found support in the energies of the
computationally derived structures wherein a 0.97 kcal/mol difference
favoring 19 was calculated (see Section 4.4.5). The computationally
predicted Keq = 0.134 was very close to the experimentally observed
equilibrium concentration Keq = 0.12, (Keq = [19′]/[19]).
(21) Furthermore, the 19F NMR chemical shifts for complex 6 in
THF/CH3OH were measured to be −121.01 ppm (Fa) and − 118.33
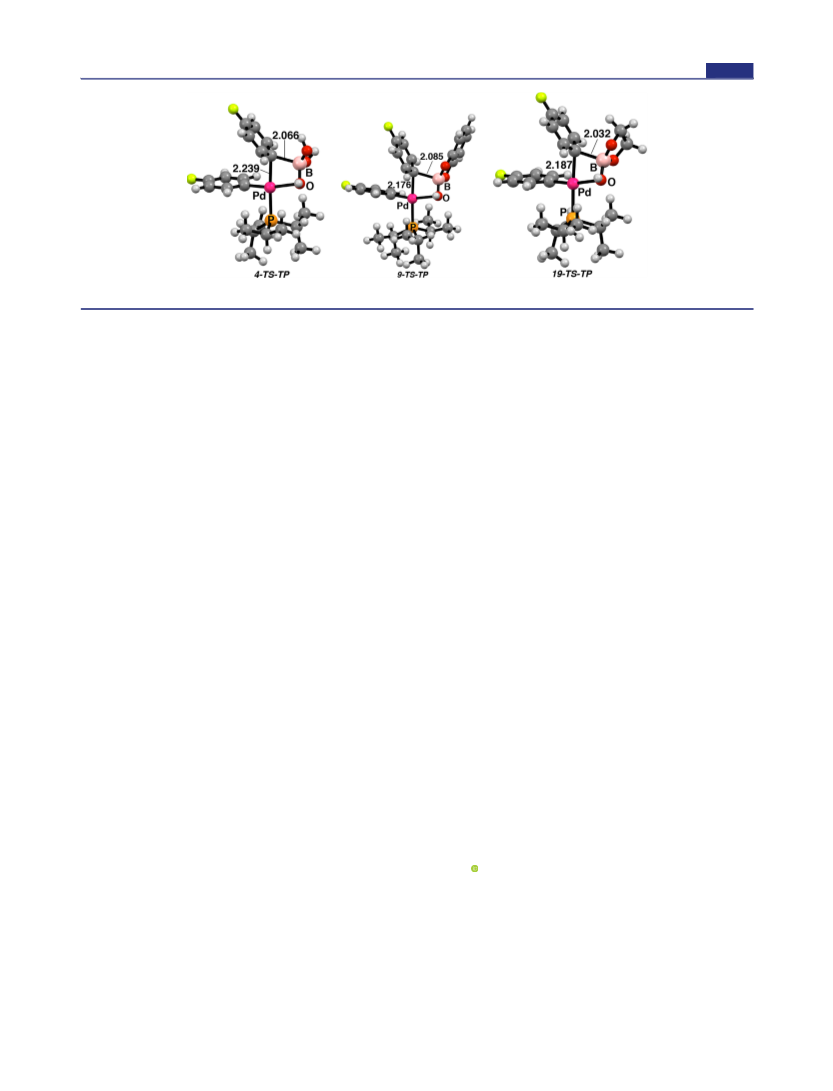
ppm (Fb) suggesting a solvent effect for the measurement of these
chemical shifts.
(22) Because of peak broadening in certain 8-B-4 complexes and rapid
formation of cross-coupling product 5 as a strong, sharp signal, the
formation of 5 was found to be more reliable for determination of the
rate.
(23) The similarity of rates for appearance of 5 and consumption of 4
suggests that transmetalation is the rate-determining step for this
process.
(24) The sigmoidal curves made extraction of a comparable rate
constant problematic and therefore were not fitted.
(25) Driver, M. S.; Hartwig, J. F. Organometallics 1997, 16, 5706−7515.
(26) The complexity of this system lends itself to other possible
interpretations. We offer an explanation that is most consistent with all
of the available data, but we cannot unambiguously exclude the
possibility that the diminished rate of 21 relative to complex 4, is the
result of the inherent reactivity of complex 21.
(27) The inability to detect a discrete intermediate with boroxine 28
results from the multitude of boron-containing oligomers that precluded
clear assignment of structure.
(3) (a) Miyaura, N.; Yamada, K.; Suzuki, A. Tetrahedron Lett. 1979, 20,
3437−3440. (b) Miyaura, N.; Suzuki, A. J. Chem. Soc., Chem. Commun.
1979, 866−867.
(4) Miyaura, N.; Yanagi, T.; Suzuki, A. Synth. Commun. 1981, 11, 513−
519.
(5) Larsen, R. D.; King, A. O.; Chen, C. Y.; Corley, E. G.; Foster, B. S.;
Roberts, F. E.; Yang, R. C.; Lieberman, D. R.; Reamer, R. A.; Tschaen, D.
M.; Verhoeven, T. R.; Reider, P. J.; Lo, Y. S.; Rossano, L. T.; Brookes, A.
S.; Meloni, D.; Moore, J. R.; Arnett, J. F.; et al. J. Org. Chem. 1994, 59,
6391−6394.
(6) Dumrath, A.; Lubbe, C.; Beller, M. Palladium-Catalyzed Cross-
̈
Coupling Reactions − Industrial Applications. Palladium-Catalyzed
Coupling Reactions; Wiley-VCH Verlag GmbH & Co. KGaA: 2013; pp
445−489.
(7) Kruger, A. W.; Rozema, M. J.; Chu-Kung, A.; Gandarilla, J.; Haight,
A. R.; Kotecki, B. J.; Richter, S. M.; Schwartz, A. M.; Wang, Z. Org.
Process Res. Dev. 2009, 13, 1419−1425.
(8) Stewart, G. W.; Brands, K. M. J.; Brewer, S. E.; Cowden, C. J.;
Davies, A. J.; Edwards, J. S.; Gibson, A. W.; Hamilton, S. E.; Katz, J. D.;
Keen, S. P.; Mullens, P. R.; Scott, J. P.; Wallace, D. J.; Wise, C. S. Org.
Process Res. Dev. 2010, 14, 849−858.
(9) De Koning, P. D.; McAndrew, D.; Moore, R.; Moses, I. B.; Boyles,
D. C.; Kissick, K.; Stanchina, C. L.; Cuthbertson, T.; Kamatani, A.;
Rahman, L.; Rodriguez, R.; Urbina, A.; Sandoval, A.; Rose, P. R. Org.
Process Res. Dev. 2011, 15, 1018−1026.
(10) (a) Cox, P. A.; Leach, A. G.; Campbell, A. D.; Lloyd-Jones, G. C. J.
Am. Chem. Soc. 2016, 138, 9145−9157. (b) Molloy, J. J.; Clohessy, T. A.;
Irving, C.; Anderson, N. A.; Lloyd-Jones, G. C.; Watson, J. B. Chem. Sci.
2017, 8, 1551−1559. (c) Cox, P. A.; Reid, M.; Leach, A. G.; Campbell, A.
D.; King, E. J.; Lloyd-Jones, G. C. J. Am. Chem. Soc. 2017, 139, 13156−
13165.
(11) The masking term was defined by Lloyd Jones and co-workers:
masking is distinct from “protection” because protection requires that a
deprotection step take place prior to use. Lennox, A. J.; Lloyd-Jones, G.
C. Isr. J. Chem. 2010, 50, 664−674.
(12) (a) Lennox, A. J.J.; Lloyd-Jones, G. C. J. Am. Chem. Soc. 2012, 134,
7431−7441. (b) Lennox, A. J. J.; Lloyd-Jones, G. C. Angew. Chem., Int.
Ed. 2012, 51, 9385−9388. (c) Butters, M.; Harvey, J. N.; Jover, J.;
Lennox, A. J. J.; Lloyd-Jones, G. C.; Murray, M. Angew. Chem., Int. Ed.
2010, 49, 5156−5160. (c1) Lennox, A. J. J.; Lloyd-Jones, G. C. Angew.
Chem., Int. Ed. 2013, 52, 7362−7370.
(13) Gonzalez, J. A.; Ogba, O. M.; Morehouse, G. F.; Rosson, N.;
Houk, K. N.; Leach, A. G.; Cheong, P. H. Y.; Burke, M. D.; Lloyd-Jones,
G. C. Nat. Chem. 2016, 8, 1067−1075.
(14) Lennox, A. J.J.; Lloyd-Jones, G. C. Chem. Soc. Rev. 2014, 43, 412−
443.
(28) (a) Carrow, B. P.; Hartwig, J. F. J. Am. Chem. Soc. 2011, 133,
2116−2119. (b) Amatore, C.; Jutand, A.; Le Duc, G. Chem. - Eur. J.
2011, 17, 2492−2503. (c) Amatore, C.; Jutand, A.; Le Duc, G. Angew.
Chem. 2012, 124, 1408−1411. (d) Schmidt, A. F.; Kurokhtina, A. A.;
Larina, E. V. Russ. J. Gen. Chem. 2011, 81, 1573−1574.
(29) We cannot unambiguously exclude the possibility that hydroxide
forms a palladium hydroxide complex. These results do not contradict
the conclusions of the studies described by Hartwig, and Amatore and
Jutand (ref 28.)
(30) Sperger, T.; Sanhueza, I.; Kalvet, I.; Schoenebeck, F. Chem. Rev.
2015, 115, 9532−9586 and references therein..
(31) García-Melchor, M.; Braga, A. A. C.; Lledos
́
, A.; Ujaque, G.;
Maseras, F. Acc. Chem. Res. 2013, 46, 2626−2634.
(32) Ortuno, M. A.; Lledos
2014, 6, 3132−3138.
(33) Calculations with the B3LYP-D3 and M06 functionals are given in
́
, A.; Maseras, F.; Ujaque, G. ChemCatChem
̃
́
(34) Besora, M.; Braga, A.; Ujaque, G.; Maseras, F.; Lledos, A. Theor.
Chem. Acc. 2011, 128, 639−646.
(35) Appleton, T. G.; Clark, H. C.; Manzer, L. E. Coord. Chem. Rev.
1973, 10, 335−422.
(15) (a) Thomas, A. A.; Denmark, S. E. Science 2016, 352, 329−332.
(b) Thomas, A. A.; Wang, H.; Zahrt, A. F.; Denmark, S. E. J. Am. Chem.
Soc. 2017, 139, 3805−3821. (c) Thomas, A. A.; Denmark, S. E. Ernest L.
Eliel, a Physical Organic Chemist with the Right Tool for the Job: Rapid
Injection Nuclear Magnetic Resonance. In Stereochemistry and Global
Connectivity: The Legacy of Ernest Eliel; Cheng, H. N., Ed.; ACS
Symposium Series; American Chemical Society: Washington, DC, 2018.
(16) This classification designates N-X-L species where N is the
number of formally valence-shell electrons about atom X, involved in
(36) In ongoing studies, all of these contributing factors will be
examined in a more thorough fashion by a comprehensive “energy
decomposition analysis” of the intrinsic reaction coordinates of at least
these three reactants. Hopffgarten, M. v.; Frenking, G. WIREs Comput.
Mol. Sci. 2012, 2, 43−62.
(37) Close inspection of the energies of the Prod-TP structures in
Figure 16 shows that the diarylpalladium species 18-Prod-TP is less
O
J. Am. Chem. Soc. XXXX, XXX, XXX−XXX

 Thomas, Andy A.
Thomas, Andy A.
 Zahrt, Andrew F.
Zahrt, Andrew F.
 Delaney, Connor P.
Delaney, Connor P.
 Denmark, Scott E.
Denmark, Scott E.