
Biochemical Pharmacology p. 1377 - 1385 (2006)
Update date:2022-08-16
Topics:
 Narimatsu, Shizuo
Narimatsu, Shizuo
 Yonemoto, Rei
Yonemoto, Rei
 Saito, Keita
Saito, Keita
 Takaya, Kazuo
Takaya, Kazuo
 Kumamoto, Takuya
Kumamoto, Takuya
 Ishikawa, Tsutomu
Ishikawa, Tsutomu
 Asanuma, Masato
Asanuma, Masato
 Funada, Masahiko
Funada, Masahiko
 Kiryu, Kimio
Kiryu, Kimio
 Naito, Shinsaku
Naito, Shinsaku
 Yoshida, Yuzo
Yoshida, Yuzo
 Yamamoto, Shigeo
Yamamoto, Shigeo
 Hanioka, Nobumitsu
Hanioka, Nobumitsu
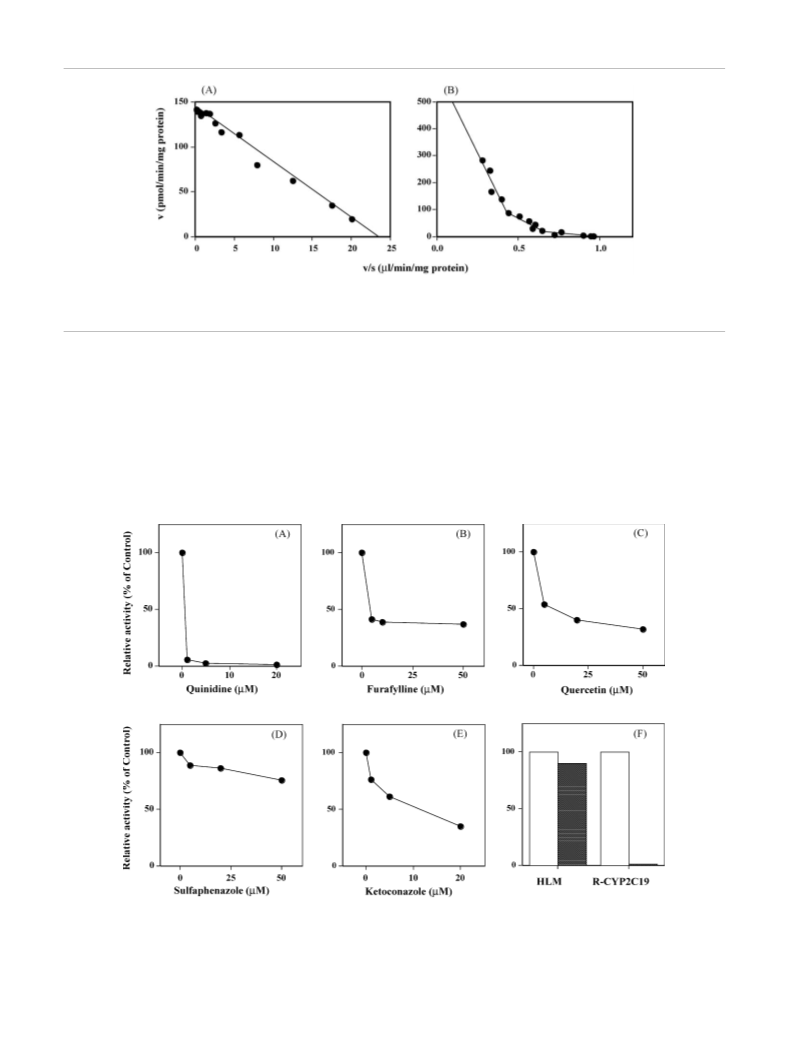
In vitro quantitative studies of the oxidative metabolism of (5-methoxy-N,N-diisopropyltryptamine, 5-MeO-DIPT, Foxy) were performed using human liver microsomal fractions and recombinant CYP enzymes and synthetic 5-MeO-DIPT metabolites. 5-MeO-DIPT was mainly oxidized to O-demethylated (5-OH-DIPT) and N-deisopropylated (5-MeO-IPT) metabolites in pooled human liver microsomes. In kinetic studies, 5-MeO-DIPT O-demethylation showed monophasic kinetics, whereas its N-deisopropylation showed triphasic kinetics. Among six recombinant CYP enzymes (CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6 and CYP3A4) expressed in yeast or insect cells, only CYP2D6 exhibited 5-MeO-DIPT O-demethylase activity, while CYP1A2, CYP2C8, CYP2C9, CYP2C19 and CYP3A4 showed 5-MeO-DIPT N-deisopropylase activities. The apparent Km value of CYP2D6 was close to that for 5-MeO-DIPT O-demethylation, and the Km values of other CYP enzymes were similar to those of the low-Km (CYP2C19), intermediate-Km (CYP1A2, CYP2C8 and CYP3A4) and high-Km phases (CYP2C9), respectively, for N-deisopropylation in human liver microsomes. In inhibition studies, quinidine (1 μM), an inhibitor of CYP2D6, almost completely inhibited human liver microsomal 5-MeO-DIPT O-demethylation at a substrate concentration of 10 μM. Furafylline, a CYP1A2 inhibitor, quercetin, a CYP2C8 inhibitor, sulfaphenazole, a CYP2C9 inhibitor and ketoconazole, a CYP3A4 inihibitor (5 μM each) suppressed about 60%, 45%, 15% and 40%, respectively, of 5-MeO-DIPT N-deisopropylation at 50 μM substrate. In contrast, omeprazole (10 μM), a CYP2C19 inhibitor, suppressed only 10% of N-deisopropylation by human liver microsomes, whereas at the same concentration the inhibitor suppressed the reaction by recombinant CYP2C19 almost completely. These results indicate that CYP2D6 is the major 5-MeO-DIPT O-demethylase, and CYP1A2, CYP2C8 and CYP3A4 are the major 5-MeO-DIPT N-deisopropylase enzymes in the human liver.
View More







Zhejiang Kangfeng Chemical Co.,LTD.
Contact:+86-579-86709687
Address:Xueshizhai Industrial Zone, Weishan Town,Dongyang City, Zhejiang Province ,China
Sichuan Sangao Biochemical Co., Ltd
Contact:+86-28-84874233
Address:19F, Bldg.2, Shudu Center, Tianfu 2nd St., Hi-tech zone, Chengdu 610041, Sichuan Province, China.
Shandong Ailitong New Material Co.,Ltd
Contact:+86-536-3226266
Address:zhongjia village, putong town , qingzhou city,Shandong Province,China
Jiangxi Lanqi Fine Chemical S&T Co., Ltd.
Contact:+86-21-64891143
Address:XinJiShan Industrial Area, Zhangshu City, JiangXi Province, China
Contact:0571-
Address:zhejing
Doi:10.1002/anie.200801426
(2008)Doi:10.1039/b006049f
(2000)Doi:10.1021/ol702078t
(2007)Doi:10.1039/C29710000246
(1971)Doi:10.1002/hlca.19500330735
(1950)Doi:10.1021/ja01020a021
(1968)