
Journal of Molecular Catalysis A: Chemical p. 76 - 82 (2016)
Update date:2022-08-17
Topics:
 Meng, Qingwei
Meng, Qingwei
 Zheng, Hongyan
Zheng, Hongyan
 Zhu, Yulei
Zhu, Yulei
 Li, Yongwang
Li, Yongwang
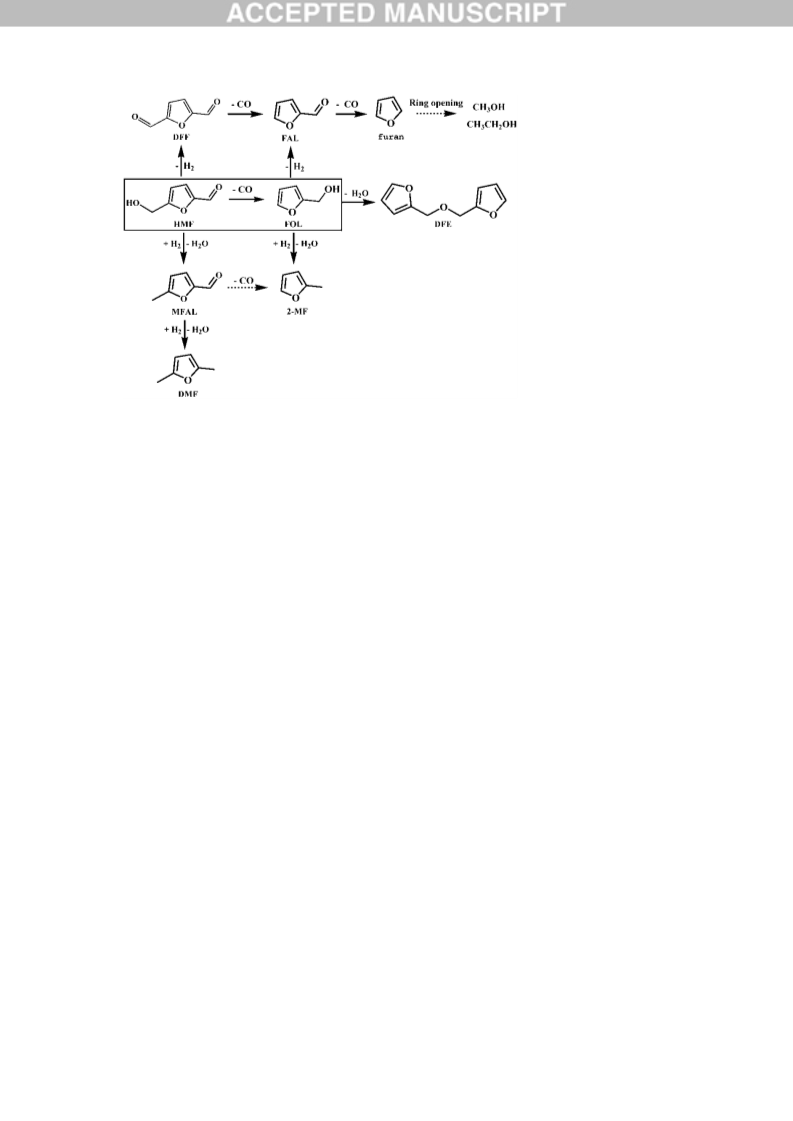
An extensive product distribution is firstly examined in the process of 5-hydroxymethylfurfural (HMF) decarbonylation over Pd-based catalysts and some interesting results are obtained. The main side reactions are due to the high activity of the furan ring-branched hydroxymethyl, which could go through hydrogenolysis, dehydrogenation and etherification. The H2 source was carefully explored and determined to be the hydroxymethyl dehydrogenation. The reactivity of the main intermediates was separately investigated and their evolution pathway was obtained. Noticeably, it is demonstrated that the elimination of the furanic ring-branched hydroxymethyl (in HMF or furfuryl alcohol) is completed by sequential dehydrogenation and decarbonylation via the intermediate of aldehyde (2, 5-diformylfuran or furfural). A comprehensive reaction pathway for HMF decarbonylation is proposed, which is significant for designing highly selective decarbonylation catalysts.
View More























Contact:86-21 60347964
Address:No.1304, West Meilong Road, Minhang District, Shanghai, China
Contact:+86-577-65618087-605
Address:Room 402, Unit 4 Xinhu Bldg. Waitan Ruian City, Zhejiang China.
Weifang Arylchem Chemical Co., LTD
Contact:86-536-5217866
Address:Development Zone, Shouguang, Shandong Province
Contact:+86-21-61318535
Address:Building 29,No.2139 Xizha Road, Fengxian District, Shanghai
Zhejiang Golden-Shell Pharmaceutical Co.,Ltd.
Contact:+86-576-87501888 / 87501988
Address:No.89 Zhongxing Road. Li'ao, Yuhuan, Zhejiang, China
Doi:10.1021/jo01269a111
(1968)Doi:10.1039/C19680000440
(1968)Doi:10.1177/1747519819901251
(2020)Doi:10.1246/cl.1981.1169
(1981)Doi:10.1016/j.apcata.2013.05.022
(2013)Doi:10.1016/j.phytochem.2012.08.019
(2013)