Journal of Sulfur Chemistry
7
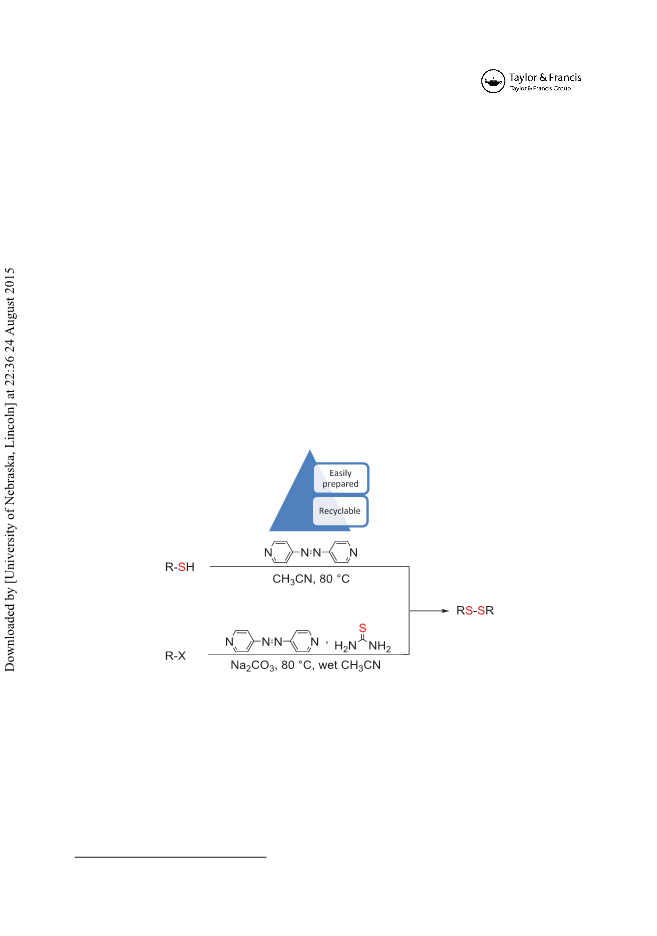
in refluxing CH3CN (4 mL) was treated with 4,4’-azopyridine (1.1 mmol, 0.202 g). The resulting
red solution was stirred at 80°C for 2 h. After decolorization of the red solution (indication of
completion of the reaction) and disappearance of benzyl mercaptan on TLC, the reaction mixture
was filtered to remove the hydrazine by-product. The filtrate was washed twice with 10% NaOH
solution (8 mL), then with saturated brine and was dried over anhydrous Na2SO4. Removal of
the volatile compounds and solvent afforded a viscous oil. The product was purified by short-
column chromatography on silica gel eluted with n-hexane. Benzyl disulfide was obtained as
white crystals and its 1H NMR and 13C NMR agreed well with the reported values.
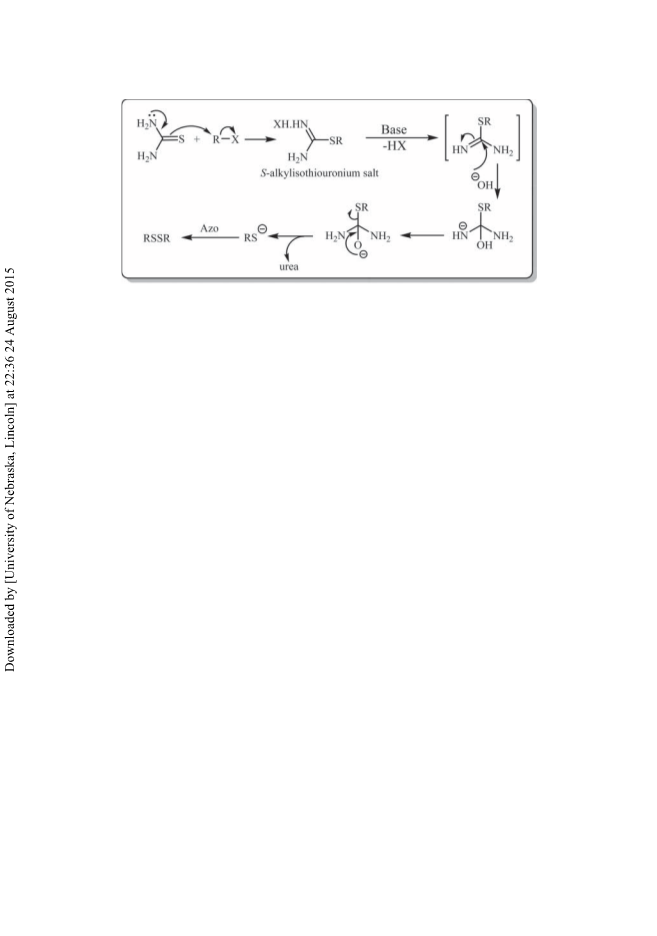
Typical procedure for direct conversion of benzyl chloride to benzyl disulfides using thiourea
and 4,4ꢀ-azopyridine in CH3CN: To a solution of thiourea (2.1 mmol, 0.160 g) and benzyl chlo-
ride (2 mmol, 0.23 mL) in wet CH3CN (3 mL CH3CN + 0.2 mL H2O), 4,4ꢀ-azopyridine (1.1
mmol, 0.202 g) and Na2CO3 (3 mmol, 0.318 g) were added. The mixture was stirred magneti-
cally at 80°C. The progress of the reaction was monitored by TLC or GC until the benzyl chloride
was consumed. After completion of the reaction, the mixture was filtered through a sintered glass
funnel to remove the produced pyridine hydrazine. The solvent was evaporated under reduced
pressure and the so-obtained residue was purified by flash chromatography on silica gel with
petroleum ether as eluent to provide benzyl disulfide.
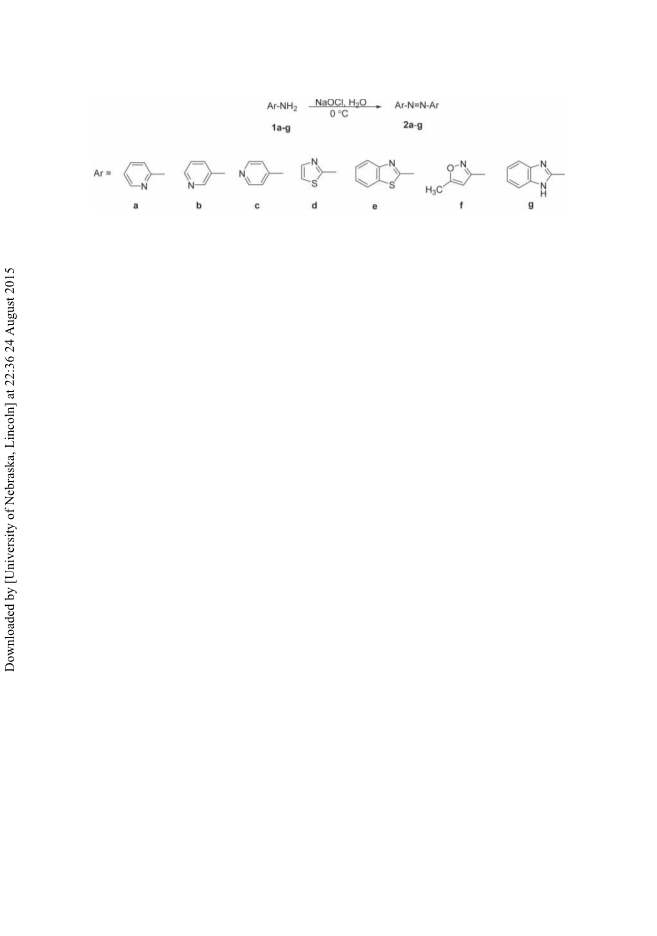
Oxidation of 4,4ꢀ-pyridinehydrazine to 4,4ꢀ-azopyridine by iodosobenzene diacetate
PhI(OAc)2: To a 50 mL flask equipped with a magnetic stirrer containing pyridinehydrazine
(0.186 g, 1.0 mmol) in DMSO (5 mL), iodosobenzene diacetate (0.322 g, 1.0 mmol) was added
in one portion and the mixture was stirred for 6 h at room temperature. H2O (20 mL) was then
added and the reaction solution was extracted with EtOAc (4 × 15 mL). The organic extracts
were combined together and dried over anhydrous sodium sulfate. Upon concentrating the solu-
tion under vacuum, azopyridine (2c) was precipitated as orange crystals (134 mg, 73%). All
the products are known compounds and were characterized by the comparison of their physical
and spectral data with those reported in the literature. Selected spectral data for representative
disulfides:
1
Dipropyl disulfide (4a) yellow liquid; H NMR (250 MHz, CDCl3): δ 0.96 (t, J = 7.1 Hz,
6H), 1.83 (sext, J = 7.1 Hz, 4H), 2.59 (t, J = 7.1 Hz, 4H); 13C NMR (62.5 MHz, CDCl3): δ
41.5, 22.7, 13.0. Anal. Calcd. C6H14S2: C, 47.95%; H, 9.39%; S, 42.66%. Found: C, 48.09%, H,
9.32%; S, 42.59%.
Dibutyl disulfide (4b) colorless oil; 1H NMR (250 MHz, CDCl3): δ 0.93 (t, J = 7.5 Hz, 6H),
1.44–1.51 (m, 8H), 2.61 (t, J = 7.7 Hz, 4H); 13C NMR (62.5 MHz, CDCl3): δ 39.2, 31.6,
21.4, 13.7. Anal. Calcd. C8H18S2: C, 53.88%; H, 10.17%; S, 35.95%. Found: C, 54.02%; H,
10.20%; S, 35.78%.
1
Dipentyl disulfide (4c) colorless oil; H NMR (250 MHz, CDCl3): δ 0.93 (t, J = 7.2 Hz,
6H), 1.36–1.41 (m, 8H), 1.61–1.1.68 (m, 4H), 2.66 (t, J = 7.4 Hz, 4H); 13C NMR (62.5 MHz,
CDCl3): δ 39.4, 29.5, 29.0, 22.1, 14.2. Anal. Calcd. C10H22S2: C, 58.19%; H, 10.74%; S, 31.06%.
Found: C, 58.10%; H, 10.67%; S, 31.23%.
Dioctyl disulfide (4d) colorless oil; 1H NMR (250 MHz, CDCl3): δ 0.79–0.85 (t, J = 6.6 Hz,
6H), 1.09–1.37 (m, 20H), 1.53–1.68 (m, 4H), 2.62 (t, J = 7.4 Hz, 4H); 13C NMR (62.5 MHz,
CDCl3): δ 39.2, 33.9, 32.7, 31.9, 29.2, 28.8, 22.6, 14.0. Anal. Calcd. C16H34S2: C, 66.14%; H,
11.79%; S, 22.07%. Found: C, 66.08%; H, 11.89%; S, 22.03%.
Dicyclohexyl disulfide (4f ) colorless oil; 1H NMR (250 MHz, CDCl3): δ 1.16–1.27 (m, 10H),
1.53–1.56 (m, 2H), 1.68–1.74 (m, 4H), 1.93–1.98 (m, 4H), 2.58–2.61 (m, 2H); 13C NMR (62.5
MHz, CDCl3): δ 49.5, 33.1, 26.0, 25.6. Anal. Calcd. C12H22S2: C, 62.55%; H, 9.62%; S, 27.83%.
Found: C, 62.66%; H, 9.53%; S, 27.81%.
1
Dibenzyl disulfide (4g) white crystal; H NMR (250 MHz, CDCl3) δ 3.65 (s, 4H), 7.16–
7.54 (m, 10H); 13C NMR (62.5 MHz, CDCl3) δ 137.3, 129.8, 128.2, 128.0, 43.9. Anal. Calcd.
C14H14S2: C, 68.25%; H, 5.73%; S, 26.02%. Found: C, 68.41%; H, 5.79%; S, 25.80%.

 Khalili, Dariush
Khalili, Dariush
 Iranpoor, Nasser
Iranpoor, Nasser
 Firouzabadi, Habib
Firouzabadi, Habib