Chemical Papers
11–14). However, aliphatic anhydrides, such as glutaric
anhydride and succinic anhydride, gave poor results under
obtained by extending reaction time to 10 h (Table 2, entries
15, 16). In these cases, dicarboxylic acids as byproducts
were generated, leading to the decrease of yields of the imi-
des. These results indicated that various anhydrides except
for aliphatic anhydrides could readily react with urea to give
their corresponding imides in ChCl/urea. It should be noted
that the yield in the second run is higher than that in the frst
run in all the cases, which can be ascribed to the slight solu-
bility of cyclic imides in ChCl/urea DES, and the dissolved
products can be extruded in the second run.
General reaction procedure
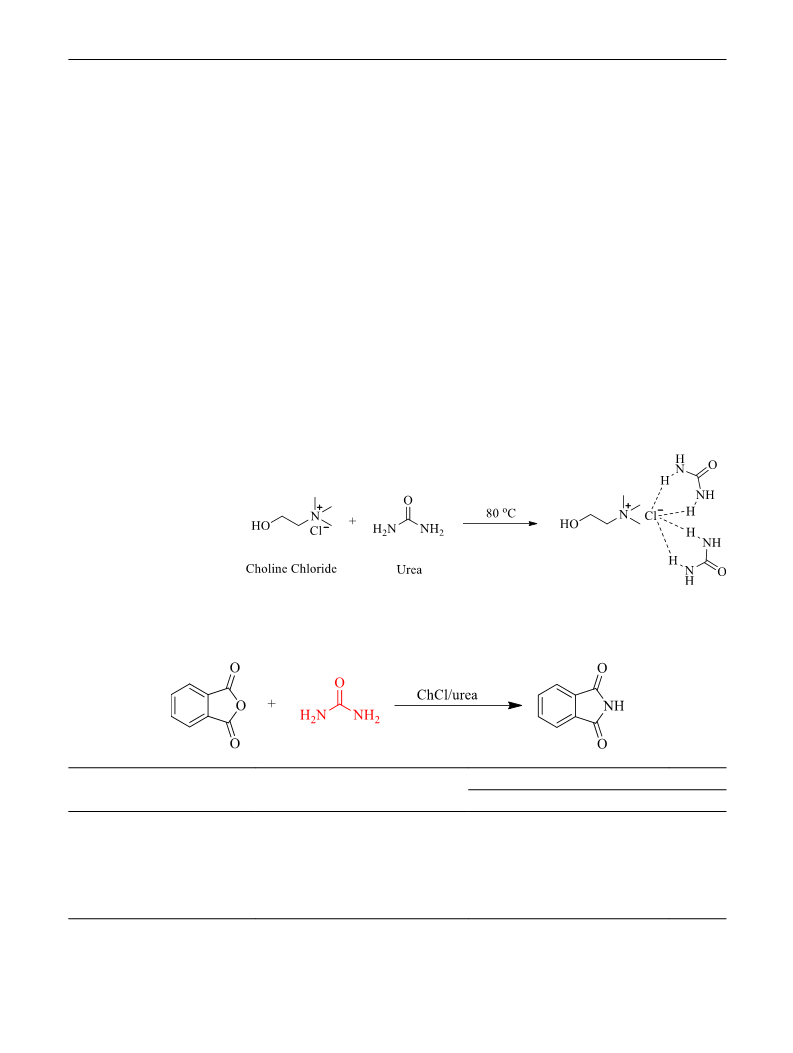
DES ChCl/urea was synthesized as described in literature
(Abbott et al. 2004). Briefy, choline chloride and urea with
a clear and transparent solution was formed (Fig. 1). The
obtained DES was used without additional purifcation.
Into a 25-mL round-bottom fask were added ChCl/urea
(5.19 g, 20 mmol), phthalic anhydride (1.48 g, 10 mmol) and
urea (0.60 g, 10 mmol) in successive, then the mixture was
heated at 140 °C for 1 h under vigorous stirring. The reac-
tion progress was monitored by GC analysis. After reaction,
the reaction mixture was cooled to room temperature, fol-
lowed by addition of 2 mL of water. The DES was dissolved
and the product was precipitated. The solid was collected
by fltration and washed thoroughly with water. The white
solid was dried thoroughly to aford the product in a yield of
84%. The DES was recovered by evaporation of water under
vacuum, and subjected to next run. The product was received
in a yield of yield of 95% in the second run.
After separation of product, the fltrate was evaporated
under vacuum to remove the water added in the separa-
tion process, and ChCl/urea was recovered directly. The
mal conditions. As shown in Fig. 2, the yield of phthal-
imide increased gradually with the recycle times of ChCl/
urea, which was due to the precipitation of the product
dissolved in the DES in previous runs. In the ffth run, the
yield reached up to 100%. The results indicated that ChCl/
urea could be readily recovered and recycled without any
deterioration.
Results and discussion
A possible reaction mechanism has been proposed as
shown in Fig. 3. Initially, DES forms hydrogen bond with
the carbonyl group of phthalic anhydride, which increases
the electrophilicity of the carbonyl group in intermediate
A, thus further promotes the nucleophilic attack of urea
on the carbonyl group to form intermediate B. Finally, B
undergoes dual intramolecular nucleophilic attacks to give
phthalimide with release of one molecule of ammonia and
carbon dioxide.
Initial experiments with phthalic anhydride as the model
substrate indicated the reaction in ChCl/urea proceeding
smoothly. Only reaction temperature had big efect on the
reaction. The results are shown in Table 1. It is worth noting
that the product phthalimide was not precipitated completely
in the frst run of ChCl/urea due to its solubility in the sys-
tem, which led to lower isolated yield of phthalimide than
generated, but the dissolved phthalimide could precipitate
gradually in the successive runs. Both the conversion of
phthalic anhydride and the yield of phthalimide increased
with the increase of temperature from 110 to 160 °C, and
the yield of phthalimide reached up to 95% at 140 °C in the
second run of ChCl/urea (Table 1, entry 4). With further
further, and phthalimide was almost quantitatively obtained
at 160 °C (Table 1, entry 6). However, temperature above
recycling of ChCl/urea difcult. Therefore, later reactions
were conducted at 140 °C.
100
80
60
40
20
0
After the determination of reaction temperature, the sub-
strate scope was investigated to demonstrate the generality
of this strategy. As shown in Table 2, phthalimide and its
gave excellent yields of their target products (Table 2, entries
1–7). 1,8-Naphthalic anhydrides and 3,3′,4,4′-biphenyltet-
racarboxylic dianhydride also aforded their corresponding
imides in excellent yields (Table 2, entries 8–10). Good
aliphatic cyclic anhydrides as substrates (Table 2, entries
1
2
3
4
5
Running times
Fig. 2 Recycling of ChCl/urea
1 3

 Liu, Luxiao
Liu, Luxiao
 Zhang, Hong-Yu
Zhang, Hong-Yu
 Yin, Guohui
Yin, Guohui
 Zhang, Yuecheng
Zhang, Yuecheng
 Zhao, Jiquan
Zhao, Jiquan