6
70
Can. J. Chem. Vol. 77, 1999
3
1
1
1
1
4
2
1
2
4
3
1
2
4
1
7
2
3
7
2
2
4
1
4
43(7), 305(8), 205(8), 147(10), 73(100); 2: 210 (M, 24),
95(14), 180(100), 165(8), 149(8), 73(13); 2.02: 205 (M –
5, 12), 177(9), 147(81), 73(100); 2.03: 261 (M – 15, 7%),
86(3), 147(100), 129(7), 73(79); 2.04: 259 (M – 15, 9%),
84(4), 147(100), 97(8), 73(41); 2.06: 418 (M, 37%),
03(16), 388(23), 314(4), 287(8), 179(6), 165(3), 73(100);
.07: 476 (M, 15%), 461(2), 358(13), 299(4), 207(3),
79(4), 147(8), 73(100); 2.08: 346(43%), 331(29), 316(56),
87(15), 241(47), 213(13), 165(11), 147(11), 73(100); 2.09:
04 (M, 46%), 389(11), 374(13), 300(8), 193(21), 73(100);
.03: 238 (M, 5%), 223(100), 208(34), 179(8), 165(7),
51(7), 73(14); 3.04: 311 (M – 15, 75%), 281(8), 238(10),
07(11), 179(11), 151(12), 133(14), 73(100); 3.05: 436 (M,
%), 305(100), 215(4), 188(6), 115(5), 73(39); 4: 418 (M,
00%), 403(35), 388(79), 373(10), 358(23), 193(8), 179(14),
3(74); 4.02: 247 (M – 15, 15%), 147(100), 73(73); 4.03:
45 (M – 15, 100%), 147(58), 143(17), 115(9), 73(98); 4.06:
35 (M – 15, 7%), 245(13), 233(24), 147(63), 133(10),
3(100); 4.07: 312 (M, 67%), 297(100), 282(35), 267(64),
53(41), 223(53), 193(20), 126(24), 73(76); 4.08: 396 (M,
5%), 338(34), 308(11), 209(90), 179(40), 147(9), 73(100);
.09: 346 (M, 20%) 331(12), 316(48), 301(16), 241(13),
79(13), 115(11), 73(100); 5: 432 (M, 68%), 417(27),
02(35), 329(8), 299(7), 193(16), 179(7), 73(100); 5.03: 490
Fig. 1. The reactivity (reflected by the amount of remaining
starting material) of the various residual Kraft lignin model
compounds examined during this work, in the presence and
absence of various metal ions: (a) displays the data as a function
of the individual metal ions added; (b) displays the data as a
function of the individual model compounds examined.
(
M, 16%), 475(3), 372(15), 360(9), 195(19), 180(13),
1
2
1
7
1
47(6), 73(100); 5.04: 410 (M, 17%), 352(6), 277(15),
20(11), 208(13), 193(31), 147(19), 73(100); 5.05: 450 (M,
7%), 435(14), 390(10), 243(14), 217(10), 251(26), 147(11),
3(100); 5.06: 538 (M, 3%), 404(17), 263(32), 231(29),
47(14), 73(100).
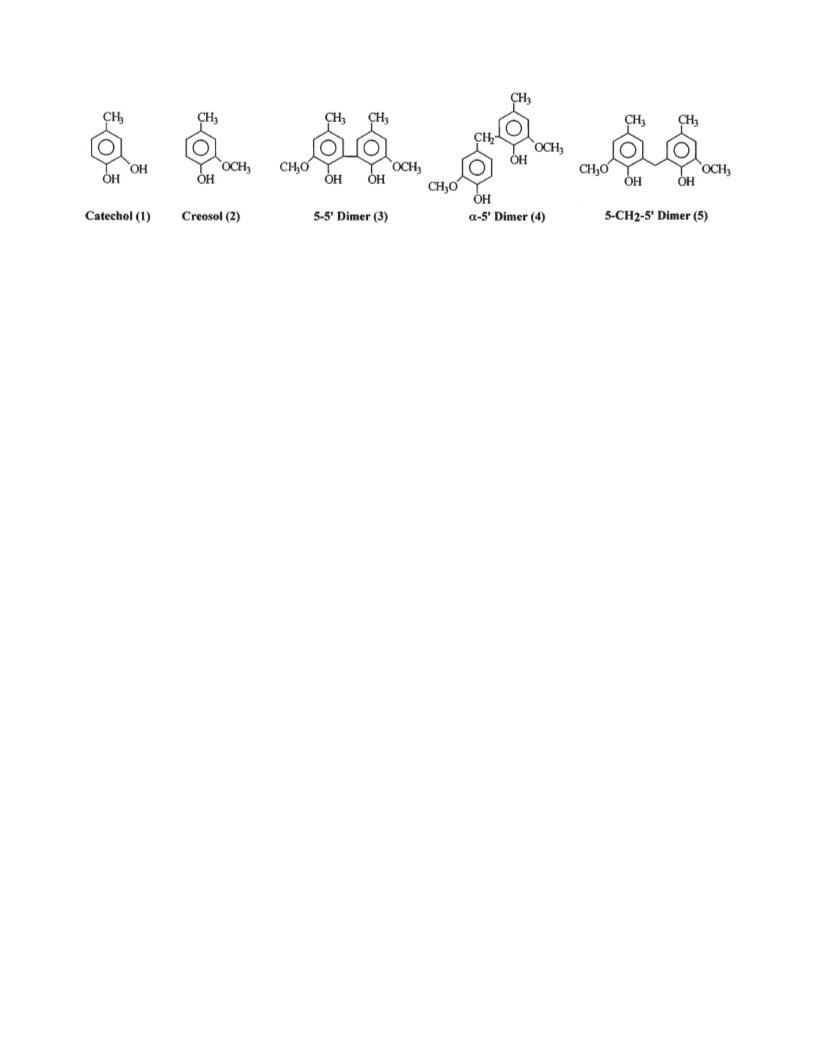
by the groups present on the aromatic rings, i.e., OH >
OCH > CH > C C . The high reactivity of catechol moi-
3
3
6 5
eties is in accord with the finding of Francis and Reeve (28).
Our data also show that the more activated the aromatic
structure, the greater its reactivity toward alkaline hydrogen
peroxide, irrespective of the metal ion being added. The
presence of metal ions did not significantly alter the above
reactivity order but did change the extent of reactions.
Johansson and Ljunggren (29) have demonstrated, using
model compounds, that during oxygen delignification the
presence of two phenolic hydroxyl groups on an aromatic
ring considerably increases its reactivity toward oxygen;
methylcatechol was found to be more reactive than creosol.
In addition, Johansson and Ljunggren (29) have shown that
the reactivity of a mononuclear aromatic structure is greater
than its dinuclear analogue. Similarly, using critical oxida-
tion potential data, Kratzl et al. (30) have arrived at the same
conclusions. In these respects, our findings are in complete
agreement with the literature claims despite different sys-
tems having been examined. Since hydroxyl and superoxide
anion radicals are the two major radical species produced
from the stepwise reduction of oxygen and from the decom-
position of hydrogen peroxide (31) and since these radicals
are more reactive toward phenolic compounds than peroxide
or oxygen, it is not surprising that the data that emerges
from the present study parallels the mechanistic information
already described for oxygen delignification.
Results and discussion
Table 1 shows the H O consumption, the total amounts
2
2
of identified products (excluding any remaining starting ma-
terials), the amounts of unidentified compounds, and those
that could not be detected by GC. The yields of products re-
covered after the reaction varied between 67 and 92%. Se-
vere degradation of model compounds with the formation of
volatile and highly water soluble products likely accounts
for the unrecovered material.
Effect of metal ions on the reactivity of hydrogen
peroxide with residual Kraft lignin model compounds
The mechanism of alkaline hydrogen peroxide
delignification of chemical pulps is complicated due to the
complexity of the lignin structure and the large variety of re-
active species formed during the process. Figures 1a and 1b
display the reactivities of the examined lignin model com-
pounds toward alkaline H O in the presence or absence of
2
2
metal ions. By presenting the same data in different ways,
two sets of evaluations and comparisons can be made: one
addressing the reactivity of the various lignin models in the
presence (or absence) of the same metal ion (Fig. 1a) and
the other the influence of the various metal ions on the same
model compound (Fig. 1b).
The plots of Fig. 1a show that, in general, the reactivities
of the five model compounds examined was of the following
order: catechol (1) > creosol (2) > α-5′ dimer (4) > 5-CH -5′
Compared to our control runs, the addition of metal ions
dramatically affected the reactivities of the examined model
compounds toward alkaline hydrogen peroxide (Fig. 1b).
The transition metals increased the extent of reaction be-
tween hydrogen peroxide and the model compounds, as evi-
denced by the decreased amounts of remaining starting
material present after the reaction (compared to the control).
2
dimer (5) > 5–5′ dimer (3). This reactivity order is what one
would expect by considering the degree of activation caused
©
1999 NRC Canada

 Sun, Yujun
Sun, Yujun
 Fenster, Michael
Fenster, Michael
 Yu, Annie
Yu, Annie
 Berry, Richard M.
Berry, Richard M.
 Argyropoulos, Dimitris S.
Argyropoulos, Dimitris S.