The decrease in activity with the increase of the IL
amount indicates that the IL interacts with the Ru catalyst. To
This is evidence that the IL adsorbs in situ to strongly
binding Ru centers through its N(CN)2 anion, and these
ensembles block the consecutive hydrogenation to cyclohex-
ane. We also observed this “cocatalytic” effect of IL in citral
hydrogenation with an ex situ prepared thin IL film on
supported nanoparticles, and ascribed it to a new kind of
examine this further, we analyzed the surface of the Ru/Al O3
2
catalyst applied in the hydrogenation experiment by means of
photoelectron spectroscopy (XPS) (Ru3d and N1s spectra:
Figures S2–S5 in the Supporting Information). The N1s
spectra confirm the presence of nitrogen, which can originate
only from the IL because the IL is the only nitrogen source in
the reaction system. These findings unequivocally indicate
that the IL is chemisorbed on the catalyst. The ratio of the
[
11b]
ligand effect,
enthalpy of H2.
expressed by a decrease of the adsorption
Because usually the adsorption enthalpy of H O on
2
ruthenium in the presence of H is roughly half of that of a
2
[
3]
surface (s) atoms of nitrogen to ruthenium is (N/Ru) = 0.22,
surface coated only with water, the hydrogen coverage
which is now decreased by the presence of the IL a) improves
the hydrophilic character of the Ru catalyst and b) diminishes
over-hydrogenation.
s
which is distinctly lower than the ratio resulting from the
chosen hydrogenation batch in the liquid phase (v) (N/Ru) =
v
3
.5. If one considers that the only binding energy detected in
the N1s spectra is at 398.0 eV and keeping in mind the XPS
results measured with catalysts in which the IL is present as a
Of course, the different solubilities of the starting
material, intermediate, and product in ILs must also be
considered (physical solvent effect). Thus, the excess values of
molar free enthalpies of mixing for mixtures of benzene,
[
11b]
thin film on the supported noble metal (SCILL systems),
a
preferred binding of the dicyanamide anion to the Ru surface
and its modification can be deduced. In contrast to Pd
catalysts (SCILL type), there are no indications of a
significant change in the electronic state of the ruthenium in
Ru/Al O after benzene hydrogenation in the presence of the
cyclohexene, or cyclohexane with [EMIM][NTf ] increase in
2
[15]
the order mentioned and their solubilities decrease corre-
spondingly. This has also been established for a number of
[
16]
other ILs. Consequently, in presence of an IL during the
selective hydrogenation of benzene, the cyclohexene, which is
formed but less soluble in the IL, is withdrawn faster from
catalyst surface, so that the further hydrogenation to cyclo-
hexane is reduced. This is also associated with the consid-
erably lower solubility of cyclohexene in water relative to that
of benzene. Cyclohexene is formed as the main product in the
initial phase of the reaction (Figures 1 and 2), and water and
IL impede its renewed adsorption. If the degree of coverage
of benzene and cyclohexene on the Ru surface are compa-
rable as the reaction advances, and their adsorption competes
with that of water, the hydrophilicity of the catalyst surface no
longer suffices for fast desorption of cyclohexene, and more
cyclohexane is formed.
2
3
IL. These findings suggest that catalyst activity is instead
reduced by an ensemble effect; that is, the geometric
arrangement or the number of Ru atoms catalyzing the
benzene hydrogenation is reduced (“diluted”) by the anion of
the IL.
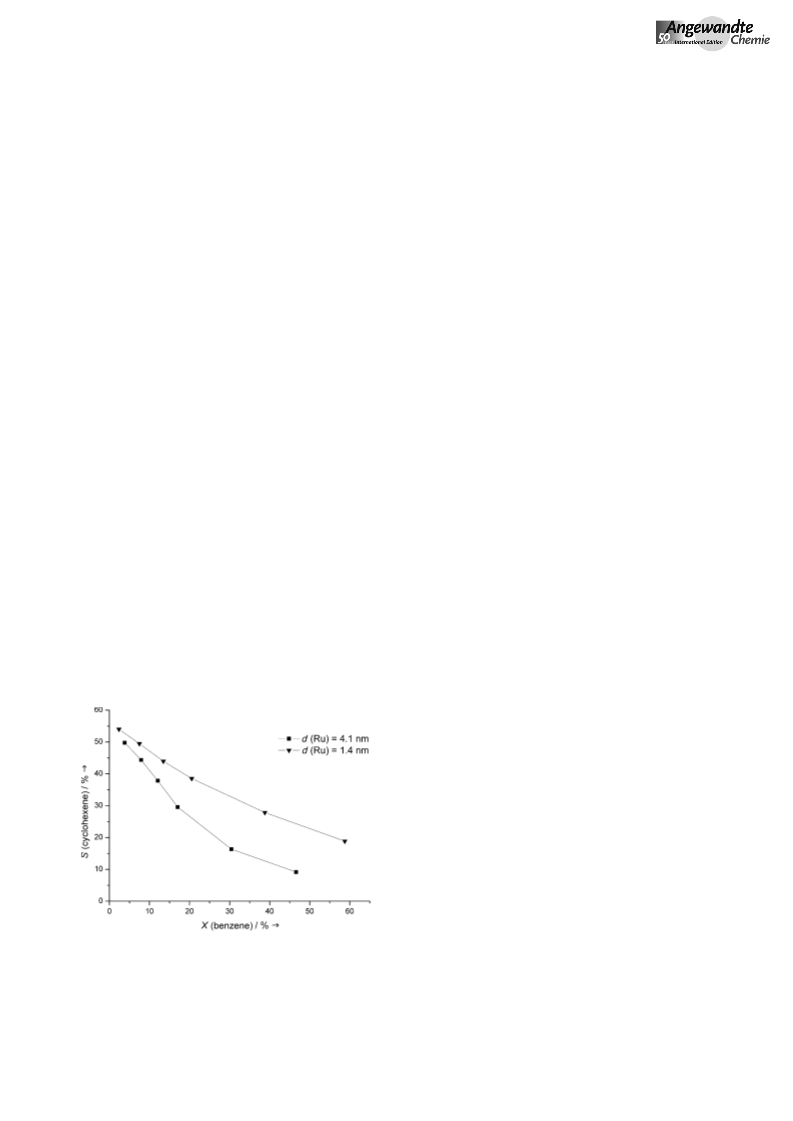
If the IL modifies the Ru surface in this way, the selective
hydrogenation of benzene to cyclohexene in the presence of
the IL should also respond to a change in Ru dispersion, in
other words, the Ru particle size (determined by H chem-
2
isorption, Table S1 in the Supporting Information). Figure 3
shows that this is precisely the case. In the case of highly
nanodisperse Ru particles in the catalyst RuAl O -14 (d =
Ru
2
3
1
.4 nm), cyclohexene selectivity was higher over a wide range
of conversion than that with the catalyst having a mean Ru
particle size of 4.1 nm, so that higher yields of cyclohexene
Analysis of the aqueous phase after the reaction using
ICP-OES revealed no ruthenium, indicating that leaching and
the formation of a homogeneous catalyst did not occur during
the reaction.
(Ycyclohexene,max = 11%) were obtained.
In summary, with the simple catalyst system described
here the extremely difficult selective hydrogenation of
benzene to cyclohexene in water as a solvent in the presence
of an IL has been successfully carried out. Cyclohexene is the
main product of the reaction in the presence of ruthenium on
Al O /H O/[B3MPyr][DCA] under moderate reaction con-
2
3
2
ditions with a high selectivity of 60% at low conversions.
Using small amounts (ppm range) of an IL based on DCA,
the addition of inorganic salts and NaOH is not necessary,
substantially simplifying the reaction and eliminating the
expensive purification of the reaction medium. These advan-
tages over the ordinary multicomponent reaction mixtures
and catalysts—along with the associated saving of costly
materials—make catalyst optimization attractive. Future
work should be focused on SCILL systems and an appropriate
catalytic reaction engineering, for example in a continuous-
[
17]
flow reactor, similar to SILP catalysis.
Figure 3. Effect of Ru particle size on the selectivity–conversion curve,
n(IL)/n(Ru-41)=2.9, n(IL)/n(Ru-14)=3.5. See Figure 1 for reaction
conditions.
Angew. Chem. Int. Ed. 2011, 50, 10453 –10456
ꢀ 2011 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Schwab, Frederick
Schwab, Frederick
 Lucas, Martin
Lucas, Martin
 Claus, Peter
Claus, Peter