CHEMCATCHEM
COMMUNICATIONS
CN of the PtÀPt bonds also increased from 3.0 to 7.5 (Table 3).
The addition of Re to Pt/b is thought to promote the growth
of Pt0 particles without significant bimetal bonding, thus re-
sulting in a drop in phenol-production performance (Table 1).
In the ZSM-5 pores, there is a definite synergistic catalytic
effect of Pt and Re in the synthesis of phenol (Table 2).
Pt/ZSM-5 (29.3%, TOF=0.70 hÀ1, defined as reacted benzene/
total Pt per hour for convenience) and Re/ZSM-5 both showed
low phenol selectivities (35.7%, negligible TOF=0.09 hÀ1). Nev-
ertheless, the addition of Re to the Pt/ZSM-5 catalyst notably
increased the selectivity from
The coexisting NH3 was converted into N2 under the phenol-
synthesis conditions (Table 1 and Table 2). Decreasing the
amount of NH3 consumption is an important challenge for the
practical application of Pt and Pt-Re/zeolites. Herein, we also
report a selective synthesis of phenol in an alternating process:
First, a Pt/b catalyst was pretreated with benzene/O2/NH3 at
453 K for 1 h, during which time neither phenol nor N2 were
evolved and no CO2 was observed. Then, the pretreated cata-
lyst was subjected to a flow of benzene/H2O for 1 h at 513 K,
during which time phenol formation was observed (Table 4
29.3% to 60.0%. Pt-Re/ZSM-5
catalysts with Pt loadings of
Table 4. Synthesis of phenol through successive benzene+O2+NH3 and benzene+H2O processes on the
Pt(1.0 wt.%)/b catalyst.
1.0–2.1 wt.% at 533 K showed
good phenol selectivities (79.5–
Cat.
Pre-reaction for 1 h
Benzene/ N2
O2/NH3 [mmol] [mmol]
Benzene+H2O post-treatment[a]
Produced
N2/phenol
82.9%,
TOF=1.20–1.90 hÀ1
;
T
[K]
CO2
T
[K]
Benzene/
H2O/N2
Phenol Phenol
[mmol] selectivity [%]
Table 2).
The Re atoms in the calcined
Pt-Re/ZSM-5 catalyst are present
as [Re7+O4] monomers, as sug-
Pt/b[c] 453 1.0:2.0:7.2
Pt/b[c] 453 1.0:2.0:7.2
Pt/b[c] 453 1.0:2.0:7.2
Pt/b[c] 453 0:2.0:0
n.d.[b]
n.d.[b]
n.d.[b]
–
n.d.[b]
n.d.[b]
n.d.[b]
–
513 1.0:62.5:2.0
513 1.0:0:64.5
66.4
0.4
28.7
3.9
>99.8
>99.8
>99.8
>99.8
93.3
trace
trace
trace
–
513
0:62.5:3.0
513 1.0:62.5:2.0
513 1.0:62.5:2.0 134.5
gested by CNRe O =3.3 at
=
Pt/b[d] 453 0.24:0.48:1.8 52.3
58.0
0.39
0.173 nm and CNReÀO =1.2 at
0.214 nm and by the Re7+ va-
lence (Re LI-edge XANES). If the
Pt-Re/ZSM-5 was treated with
NH3 at 553 K, the Re7+ mono-
mers transformed into Reꢁ3+
[a] Phenol: total amount produced within 50–90 min. Selectivity [%]: (phenol produced in the benzene+H2O
process)/[(phenol produced in the benzene+H2O process)+(CO2 produced during the pre-reaction/6)]ꢁ100.
[b] Not detected by GC. [c] Catalyst (1 g). [d] Catalyst (2.5 g).
clusters, with oxo-ligands at 0.173 and 0.202 nm and ReÀRe(Pt)
bonds at 0.274 and 0.303 nm, presumably affording PtÀRe bi-
metal clusters in the ZSM-5 pores. The Pt-Re/ZSM-5 catalyst
was activated by the NH3 pretreatment and the promotion of
Re species may be ascribed to the formation of bimetal clus-
ters. However, under the catalytic conditions, most of the bi-
metal clusters decomposed into Re monomers and Pt clusters.
The Pt-Re/ZSM-5 catalyst showed heterogeneous Pt states,
which makes discussion on the active structure of the Pt-Re/
ZSM-5 catalyst difficult. The synergistic aspect of the Pt-Re/
ZSM-5 catalyst may be simulated by using DFT calculations,
which is currently under investigation.
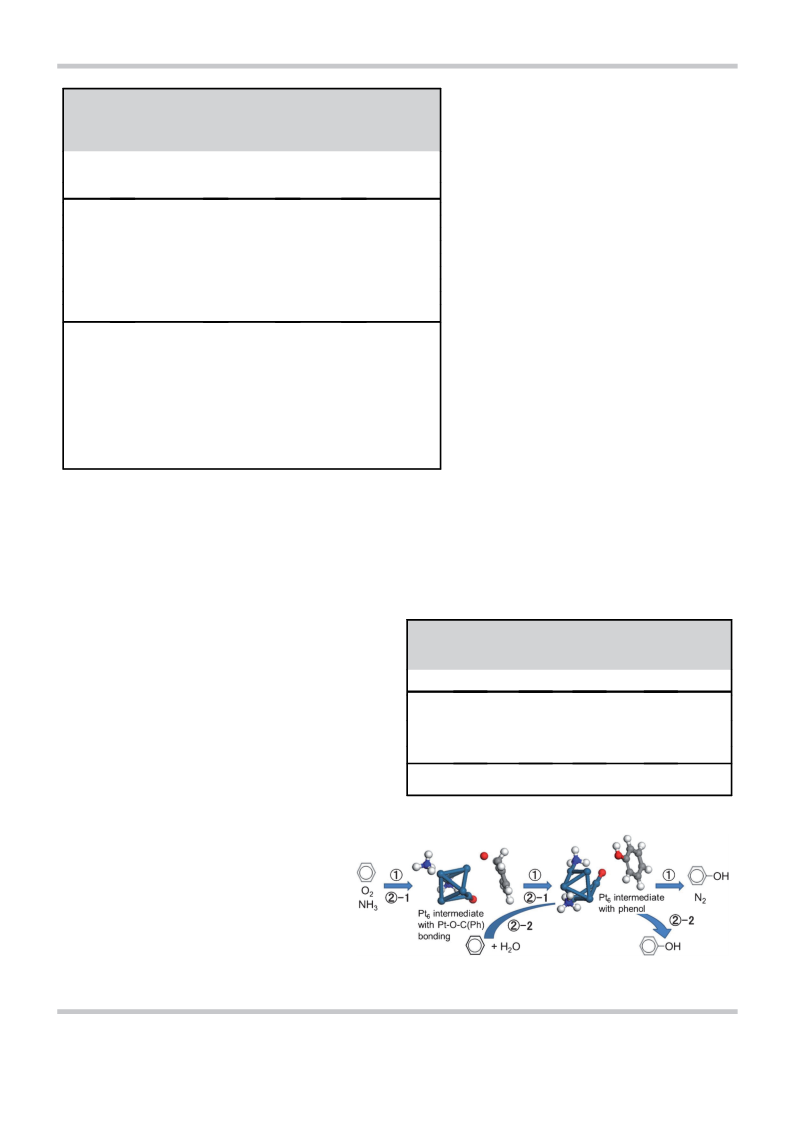
and Scheme 1). The total amount of phenol that was produced
in the benzene/H2O flow on Pt(1 wt.%)/b (1 g) was 66.4 mmol
and no CO2 was produced, which indicated a phenol selectivity
of >99.8%, considering the sensitivity of GC for CO2 detection.
Thus, the relative amount of as-produced N2 (NH3 consump-
tion) to phenol was suppressed in this alternating processes
(Table 4). If the benzene/O2/NH3-pretreated catalyst was sub-
jected to a flow of H2O alone, 28.7 mmol of phenol was ob-
tained. These results indicate that the phenol production is not
ascribed to the benzene+H2O reaction but rather to the
H2O(+benzene)-promoted desorption of phenol that had been
produced during the pre-reaction of benzene/O2/NH3 at 453 K,
although a small contribution of the benzene+H2O reaction to
the phenol synthesis is not excluded.[10] The addition of H2O to
the benzene/O2/NH3 feed decreased the phenol selectivity. The
amount of phenol that was produced in 1 h ranged from 0.5–
6.8-times the total amount of Pt on the Pt/b and Pt-Re/ZSM-5
catalysts. Thus, the alternating process for the pre-reaction pro-
cess with benzene/O2/NH3 and the post-treatment process
with benzene/H2O is a promising method for the selective pro-
duction of phenol (83–99.8% selectivity) with minimum NH3
consumption, as shown in the Supporting Information, Fig-
ures S6 and S7, in which the alternating processes could be re-
peated without significant loss in performance.
Notably, the catalytic performance largely depended on the
source and type of zeolite (see the Supporting Information,
SI 4 and Figure S5). The activity of the Pt-Re/ZSM-5 (TOSOH;
SiO2/Al2O3, 40:1) catalyst was one third of that of the Pt-Re/
ZSM-5 (Zeolyst; SiO2/Al2O3, 30:1) catalyst and the selectivity
was also lower (Table 2). The activity and selectivity of the CVD
Re/ZSM-5 catalyst (SiO2/Al2O3, 19:1) were sensitive to the quali-
ty and purity of the zeolite framework.[9] The catalytic perfor-
mance decreased in the following order of the type of zeolite:
b>ZSM-5>Y>mordenite. For comparison, amorphous silica/
alumina (SiO2/Al2O3, 30:1) was used as a support, but the as-
obtained Pt/SiO2-Al2O3 and Pt-Re/SiO2-Al2O3 were almost inac-
tive in the phenol synthesis (0% and 1.1% selectivity, respec-
tively), unlike the Pt and Pt-Re/zeolites. These results indicate
a confinement effect on the phenol synthesis, thus showing
the importance of pore structure, acidity, and quality of the
crystalline zeolite.
In conclusion, we found that the Pt/b and Pt-Re/ZSM-5 cata-
lysts could selectively promote the direct synthesis of phenol
from benzene and O2, regulated by NH3, as well as from alter-
nating process that offered decreased NH3 consumption.
ꢀ 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ChemCatChem 0000, 00, 1 – 4
&
3
&
ÞÞ
These are not the final page numbers!

 Wang, Linsheng
Wang, Linsheng
 Yamamoto, Sadaaki
Yamamoto, Sadaaki
 Malwadkar, Sachin
Malwadkar, Sachin
 Nagamatsu, Shin-Ichi
Nagamatsu, Shin-Ichi
 Sasaki, Takehiko
Sasaki, Takehiko
 Hayashizaki, Kenichiro
Hayashizaki, Kenichiro
 Tada, Mizuki
Tada, Mizuki
 Iwasawa, Yasuhiro
Iwasawa, Yasuhiro