
Journal of Physical Chemistry p. 6266 - 6271 (1984)
Update date:2022-08-17
Topics:
 Powell, M. F.
Powell, M. F.
 Morimoto, H.
Morimoto, H.
 Erwin, W. R.
Erwin, W. R.
 Gordon, B. E.
Gordon, B. E.
 Lemmon, R. M.
Lemmon, R. M.
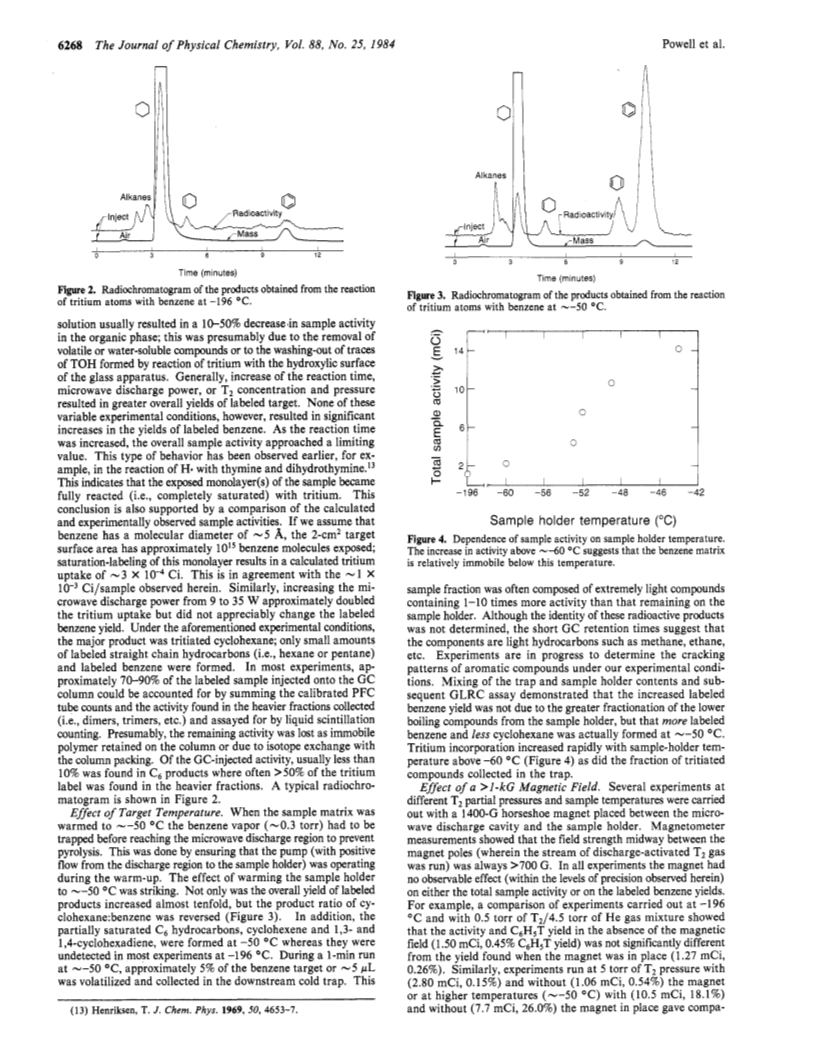
Reaction of thermal tritium atoms, generated by microwave activation os T2 gas, with benzene and biphenyl was studies at ca.-50 and -196 deg C.The saturation reactions (i.e.,benzene->cyclohexane-t6)predominated over isotope exchange (i.e.,benzene->benzene-t( at -196 deg C.However, significant exchange labeling occurred at ca.-50 deg C, with a concomitant reduction in the yields of saturated products.This reversal in labeled product yields at the different temperatures is due, in part, to the faster rate of H expulsion from the intermediate cyclohexadienyl radical at -50 deg C and to the increased mobility of the warmer matrix that retards multiple T- reactions with the same aryl molecule by covering up singly tritiated intermediates.The less volatile aryl compound, biphenyl, was labeled in a diffusionally active matrix of either benezene or cyclohexane, whereas it could not be labeled otherwise.
View More




Doi:10.1021/jp076169h
(2008)Doi:10.1016/S0040-4039(01)91777-0
(1974)Doi:10.1023/A:1015045216923
(2001)Doi:10.1039/d1cc00587a
(2021)Doi:10.1016/j.jallcom.2017.05.044
(2017)Doi:10.1021/acs.jnatprod.6b00941
(2017)