
Journal of the Chinese Chemical Society p. 1211 - 1222 (2007)
Update date:2022-08-11
Topics:
 Aboul-Gheit, Noha A. K.
Aboul-Gheit, Noha A. K.
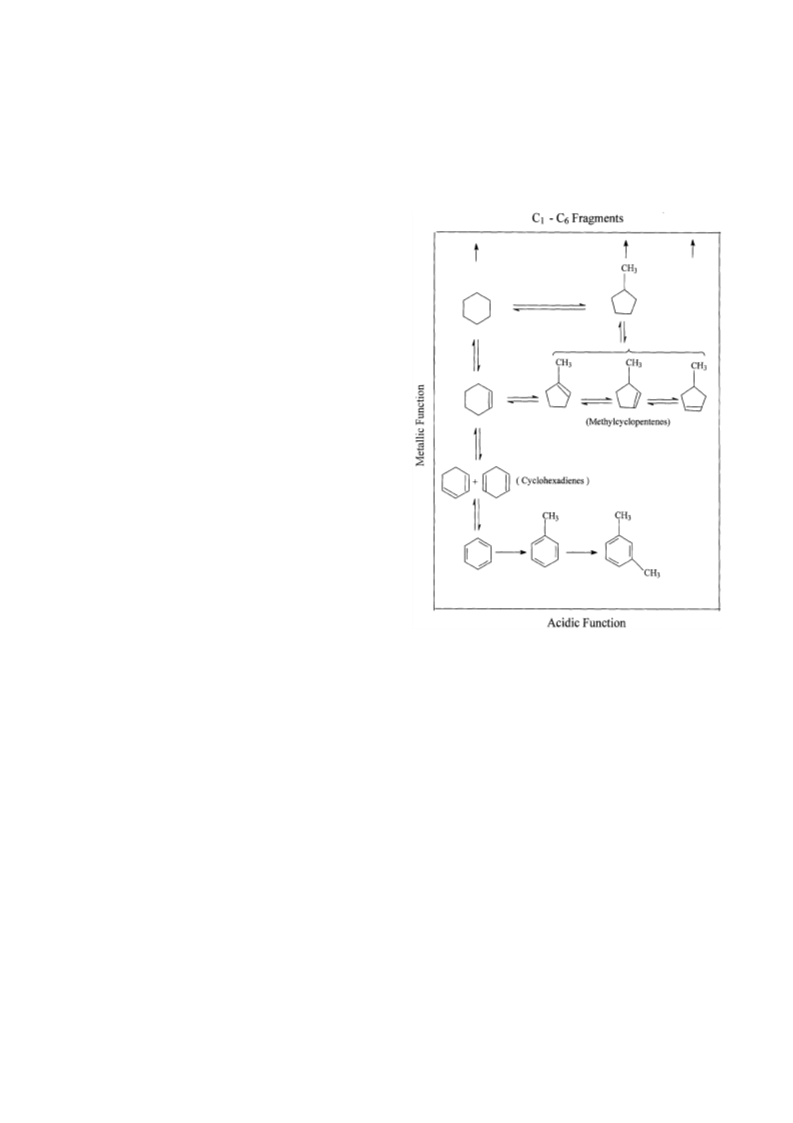
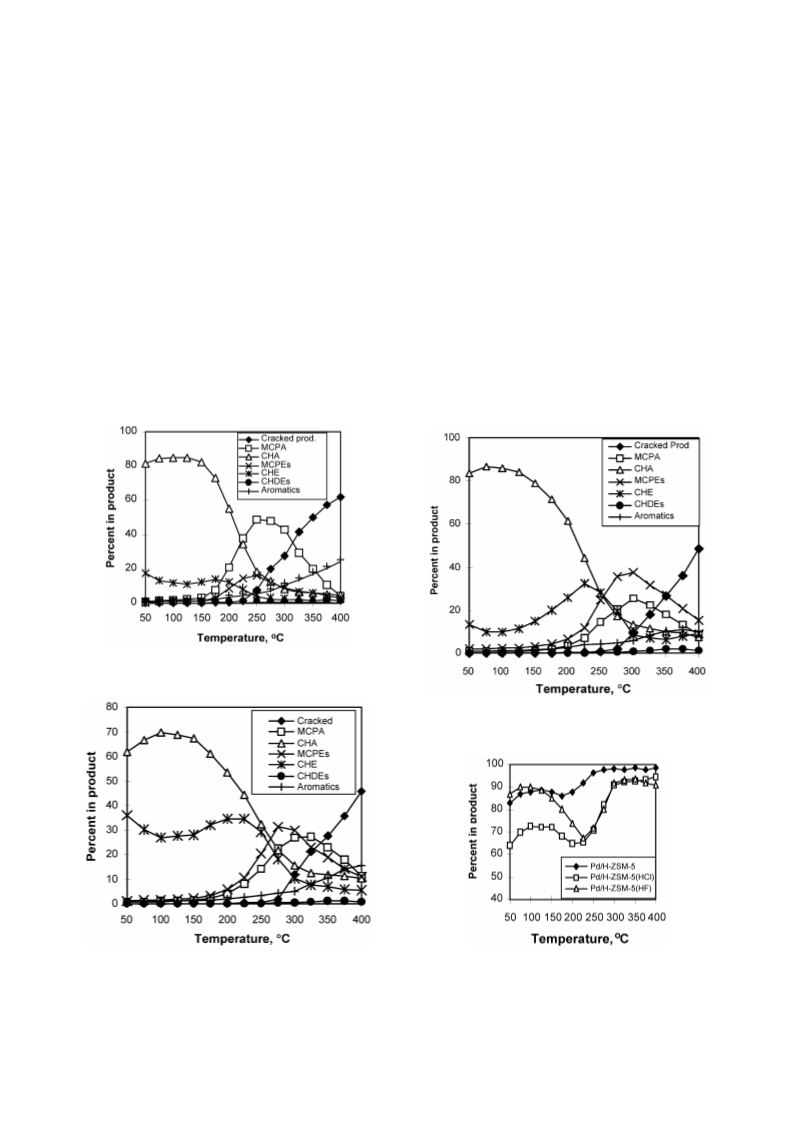
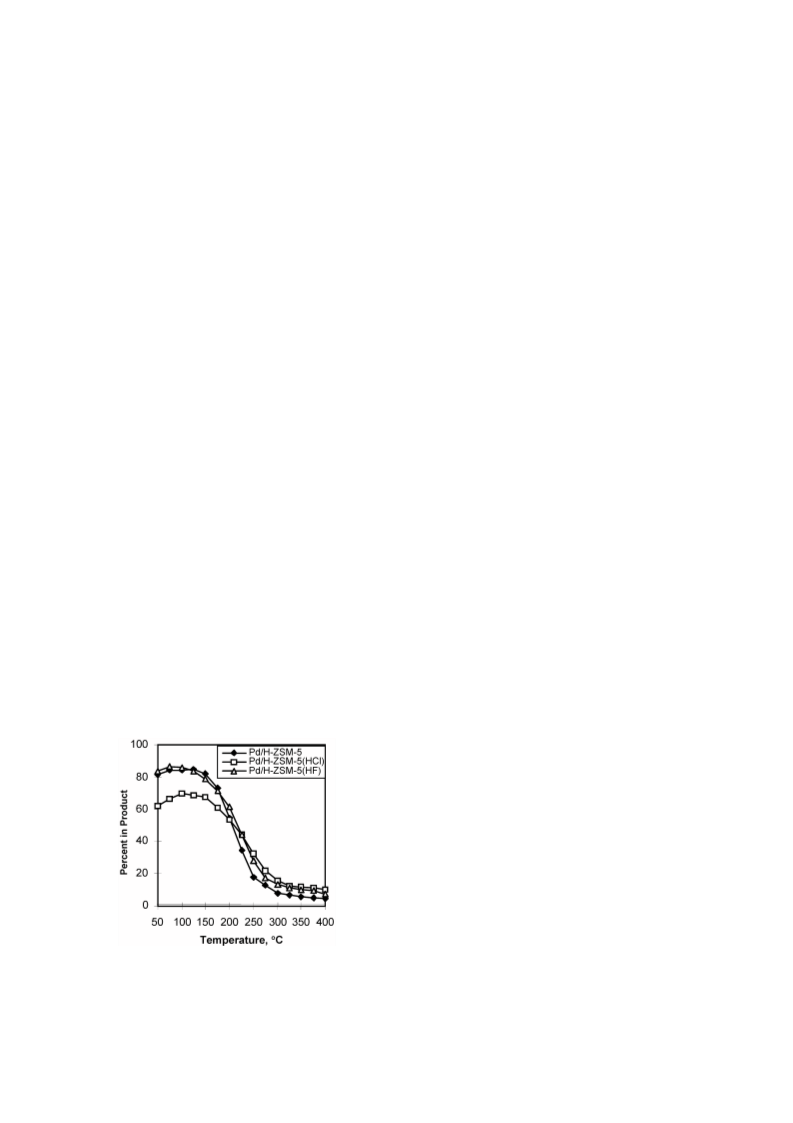
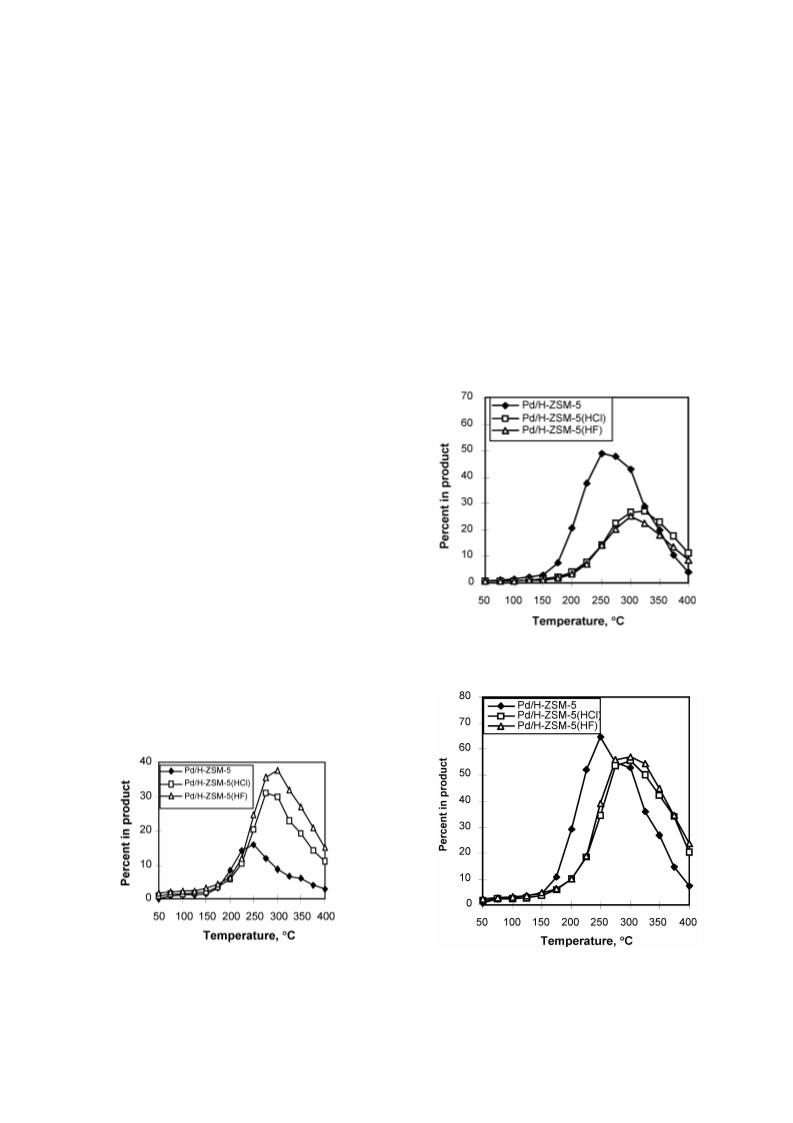
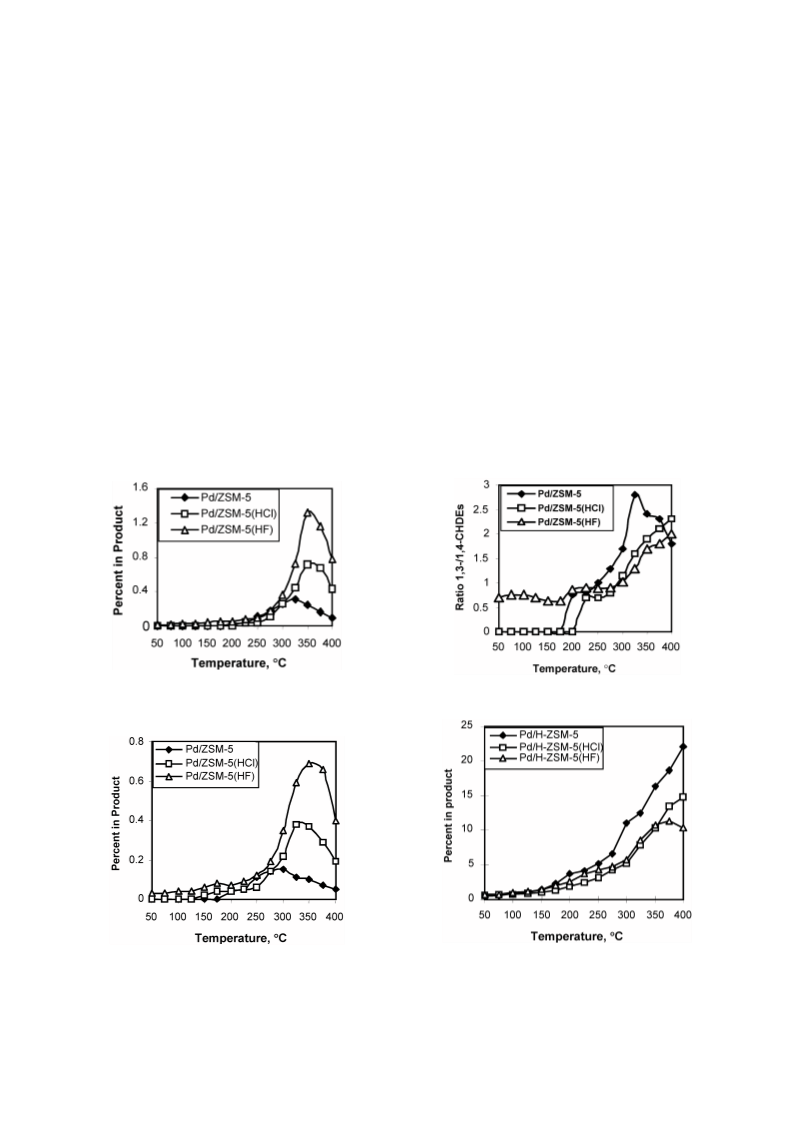
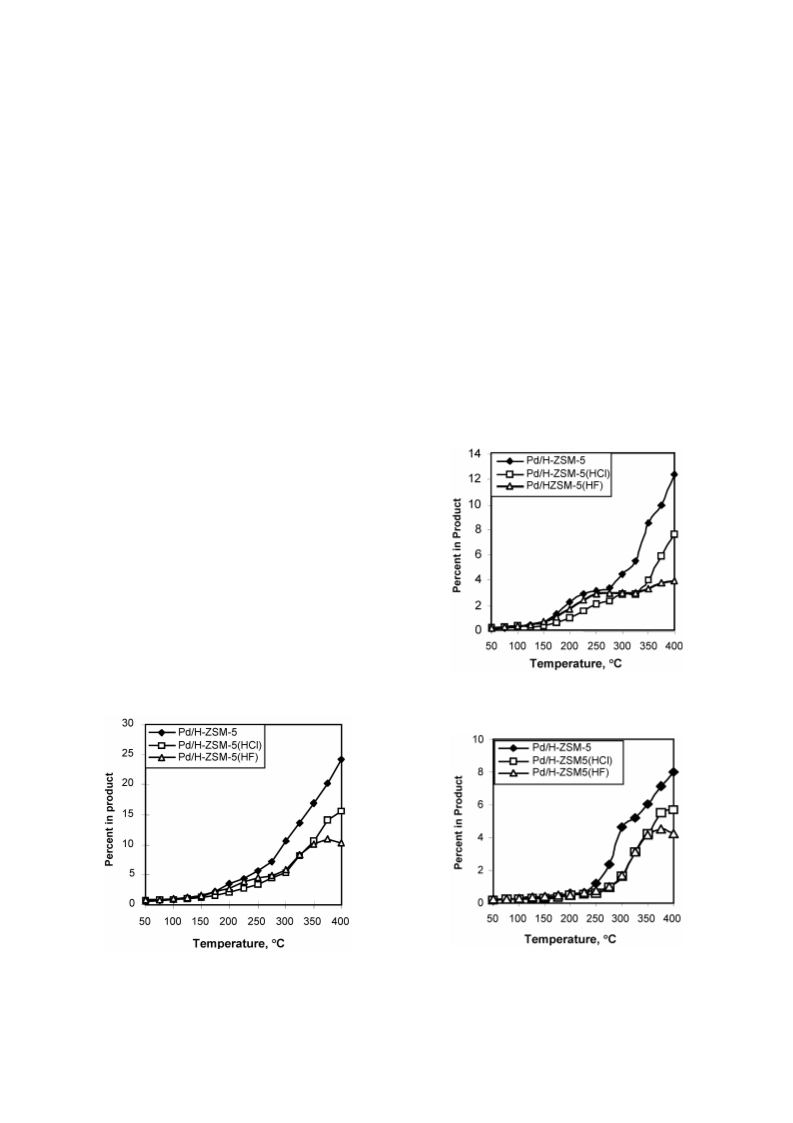
Cyclohexene (CHE) hydroconversion was performed in a flow reactor at atmospheric pressure and temperatures of 50-400 °C using: Pd/H-ZSM-5, Pd/H-ZSM-5(HCl), and Pd/H-ZSM-5(HF) catalysts. These catalysts were characterized for acid site strength distribution via NH3 TPD, Pd dispersion via H2 chemisorption, TPR via reduction of the metal oxide in the catalysts and XRD for tracing crystallinity. The hydroconversion steps proceeded as follows: CHE → Cyclohexane (CHA); CHE → Methylcyclopentenes (MCPEs) → Methylcyclopentane (MCPA); CHE → Cyclohexadienes (CHDEs) → Benzene → Alkylbenzenes; CHE and others → Hydrocracked products. The overall hydroconversion of CHE was achieved in the catalyst order: Pd/H-ZSM-5 > Pd/H-ZSM-5(HF) > Pd/H-ZSM-5(HCl). CHE hydrogenation step was the major reaction at low temperatures which significantly inhibited via HCl treatment, but slightly enhanced via HF treatment. At medium temperatures, on all catalysts, isomerisation to MCPEs and MCPA increase to a maximum then a decline with a further increase of temperature. The overall isomerisation of CHE was highest on the untreated catalyst. During the higher temperature range, dehydrogenation, alkylation and hydrocracking were increased with temperature. Dehydrogenation of CHE always yielded larger amounts of 1,3-CHDE than 1,4-CHDE. These cyclohexadienes were produced in the catalyst order: Pd/H-ZSM-5(HF) > Pd/H-ZSM-5(HCl) > Pd/H-ZSM-5. In general, benzene alkylation to toluene exceeded that of xylenes, indicating that the second methylation is more difficult than the first. However, the catalytic activities for benzene and toluene production were in the order: Pd/H-ZSM-5 ? Pd/H-ZSM-5(HCl) > Pd/H-ZSM-5(HF), whereas for xylenes production, Pd/H-ZSM-5 ? Pd/H-ZSM-5(HF) > Pd/H-ZSM-5(HCl). Intrapore diffusion plays an important role during the dehydrogenation reactions as well as during the interconversion of individual aromatic hydrocarbons.
View More


Doi:10.1002/jhet.1833
(2015)Doi:10.1016/j.ejmech.2018.06.025
(2018)Doi:10.1021/ja0261630
(2002)Doi:10.1002/anie.202016284
(2021)Doi:10.1002/anie.201302800
(2013)Doi:10.1016/S0957-4166(97)00002-5
(1997)