10.1002/cctc.201701696
ChemCatChem
FULL PAPER
added dropwise to the slurry and stirred for another 5 min. The nominal
Zn/Ru3+ molar ratio was 6.48 to guarantee the entire reduction of Ru. The
reaction during galvanic replacement was 3Zn + 2Ru3+ = 3Zn2+ + 2Ru, as
the standard reduction potential of the Zn2+/Zn couple (0.76 V versus
the standard hydrogen electrode, SHE) is much lower than that of the
Ru3+/Ru couple (0.68 V versus SHE). After galvanic replacement, aside
from residual metallic Zn combining with Ru, there were also oxidized
zinc species arising from the hydrolysis of Zn2+ or the oxidation of
metallic Zn by dissolved oxygen.[35] Hence, the black slurry was cooled
down to room temperature, and hydrochloric acid (37%) was added
dropwise under mild stirring to adjust the content of metallic Zn and also
to remove the oxidized zinc species that might cover the catalyst by
reactions of Zn + 2HCl = ZnCl2 + H2 and ZnO + 2HCl = ZnCl2 + H2O,
respectively. Finally, the solids were washed with deionized water until
neutrality and removal of the chloride ions by AgNO3 test. Surface
analysis by XPS also confirmed that Cl was absent. The as-prepared
Acknowledgements
This work was supported by the National Key R&D program of
China (2016YFB0301600), the NSF of China (21373055,
21703024), the Science & Technology Commission of Shanghai
Municipality (08DZ2270500), the Beijing Synchrotron Radiation
Facility (BSRF), the SINOPEC (S411063), the Scientific and
Technological Research Program of Chongqing Municipal
Education Commission (KJ1500305), and the Science
Technology Commission of Chongqing Municipality
(cstc2016jcyjA0392).
&
Keywords: Galvanic replacement • Ru–Zn • benzene •
hydrogenation • cyclohexene
[1]
[2]
L. Lloyd, Handbook of Industrial Catalysis Springer, New York, 2011.
K. Weissermel, H. J. Arpe, Industrial Organic Chemistry, 4th ed. Wiley-
VCH, Weinheim, 2003.
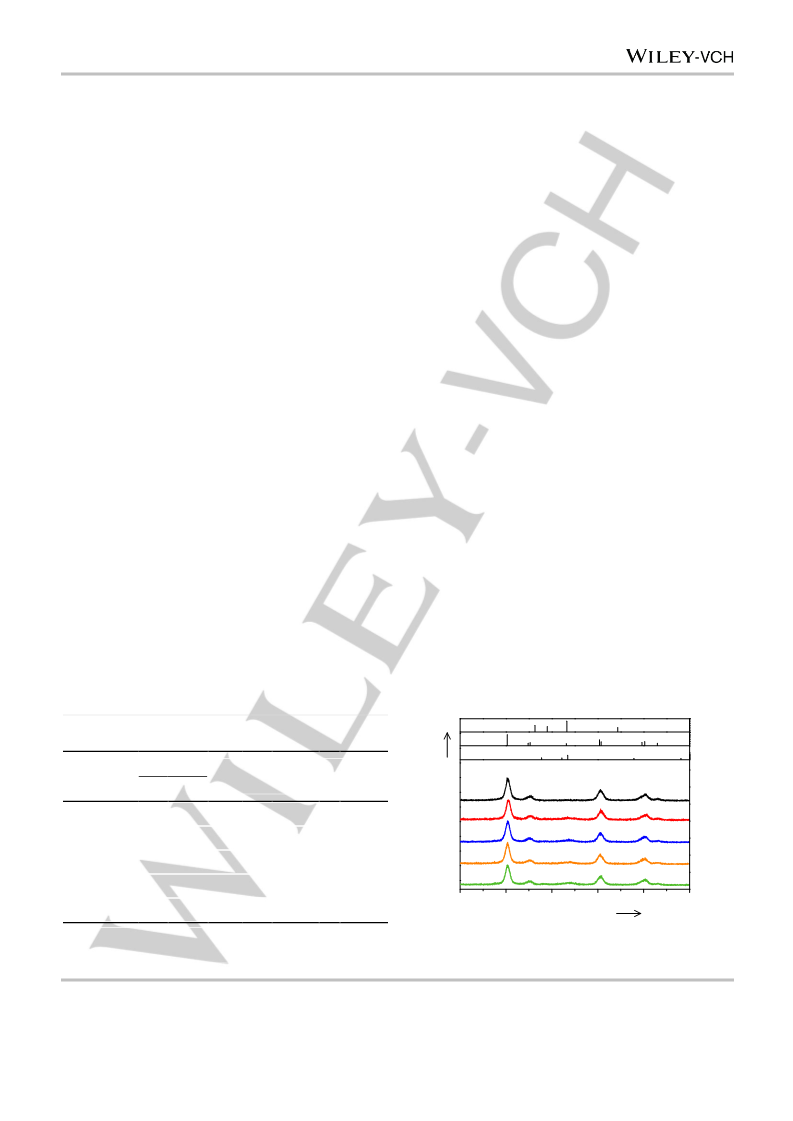
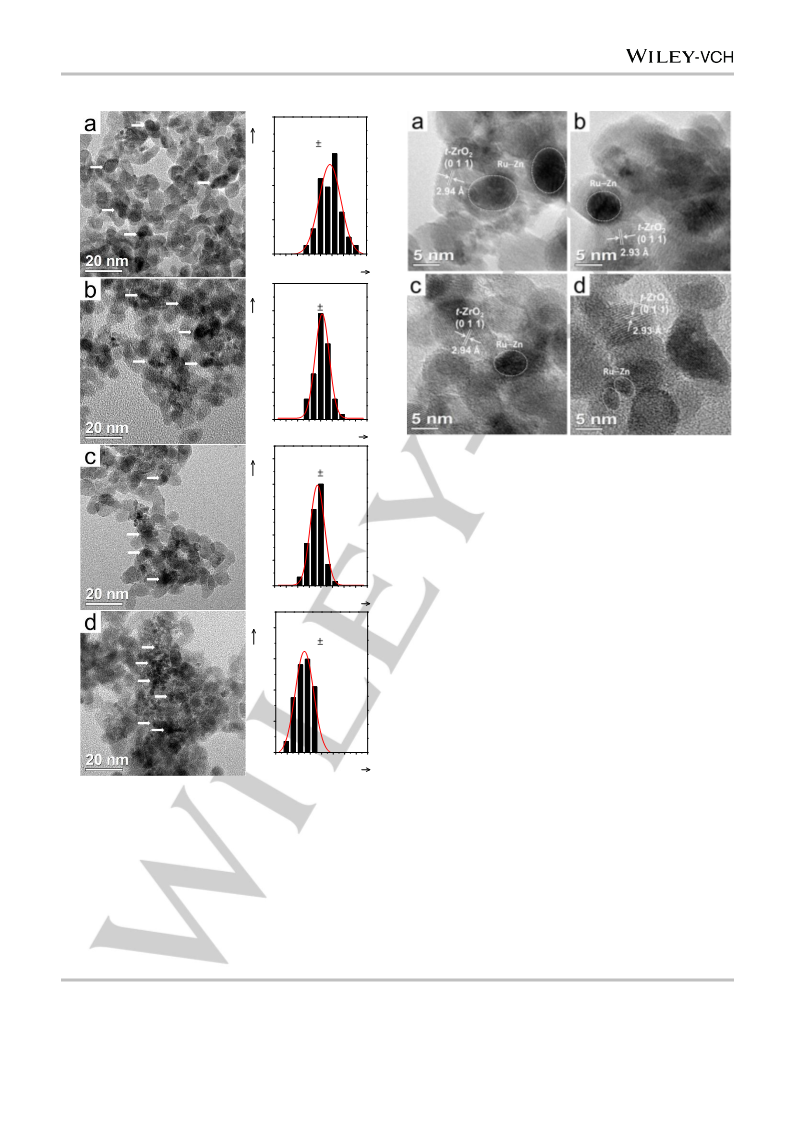
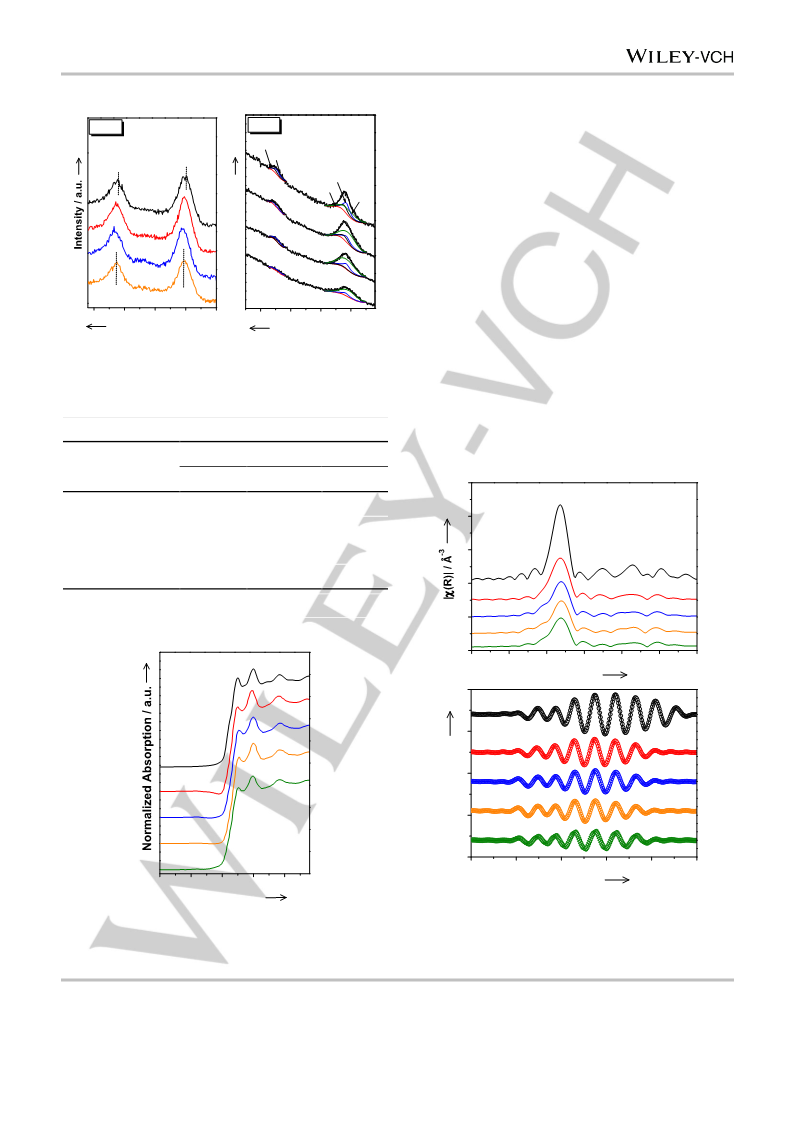
nanocomposite catalysts were denoted as Ru–Zn/ZrO2(x), where
x
represents the nominal HCl/Zn molar ratio of 1.32, 1.46, 1.52, or 1.85.
[3]
[4]
L. Foppa, J. Dupont, Chem. Soc. Rev. 2015, 44, 18861897.
Y. Pei, G. B. Zhou, N. Luan, B. N. Zong, M. H. Qiao, F. Tao, Chem. Soc.
Rev. 2012, 41, 81408162.
HCl
H2O
RuCl3
[5]
[6]
H. Nagahara, M. Konishi, Eur. Patent 220525, 1986.
J. A. Anderson, M. F. García, Supported Metals in Catalysis Imperial
College Press, London, 2005.
3Zn + 2Ru3+ 3Zn2+ + 2Ru
[7]
[8]
[9]
H. Z. Liu, S. G. Liang, W. T. Wang, T. Jiang, B. X. Han, J. Mol. Catal. A
2011, 341, 3541.
T. T. Zhang, Z. M. Wang, Q. Q. Zhao, F. Li, W. Xue, J. Nanomater.
2015, 2015, 18.
J. Q. Wang, Y. Z. Wang, S. H. Xie, M. H. Qiao, H. X. Li, K. N. Fan, Appl.
Catal. A 2004, 272, 2936.
Zn + 2HCl ZnCl2 + H2
ZnO + 2HCl ZnCl2 +H2O
t-ZrO2
Ru
Zn
Scheme 2. Illustration of the formation process of the RuZn/ZrO2
nanocomposite catalysts via galvanic replacement followed by acid treatment.
[10] G. B. Zhou, X. H. Tan, Y. Pei, K. N. Fan, M. H. Qiao, B. Sun, B. N.
Zong, ChemCatChem 2013, 5, 24252435.
[11] J. L. Liu, L. J. Zhu, Y. Pei, J. H. Zhuang, H. Li, H. X. Li, M. H. Qiao, K. N.
Fan, Appl. Catal. A 2009, 353, 282287.
Catalytic testing
[12] X. H. Yan, Q. Zhang, M. Q. Zhu, Z. B. Wang, J. Mol. Catal. A 2016, 413,
8593.
[13] P. Zhang, T. B. Wu, T. Jiang, W. T. Wang, H. Z. Liu, H. L. Fan, Z. F.
Zhang, B. X. Han, Green Chem. 2013, 15, 152159.
[14] W. Xue, Y. Song, Y. J. Wang, D. D. Wang, F. Li, Catal. Commun. 2009,
11, 2933.
[15] S. U. Nandanwar, A. A. Dabbawala, M. Chakraborty, H. C. Bajaj, S.
Mukhopadhyay, K. T. Shenoy, Res. Chem. Intermed. 2016, 42,
15571569.
[16] S. J. Bao, S. N. Huang, Y. Liu, Y. R. Hu, W. P. Wang, M. F. Ji, H. L. Li,
N. X. Zhang, C. Z. Song, S. F. Duan, Nanoscale 2017, 9, 72847296.
[17] P. S. Zangabad, M. Karimi, F. Mehdizadeh, H. Malekzad, A. Ghasemi,
S. Bahrami, H. Zare, M. Moghoofei, A. Hekmatmanesh, M. R. Hamblin,
Nanoscale 2017, 9, 13561392.
Partial hydrogenation of benzene was conducted in a 500 ml Hastelloy
autoclave stirred mechanically. After charging 1.0 g of catalyst, 100 ml of
H2O, 2.0 g of ZnSO47H2O, and 50 ml of benzene, the autoclave was
sealed and purged with H2 several times to expel air. The reaction
conditions were the temperture of 413 K, the H2 pressure of 5.0 MPa,
and the stirring rate of 1200 rpm, which are typical for this
reaction.[3,4,715] During the reaction, a small aliquot of the sample was
discharged from the autoclave at intervals and analyzed on a GC122 gas
chromatograph fitted with a thermal conductivity detector (TCD) and a
PEG-20M stainless steel packed column. The catalysts were evaluated
at least in duplicate, and the results from replicate runs agreed to within
2%.
[18] J. Reguera, J. Langer, D. J. de Aberasturi, L. M. Liz-Marzán, Chem.
Soc. Rev. 2017, 46, 38663885.
[19] A. G. M. da Silva, T. S. Rodrigues, S. J. Haigh, P. H. C. Camargo,
Chem. Commun. 2017, 53, 71357148.
[20] D. P. Liu, W. Li, X. L. Feng, Y. Zhang, Chem. Sci. 2015, 6, 70157019.
[21] S. Chen, H. Y. Zhang, L. Y. Wu, Y. F. Zhao, C. L. Huang, M. F. Ge, Z.
M. Liu, J. Mater. Chem. 2012, 22, 91179122.
[22] Z. J. Wu, Y. Z. Mao, X. X. Wang, M. H. Zhang, Green Chem. 2011, 13,
13111316.
[23] S. Y. Song, X. C. Liu, J. Q. Li, J. Pan, F. Wang, Y. Xing, X. Wang, X. G.
Liu, H. J. Zhang, Adv. Mater. 2017, 29, doi: 10.1002/adma.201700495.
[24] X. H. Xia, Y. Wang, A. Ruditskiy, Y. N. Xia, Adv. Mater. 2013, 25,
63136333.
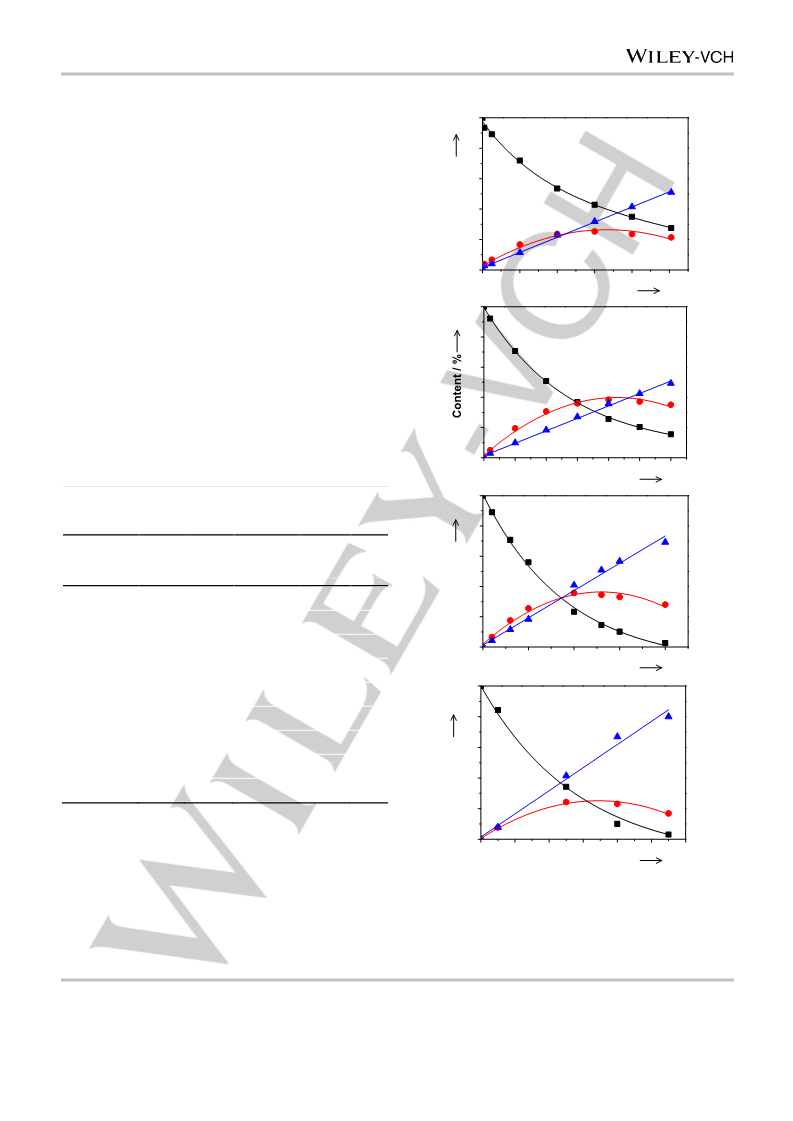
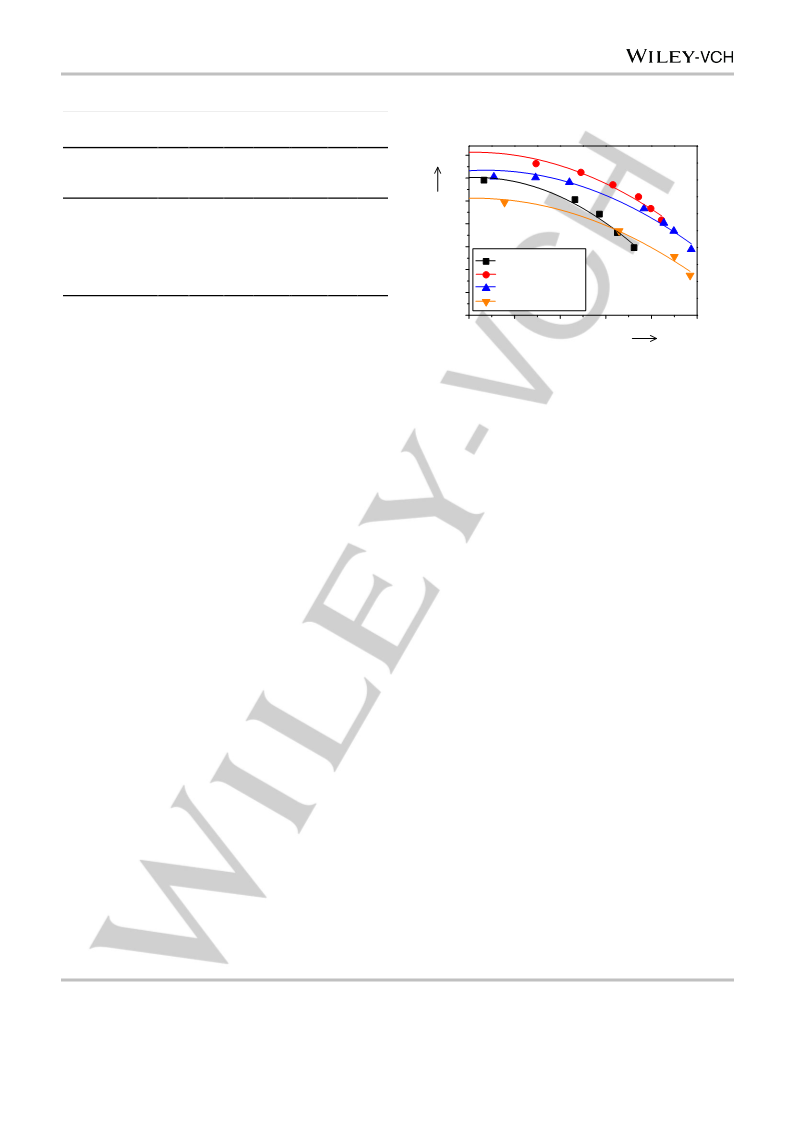
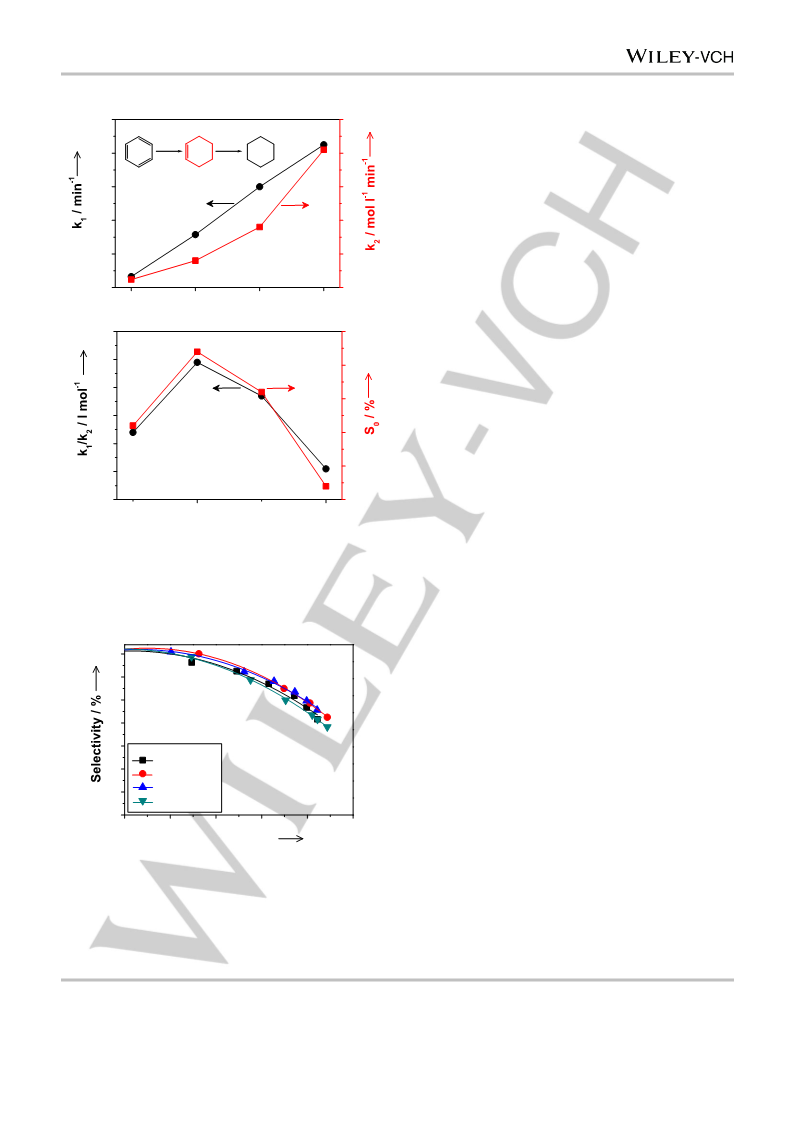
The TOF of benzene and the S0 of cyclohexene were used to denote
the activity and selectivity of the catalyst, respectively. For the calculation
of the TOF, the r0, i.e., the moles of benzene converted per gram of the
catalyst per minute at zero reaction time (t), was obtained first by
referring to the procedures proposed previously.[36] To figure out r0, a
polynomial equation was used to fit the benzene content–t curve. Then,
the equation was differentiated, and r0 was obtained by substituting zero
for t. The TOF was calculated using the equation of TOF = r0 MRu/(DRu
W),[30] where MRu was the molar mass of Ru, and W was the loading of
Ru. The S0 was acquired through the extrapolation of the fitted
cyclohexene selectivity–t curve to t of zero.
This article is protected by copyright. All rights reserved.

 Zhou, Gongbing
Zhou, Gongbing
 Wang, Hao
Wang, Hao
 Tian, Jing
Tian, Jing
 Pei, Yan
Pei, Yan
 Fan, Kangnian
Fan, Kangnian
 Qiao, Minghua
Qiao, Minghua
 Sun, Bin
Sun, Bin
 Zong, Baoning
Zong, Baoning